Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Tomo 53(2). ISSN (en línea) 1853-8665. Año 2021.
Original article
Effect of thermosonication on enzymatic oxidation and physicochemical properties of soursop (Annona muricata) pulp
Efecto de la termosonicación en la oxidación enzimática, y las propiedades físico-químicas de la pulpa de guanábana (Annona muricata)
Victor Manuel Gelvez Ordóñez 1,
Ivan Daniel López Castilla 2,
Luis Eduardo Ordoñez-Santos 3*
1 Universidad de Pamplona. Facultad de Ingenierías y Arquitectura. Departamento de alimentos. Villa del Rosario. Norte de Santander. Colombia.
2 Universidad de Pamplona. Semillero de Innovaciones Alimentarias - SIAL. Extensión Villa del Rosario. Colombia.
3 Universidad Nacional de Colombia. Facultad de Ingeniería y Administración. Departamento de Ingeniería. Colombia.
* leordonezs@unal.edu.co
Recepción: 27/05/2020
Aceptación: 03/06/2021
Abstract
Soursop is an exotic tropical fruit with marked commercial importance. However, its post-harvest presents losses of around 30%. The objective of this research was to study the effect of thermosonication (TS) on physicochemical properties and enzymatic oxidation of soursop pulp. Four treatments were evaluated: control (0 min), ultrasound at 20 °C (US20), 40 °C (US40), and 60 °C (US60) for 25 minutes. Concentration of vitamin C, and peroxidase (POD) and polyphenoloxidase (PPO) inactivation in the soursop pulp, were assessed. The results indicate a loss of vitamin C of 2.63%; 13.16% and 28.95% for temperatures at 20, 40 and 60 °C, respectively. On the other hand, partial inactivation of 48.41%; 69.83% and 74.24% of POD, and 56.38%; 74.47% and 81.91% of PPO enzymes were achieved at temperatures of 20, 40 and 60 °C, respectively. Finally, it was concluded that ultrasound combined with heat (TS) can inactivate POD and PPO enzymes in the soursop pulp, achieving considerable retention of vitamin C.
Keywords: pH, Soluble solids total, Polyphenoloxidase, Peroxidase, Vitamin C
Resumen
La guanábana es una fruta tropical exótica tiene una marcada importancia comercial, sin embargo, presenta pérdidas de alrededor del 30% en poscosecha. El objetivo del trabajo fue estudiar el efecto de la termosonicación (TS) sobre propiedades fisicoquímicas y la oxidación enzimática de la pulpa de guanábana. Los tratamientos evaluados fueron control (0 min), ultrasonido a 20 °C (US20), a 40 °C (US40), y a 60 °C (US60) durante 25 minutos. Se evaluaron principalmente la concentración de vitamina C, la inactivación de la peroxidasa (POD) y la polifenoloxidasa (PPO) en la pulpa de guanábana. Los resultados indican que se observó una pérdida de vitamina C en un 2,63; 13,16 y 28,95% para temperaturas a 20, 40 y 60 °C, respectivamente. Por otro lado, una inactivación parcial de las enzimas, POD, en 48,41; 69,83 y 74,24% y para PPO en 56,38; el 74,47 y el 81,91% se logró a temperaturas de 20, 40 y 60 ° C, respectivamente. Finalmente, se demostró que el ultrasonido combinado con calor (TS) inactivan en un gran porcentaje las enzimas POD y PPO en la pulpa de guanábana logrando importantes niveles de retención de la vitamina C.
Palabras clave: pH, sólidos solubles totales, polifeniloxidasa, peroxidase, vitamina C
Introduction
Quality of minimally processed fruits is widely valued by consumers. During elaboration, the enzymes Peroxidase (POD) and Polyphenoloxidase (PPO) trigger enzymatic processes affecting product natural color. According to Raimbault et al. (2010) these two enzymes produce quinones after oxidation of phenolic decolorizing compounds. In order to increase shelf life and improve product appearance, inactivation of these two enzymes must be total or partial (>70%). According to Soysal (2008) and Goyeneche et al. (2013) fruit and vegetable products subjected to thermal treatments at 60 and 95 °C stop enzymatic browning after protein inactivation, whereas Castro et al. (2008) reported that along with inactivation, it may also degrade nutrients such as vitamin C. In this sense, the demand for fresh products has prompted research to find non-thermal treatments such as ultrasound (US), an inaudible acoustic wave frequency higher than 20kHz (33). Islam et al. (2014) and Abid et al. (2014) evaluated the effects of US enzymatic inactivation and indicated that acoustic waves alter enzymatic tertiary structure leading to biological activity loss. In another work, Sulaiman et al. (2015) studied the effect of thermal US on PPO inactivation observing an increase in the inactivation rate after 10 minutes of US. Jang and Moon (2011) studied the effects of US and ascorbic acid on PPO and POD activity in fresh apples during storage. Liu et al. (2017) studied the effect of US on PPO in eastern sweet melon, reporting that high intensity US generated enzymatic conformational changes, causing inactivation.
Soursop is an exotic fruit with high levels of bioactive compounds, and great agro-industrial potential given its high recovery yields and excellent sensory characteristics (3). However, as stated, this fruit is susceptible to enzymatic browning (6). In Colombia, browning is one main rejection factor, generating losses of around 30% in post-harvest handling of this fruit. Therefore, research in this matter is required. The objective of this work was to study the effect of thermosonication (TS) on the physicochemical properties and enzymatic oxidation of soursop pulp.
Materials and methods
Chemicals
Phosphate buffer (pH 7.5), polyvinylpolypyrrolidone (PVPP), triton X-100, potassium iodide [90%], and hydrochloric acid [37%] (V/V) were obtained from Merck. Guaiacol [98%], hydrogen peroxide [30%], catechol [99%] and Arsenic (III) oxide [99.995%] were purchased from Sigma–Aldrich.
Pulp preparation
Five kg of fresh ripen soursop (Annona muricata) were purchased from a local supermarket in the city of Cúcuta, Colombia. These fruits were immediately taken to the Research Laboratory of the Food Innovations Group of the University of Pamplona, Villa del Rosario, Colombia. After peeling, the seeds were manually removed. Then, the pulp was vacuum packed (95%) and stored under refrigeration (4 °C) until further processing or analysis.
Treatment with thermosonication (TS)
Fruit pulp was divided in four samples, control (0 min), ultrasonic at 20 °C (US20), ultrasonic at 40 °C (US40), and ultrasonic at 60 °C (US60). Pulp samples (150 g) were vacuum-packed at 95%, and treated in an ultrasonic bath system (Elmasonic E equipment Model: E60H), with 40 kHz frequency and maximum ultrasonic power of 100 W. Internal dimension: 300 mm x 151 mm x 150 mm. All treatments were carried out in the dark, immediately cooled by immersion in an ice water bath and stored at 4 °C until further analysis.
Physicochemical properties
Total soluble solids (SST) were measured by refractometry (ATC-FG113), and pH was measured using an Orion 210ª electronic pH meter previously calibrated following AOAC (1990). Vitamin C was quantified by titration of ascorbic acid according to Santos and Daghastanli (2001).
Peroxidase (POD) extraction and activity
Enzymatic extraction was carried out after Silva et al. (2015) modified. Twenty g of soursop pulp were mixed with 100 mL distilled water, and centrifuged at 10000 g, 7 °C, for 10 minutes. Then, 1.5 ml of the extract was mixed with 320 ųL of guaiacol 5% (V / V) and 160 ųL of H2O2 [0.147 M] (V / V). Absorbance was recorded using a spectrophotometer (ThermoSpectronic Genesys 20) at 470 nm, every 20 seconds for 4 minutes, at 25 °C. Absorbance was related as a function of time. Enzymatic activity was obtained by equation 1:
Eq. 1 AE/e = (M) / (Ke),
where:
AE / e = enzymatic activity of the treated or not treated sample,
M = rate of reaction progress (ΔAbs / Δtim), and Ke: extinction coefficient.
Residual POD activity (%), resulted from equation 2:
Eq. 2 AER (%) = ((ΔE/Ao)*100),
where:
ΔE = enzymatic activity of the thermosonicated sample,
Ao = enzymatic activity of the control sample (untreated),
AER = residual enzymatic activity.
Oxidation caused by hydrogen peroxide reduces Guayacol to Tetraguayacol, generating brown coloration. According to Liburdi et al. (2019), the molar extinction coefficient of tetraguayacol is 26.6 mM-1cm-1 for an absorbance of 470 nm. This value is used to calculate enzymatic activity of POD after treatments with TS using eq. 1 and 2.
Polyphenoloxidase (PPO) extraction and activity
Enzymatic extraction was carried out according to Campo and Gélvez (2011) with modifications. Ten g of soursop pulp were mixed with 25 mL of 0.2M phosphate buffer solution (pH 6.5) containing 4% polyvinylpolypyrrolidone (PVPP) and 1% Triton X-100, homogenized for 3 minutes and then centrifuged (10000 rpm / 30 min at 4 °C). Seventy-five μL of the extract were extracted and mixed with 3 mL of 0.007M catechol in 0.05M phosphate buffer (pH 6.5) as enzyme substrate. Absorbance was recorded using a spectrophotometer (ThermoSpectronic Genesys 20) at 420 nm, every 20 seconds, for 4 minutes, at 25 °C. Absorbance was related as a function of time. This value is used to calculate enzymatic activity of PPO after treatments with TS using eq.1 and 2, calculating residual activity of PPO (%). According to Palmer (1963), Catecol is reduced to Benzoquinone by O2 with an extinction coefficient of 2.6 M-1cm-1 at 420 nm.
Statistical analysis
Results are expressed as means ± standard deviations. Treatment effects (control, US20, US40, and US60) on individual dependent variables (PPO and POD activity, Vitamin C, total solids and pH) were evaluated by one-way ANOVA. Significant differences between means were determined by the Tukey pairwise comparison test at a significance level of P < 0.05. Statistical analyses were conducted using the SPSS 18 program for Windows. All treatments were carried out in triplicate.
Results and discussion
Analysis of physicochemical properties
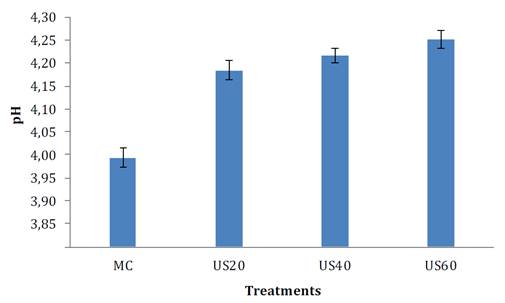
Figure 1 shows pH values of the treated and untreated samples. This variable increased significantly (p <0.05) after TS and temperature due to cavitation, after breaking pulp hydroxyl bonds (OH-). These results were also reported by Avalos et al. (2015) and Campo et al. (2011), indicating that with US in exposure time range of 15 and 30 minutes, a slight increase in cherimoya pulp pH, occurred. In contrast, Porras et al. (2011), reported a decrease in pH in mango pulp for times greater than 30 minutes. In the present study, pH reduces PPO and POD enzymatic actions, allowing pulp stability. In addition to the inactivation of these enzymes, the OH- group is associated with antimicrobial and antioxidant activities that allow preserving and improving the shelf life of food (2). According to Avalos et al. (2015), this slight increase is related to the release of volatile aromatic substances given cavitation and temperature effects in the TS treatment, favoring organoleptic properties of soursop pulp.
MC= control samples, US20= ultrasonic at 20 °C, US40= ultrasonic at 40 °C, and US60= ultrasonic at 60 °C.
MC= muestra control, US20 = ultrasónico a 20 °C, US40 = ultrasónico a 40 °C y US60 = ultrasónico a 60 °C.
Figure 1. Soursop pulp pH treated and untreated with thermosonication at different temperatures.
Figura 1. Valores de pH de pulpa de guanábana tratados y no tratados con termosonicación a diferentes temperaturas.
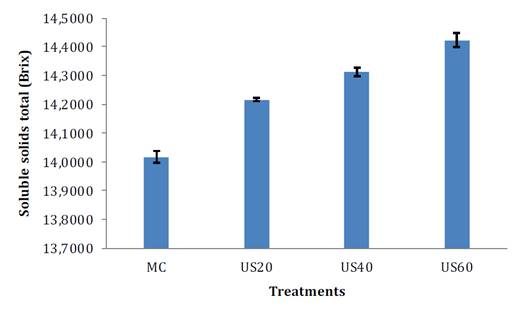
Figure 2 shows SST (°Brix) of the treated and untreated samples. SST increased significantly (p <0.05) after TS and temperature, as observed for mango pulp, apple juice and cherimoya pulp, (1, 5, 25). During ultrasound treatments, polysaccharide diffusion coefficient and solubility increased, mainly due to cavitation in the plant matrix, fracturing cell walls (32). An alternative explanation states that increasing SST (°Brix) is given by micro evaporation of water during ultrasonic cavitation (17, 25). According to Makino et al. (1983), cavitation causes a dissociation of water molecules into hydrogen ions (H+) and hydroxyl groups (OH-) increasing pulp ° Brix. Figure 3 shows vitamin C values of the treated and untreated samples.
MC= control samples, US20= ultrasonic at 20 °C, US40= ultrasonic at 40 °C, and US60= ultrasonic at 60 °C.
MC= muestra control, US20 = ultrasónico a 20 °C, US40 = ultrasónico a 40 °C y US60 = ultrasónico a 60 °C.
Figure 2. Total soluble solids of soursop pulp treated and not treated with thermosonication at different temperatures.
Figura 2. Valores de sólidos solubles totales de pulpa de guanábana tratada y no tratada con termosonicación a diferentes temperaturas.
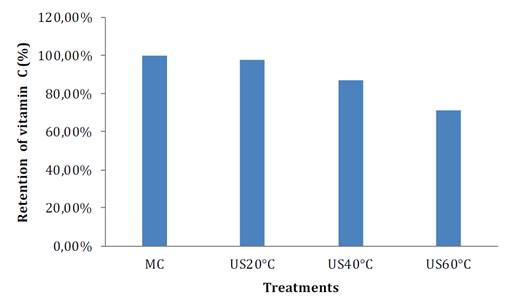
MC= control samples, US20= ultrasonic at 20 °C, US40= ultrasonic at 40 °C, and US60= ultrasonic at 60 °C.
MC= muestra control, US20 = ultrasónico a 20 °C, US40 = ultrasónico a 40 °C y US60 = ultrasónico a 60 °C.
Figure 3. Vitamin C before and after thermosonication.
Figura 3. Vitamina C presente en la muestra antes y después del tratamiento con termosonicación.
Significant differences (p <0.05) in vitamin C retention among control sample and treatments were found with temperatures around 40 and 60 °C. The lowest vitamin C retention value (71.05%) occurred for TS around 60 °C, followed by TS at 40 °C with 86.84%. On the contrary, no significant differences could be found for TS at 20 °C. Our results agree with those reported by Dabir and Ananthanarayan (2018) in custard apple. These authors found no significant effect between treated and untreated samples with US at room temperatures. However, Abid et al. (2014) indicated that under TS, when temperature raised above 40 °C, a significant loss of vitamin C occurred. The synergistic effect of radical formation (OH-and H+) and temperature during thermosonication, triggers hydrolysis, decarboxylation, regrouping and cyclization reactions, initiating the formation of compounds like 2,3-diketogulonic, 3-hydroxy acids -2-pirone, and 2-furoic, losing nutritional value (23).
Effects of thermosonication on POD and PPO enzymatic activity in soursop
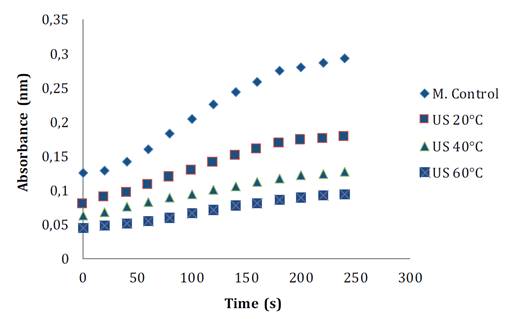
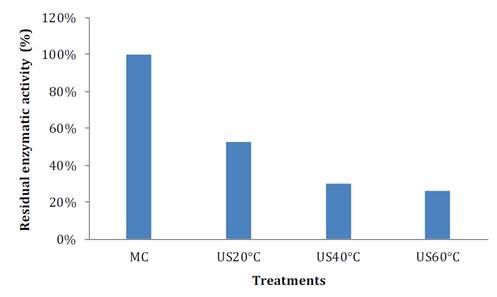
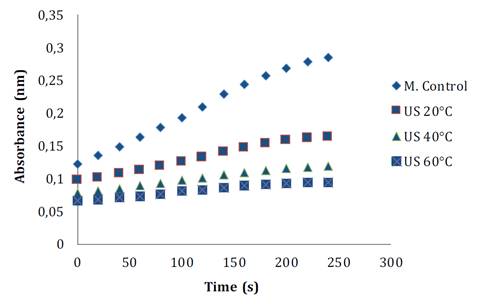
During fruit processing and storage, PPO and POD enzymes can affect product final quality. PPO is involved in the oxidation of polyphenols to o-quinones, which subsequently polymerize into undesirable brown, red, or black pigments in the presence of oxygen (18). These authors also comment that POD, an antioxidant enzyme, catalyzes the conversion of hydrogen peroxide to water using polyphenols as hydrogen donors. Figure 4 shows POD reaction before and after the treatment. Its reaction rate decreases with TS and temperature. POD residual enzymatic activity under TS around 20 °C, is 52.59% compared to the control sample, whereas when temperature is increased, only 30.17 and 25.86% are observed at 40 and 60 °C, respectively (figure 4A). Other studies also report a reduction in POD activity after ultrasound. In bayberry juice, residual activity levels resulted in between 9.49-73.14%, while in pear juice, inactivation levels reached between 4.30-43.2% (9, 27).
MC= control samples, US20 = ultrasonic at 20 °C, US40 = ultrasonic at 40 °C and US60 = ultrasonic at 60 °C.
MC=Muestra control, US20 = ultrasónico a 20 °C, US40 = ultrasónico a 40 °C y US60 = ultrasónico a 60 °C.
Figure 4. Progression curve of (Abs vs Time) soursop peroxidase reaction before and after TS treatments.
Figura 4. Curva de progreso de la reacción (Abs vs Time) de la peroxidasa de guanábana antes y después de los tratamientos con TS.
MC= control samples, US20= ultrasonic at 20 °C, US40= ultrasonic at 40 °C, and US60= ultrasonic at 60 °C.
MC= muestra control, US20 = ultrasónico a 20 °C, US40 = ultrasónico a 40 °C y US60 = ultrasónico a 60 °C.
Figure 4A. Residual enzymatic activity of peroxidase in soursop after thermosonication at different temperatures.
Figura 4A. Actividad enzimática residual de peroxidasa en guanábana después del tratamiento con termosonicación a diferentes temperaturas.
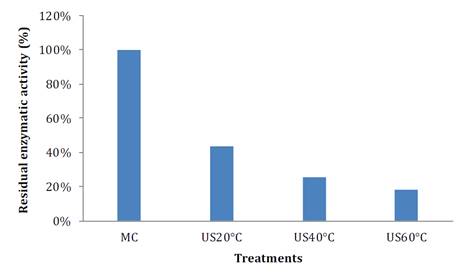
On the other hand, figure 5 shows PPO behaviour before and after the treatment. PPO reaction rate also decreased with TS and temperature. Its residual enzymatic activity with TS around 20 °C, was 43.62% compared to the control sample, whereas when temperature is increased, only 25.53 and 18.09% were observed for 40 and 60 ° C, respectively (figure 5A). Accordingly, in bayberry juice, registered residual activity levels reached between 0.08 and 53.23%, while in pear juice reported inactivation levels resulted between 1.91-89.33% (9, 27).
MC= control samples, US20= ultrasonic at 20 °C, US40= ultrasonic at 40 °C, and US60= ultrasonic at 60 °C.
MC= muestra control, US20 = ultrasónico a 20 °C, US40 = ultrasónico a 40 °C y US60 = ultrasónico a 60 °C.
Figure 5. Progression curve of the reaction (Abs vs Time) of soursop polyphenoloxidase before and after TS.
Figura 5. Curva de progreso de la reacción (Abs vs Tim) de la polifenoloxidasa de guanábana antes y después de los tratamientos con TS.
MC= control samples, US20= ultrasonic at 20 °C, US40= ultrasonic at 40 °C, and US60= ultrasonic at 60 °C.
MC= muestra control, US20 = ultrasónico a 20 °C, US40 = ultrasónico a 40 °C y US60 = ultrasónico a 60 °C.
Figure 5A. Residual enzymatic activity of polyphenoloxidase in soursop after thermosonication at different temperatures.
Figura 5A. Actividad enzimática residual de polifenoloxidasa en guanábana después del tratamiento con termosonicación a diferentes temperaturas.
According to Ercan and Soysal (2011) ultrasonic generated heat, and exposure time leading to increased sample temperatures, allow the reduction of POD and PPO activities. When US is combined with temperature, a synergic effect generates enzymatic dissociation of the prosthetic group, significantly decreasing (p<0.05) the content of the structure of the helix/alpha and causing conformational changes in the secondary and tertiary structures of POD and PPO after cavitation effect. This finally leads to partial or total protein denaturation (14, 20, 22).
Anaya-Esparza et al. (2017) reported that US generates micro-streaming phenomena that can interrupt polypeptide Van der Waals interactions and hydrogen bonds. In addition, free radicals generated during US can react with disulfide bonds, destabilizing the enzymatic conformation, or oxidizing amino acids related to enzymatic catalytic activity and stability, such as tryptophan, tyrosine, histidine, and cysteine. According to Dabir and Ananthanarayan (2018), complete inactivation of POD and PPO is achieved at 5 minutes under an ultrasonic power of 85 W and 90 W respectively, followed by 21.6% loss of vitamin C. In this study, TS at 40 °C resulted the most efficient treatment inactivating POD in 69.83% and PPO, in 74.47%, while retaining 86.84% vitamin C. On the other hand, TS and its lower temperatures (<60 °C), favoured vitamin C retention, in comparison with conventional thermal treatments using temperatures higher than 70 °C. According to Burdurlu et al. (2006), greater temperature decreases vitamin C.
Conclusions
TS treatment in soursop pulp (Annona muricata) affects its physicochemical properties, increases pH and soluble solids, while decreases vitamin C retention. The evaluated TS treatments inactivated POD and PPO enzymes in a range of 48.41-74.24% and 56.38-81.91%, respectively. Maximum POD (69.83%) and PPO (74.47%) inactivation were reached with 40 kHz and 40 °C TS, keeping vitamin C at 86.84%. Further research on POD and PPO inactivation in soursop is needed, Studies demonstrating that TS could be more efficient than blanching and bleaching, should be developed. Given that lower temperatures do not need? additional chemicals, this constitutes a product and environmentally friendly proposal.
1. Abid, M.; Jabbar, S.; Hu, B.; Hashim, M. M.; Wu, T.; Lei, S.; Khan, M. A.; Zeng, X. 2014. Thermosonication as a potential quality enhancement technique of apple juice. Ultrasonics Sonochemistry 21: 984-990.
2. Aguilar-Méndez, M. A.; Campos-Arias, M. P.; Quiroz-Reyes, C. N.; Ronquillo-de Jesús, E.; Cruz- Hernández, M. A. 2020. Fruit peels as sources of bioactive compounds with antioxidant and antimicrobial properties. Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina. 52(1): 360-371.
3. Anaya-Esparza, L. M.; Velázquez-Estrada, R. M.; Roig, A. X.; García, H. S.; Sayago-Ayerdi, S. G.; Montalvo González, E. 2017. Thermosonication: An alternative processing for fruit and vegetable juices. Trends in Food Science & Technology 61: 26-37.
4. AOAC. 1990. Official methods of analysis. 14th ed. p. 264-275. Gaithersburg, MD. USA: AOAC International.
5. Avalos, F. R.; Azahuanche, F. R. P.; Arquiño, M. J. O.; Jáuregui, G. B.; Tang, M. V.; Horna, A. C. F. 2015. Effect of exposition time to ultrasound on physicochemical, rheological, and microbiological characteristics of “chirimoya” Annona cherimola Mill. (Annonaceae) pulp. Arnaldoa 22: 367-380.
6. Bora, P. S.; Holschuh, H. J.; da Silva Vasconcelos, M. A. 2004. Characterization of polyphenol oxidase of soursop (Annona muricata l.) fruit and a comparative study of its inhibition in enzyme extract and in pulp. CYTA Journal of Food 4: 267-273.
7. Burdurlu, H. S.; Koca, N.; Karadeniz, F. 2006. Degradation of vitamin C in citrus juice concentrates during storage. Journal of Food Engineering. 74: 211-216.
8. Campo Vera, Y.; Gélvez, M. V. 2011. Efecto de la termosonicaciòn sobre las propiedades fisicoquímicas del hongo comestible (Pleurotus ostreatus) fresco empacado al vacío. Bistua: Revista de La Facultad de Ciencias Básicas. 9(2): 55-63.
9. Cao, X.; Cai, C.; Wang, Y.; Zheng, X. 2018. The inactivation kinetics of polyphenol oxidase and peroxidase in bayberry juice during thermal and ultrasound treatments. Innovative food science & emerging technologies. 45: 169-178.
10. Castro, S. M.; Saraiva, J. A.; Lopes-da-Silva, J. A.; Delgadillo, I.; Van Loey, A.; Smout, C.; Hendrickx, M. 2008. Effect of thermal blanching and of high pressure treatments on sweet green and red bell pepper fruits (Capsicum annuum L.). Food Chemistry. 107: 1436-1449.
11. Dabir, M. P.; Ananthanarayan, L. 2018. Effect of Heat Processing and Ultrasonication Treatment on Custard Apple Peroxidase Activity and Vitamin C. International Journal of Food Engineering, 13(1). https://doi.org/10.1515/ijfe-2015-0209
12. Ercan, S. Ş.; Soysal, Ç. 2011. Effect of ultrasound and temperature on tomato peroxidase. Ultrasonics Sonochemistry. 18: 689-695.
13. Goyeneche, R.; Di Scala, K.; Roura, S. 2013. Biochemical characterization and thermal inactivation of polyphenol oxidase from radish (Raphanus sativus var. sativus). LWT-Food Science and Technology. 54: 57-62.
14. Huang, N.; Cheng, X.; Hu, W.; Pan, S. 2015. Inactivation, aggregation, secondary and tertiary structural changes of germin-like protein in Satsuma mandarine with high polyphenol oxidase activity induced by ultrasonic processing. Biophysical Chemistry. 197: 18-24.
15. Islam, M. N.; Zhang, M.; Adhikari, B. 2014. The inactivation of enzymes by ultrasound-a review of potential mechanisms. Food Reviews International. 30: 1-21.
16. Jang, J. H.; Moon, K. D. 2011. Inhibition of polyphenol oxidase and peroxidase activities on fresh-cut apple by simultaneous treatment of ultrasound and ascorbic acid. Food Chemistry. 124: 444-449.
17. Knorr, D.; Zenker, M.; Heinz, V.; Lee, D. U. 2004. Applications and potential of ultrasonics in food processing. Trends in Food Science & Technology. 15: 261-266.
18. Lee, J. H.; Kasote, D. M.; Jayaprakasha, G. K.; Avila, C. A.; Crosby, K. M.; Patil, B. S. 2021. Effect of production system and inhibitory potential of aroma volatiles on polyphenol oxidase and peroxidase activity in tomatoes. Journal of the Science of Food and Agriculture. 101(1): 307-314.
19. Liburdi, K.; Benucci, I.; Esti, M. 2019. Effect of microwave power and blanching time in relation to different geometric shapes of vegetables. LWT - Food Science and Technology. 99: 497-504.
20. Liu, S.; Liu, Y.; Huang, X.; Yang, W.; Hu, W.; Pan, S. 2017. Effect of ultrasonic processing on the changes in activity, aggregation and the secondary and tertiary structure of polyphenol oxidase in oriental sweet melon (Cucumis melo var. makuwa Makino). Journal of the Science of Food and Agriculture. 97: 1326-1334.
21. Makino, K.; Mossoba, M. M.; Riesz, P. 1983. Chemical effects of ultrasound on aqueous solutions. Formation of hydroxyl radicals and hydrogen atoms. The Journal of Physical Chemistry. 87: 1369-1377.
22. O’donnell, C. P.; Tiwari, B. K.; Bourke, P.; Cullen, P. J. 2010. Effect of ultrasonic processing on food enzymes of industrial importance. Trends in Food Science & Technology. 21(7): 358-367.
23. Ordóñez-Santos, L. E.; Martínez-Girón, J.; Arias-Jaramillo, M. E. 2017. Effect of ultrasound treatment on visual color, vitamin C, total phenols, and carotenoids content in Cape gooseberry juice. Food Chemistry. 233: 96-100.
24. Palmer, J. K. 1963. Banana polyphenoloxidase. Preparation and properties. Plant Physiology 38: 508-513.
25. Porras, O.; González, G.; Castellanos, A.; Ballesteros, J.; Pacheco, M. 2011. Effect of the application of ultrasound waves on the physicochemical, rheological and microbiological properties of mango pulp (Mangifera indica l.) Common variety. Alimentos Hoy. 20(23): 52-77.
26. Raimbault, A. K.; Marie-Alphonsine, P. A.; Horry, J. P.; Francois-Haugrin, M.; Romuald, K.; Soler, A. 2010. Polyphenol oxidase and peroxidase expression in four pineapple varieties (Ananas comosus L.) after a chilling injury. Journal of Agricultural and Food Chemistry. 59: 342-348.
27. Saeeduddin, M.; Abid, M.; Jabbar, S.; Wu, T.; Hashim, M. M.; Awad, F. N.; Hu, B.; Lei, S.; Zeng, X. 2015. Quality assessment of pear juice under ultrasound and commercial pasteurization processing conditions. LWT-Food Science and Technology. 64(1): 452-458.
28. Santos, H. L.; Daghastanli, K. R. P. 2001. Using a classical method of vitamin C quantification as a tool for discussion of its role in the body. Biochemistry and Molecular Biology Education. 29(3). 110-114.
29. Silva, L. C. A.; Almeida, P. S.; Rodrigues, S.; Fernandes, F. A. N. 2015. Inactivation of polyphenoloxidase and peroxidase in apple cubes and in apple juice subjected to high intensity power ultrasound processing. Journal of Food Processing and Preservation. 39: 2081-2087.
30. Soysal, Ç. 2008. Kinetics and thermal activation/inactivation of starking apple polyphenol oxidase. Journal of Food Processing and Preservation. 32: 1034-1046.
31. Sulaiman, A.; Soo, M. J.; Farid, M.; Silva, F. V. M. 2015. Thermosonication for polyphenoloxidase inactivation in fruits: Modeling the ultrasound and thermal kinetics in pear, apple and strawberry purees at different temperatures. Journal of Food Engineering. 165: 133-140.
32. Tahmouzi, S. 2014. Optimization of polysaccharides from Zagros oak leaf using RSM: antioxidant and antimicrobial activities. Carbohydrate polymers. 106: 238-246.
33. Ulloa, J. A.; Ulloa, P. R.; Ramírez, J. C.R.; Rangel, B. E. U. 2013. Ultrasonido: aplicaciones en el campo de los alimentos. Revista Fuente Nueva Época Año. 4(14): 1-13.
Acknowledgements
This work was supported by Universidad de Pamplona, Norte de Santander, Colombia.