Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Tomo 53(2). ISSN (en línea) 1853-8665. Año 2021.
Original article
Implementation of ice plant (Mesembryanthemum crystallinum L.) production under semi-controlled conditions
Implementación de la producción de lechuga glacial (Mesembryanthemum crystallinum L.) bajo condiciones semicontroladas
María del Carmen Rodríguez-Hernández 1
1 Universidad de Alicante. Departamento de Ciencias de la Tierra y del Medio Ambiente. Alicante. Apdo. Correos 99, 03080. España.
Abstract
Ice plant (Mesembryanthemum crystallinum L.) is regarded as a drought and saline stress-tolerant plant with many biological properties, and especially valued in gourmet cuisine. The objective of this work was to find an optimum plant cultivation mode to produce edible parts under greenhouse conditions. Three soilless media were evaluated: peat, vermiculite and hydroponic culture. Peat pot culture yielded reduced biomass. Vermiculite and hydroponics, however, led optimum M. crystallinum growth. Plants grown in vermiculite and irrigated with nutrient solution presented a significantly higher yield. In fact, vermiculite-grown plants presented enhanced leaf area and leaf fresh weight as well as high foliar N, Mg, Mn, Fe, Na, chlorophyll and carotenoid concentrations. To conclude, greater succulence and Na concentration in edible parts of the vermiculite-grown glacier lettuce can offer consumers a more interesting taste, consistency and improved nutrient contents.
Keywords: Mesembryanthemum crystallinum; Hydroponic culture; Substrates; Pot culture.
Resumen
La lechuga glacial (Mesembryanthemum crystallinum L.) muestra tolerancia a los estreses salino e hídrico, y posee diversas actividades biológicas que han permitido su revalorización como planta comestible en la alta cocina. El objetivo del presente trabajo fue evaluar el óptimo modo de cultivo para la producción de hojas de M. crystallinum bajo condiciones de invernadero. Para ello, se compararon tres formas de cultivo sin suelo: turba, vermiculita y cultivo hidropónico. El crecimiento en maceta con turba no resultó ser adecuado para la lechuga glacial, provocando una baja producción de biomasa. Sin embargo, tanto el cultivo en vermiculita como hidropónico permitieron un óptimo crecimiento de M. crystallinum, mostrando una producción significativamente superior las plantas crecidas en vermiculita mediante riego con solución nutritiva. La lechuga glacial desarrollada en vermiculita incrementó su área y materia fresca foliar, junto con elevadas concentraciones de N, Mg, Mn, Fe, Na, clorofilas y carotenoides en hojas. Además, el aumento de la suculencia y la concentración de Na en las partes comestibles de las plantas cultivadas en vermiculita pueden ofrecer un sabor, consistencia y contenido en nutrientes de mayor interés para el consumidor.
Palabras clave: Mesembryanthemum crystallinum; Cultivo hidropónico; Sustratos; Cultivo en maceta.
Originales: Recepción: 22/06/2020
Aceptación: 12/10/2021
Introduction
Mesembryanthemum crystallinum, commonly known as ice plant or glacier lettuce, is regarded as a stress-tolerant plant. During the dry season and periods of water stress, the photosynthesis mode switches from C3 to Crassulacean Acid Metabolism (CAM) (9). This halophytic plant possesses specialised trichomes called epidermal bladder cells, which have various functions including: water storage, salt accumulation, protection from UV rays and a role in plant defence (7). When mature plants die, the stored salt is leached into the soil thereby dramatically increasing salinity, making it difficult for other native species to grow (10, 20, 32). However, due to its ability to accumulate salt, the plant has been employed for soil desalination and proposed for bioremediation (2). The ice plant can also rapidly absorb soil moisture and help to build up high nitrate levels (13).
The ice plant’s numerous biological properties include antioxidant activities, performed by betacyanin and other flavonoids, as well as antimicrobial activities (18, 25). Leaves and stems are used raw or cooked. With its succulent, mellow, slightly salty-tasting leaves, the species is considered a fashionable plant sold in delicatessen shops (16, 30). The product, however, is highly perishable. The wild nature of this species suggests it could be cultivated throughout the year, with little watering and care, based on a different maintenance approach (1). Therefore, the objective was to conduct a greenhouse production experiment and assess optimum ice plant growth conditions in soilless media.
Materials and methods
Plant material and growth conditions
Mesembryanthemum crystallinum seeds were collected from wild plants in Alicante (in South East Spain). They were surface disinfected with 0.5% NaClO for 2 h and pre-hydrated with aerated, distilled water for 22 h. Germination subsequently took place in vermiculite, hydrated with distilled water and maintained in a growth chamber at 24°C day/night (D/N) air temperature (T) and 70% D/N relative humidity (RH) (29). Chamber light conditions were 16 h light-8 h dark cycle with photosynthetically active radiation of 400 μmol m-2s-1, provided by a combination of fluorescent tubes (Philips TLD 36W/83, Germany and Silvana F36W/GRO, USA). After 20 days, seedlings were transferred to a greenhouse under semi-controlled conditions of T D/N: 25/18°C; RH D/N: 60/80% and received natural daylight (mean photosynthetic photon flux rate of 400 μmol m-2 s-1) (3) (Photo 1).
Photo 1: Experimental site and ice plants grown under different mode of cultivation.
Foto 1: Unidad de experimentación vegetal y plantas de lechuga glacial crecidas bajo diferentes modos de cultivo.
During the transplant, plants were divided into three homogeneous groups of 10 plants. Each group underwent different growth conditions: 1) seedlings transplanted to plastic peat pots (V=1 L) watered weekly with 300 mL demineralised water; 2) seedlings inserted into 1 L plastic vermiculite pots watered weekly with 300 mL Hoagland nutrient solution (17); 3) seedlings grown in hydroponic containers (V=1 L) with Hoagland aerated nutrient solution, which was replaced weekly. Hoagland solution has been widely used for glacier lettuce growth (3, 5, 24, 29). Peat was mainly composed of pine bark, with 86% of organic matter based on dry weight, 49 mS m-1 conductivity, pH 7 and grain size <10 mm. After 45 days, the plants were harvested and the different determinations were performed. At the end of the experiment, the electrical conductivity (EC) of the hydroponic, vermiculite and peat culture was 1.38, 0.57 and 0.63 mS cm-1, respectively.
Growth parameters
Plant dry weight (DW) was determined after oven drying at 80°C until constant weight. Shoot and root lengths were also measured. Leaf area was measured using the app “Easy Leaf Area Free” (12), while Specific leaf area (SLA) was calculated as the ratio of the leaf area to leaf dry weight.
Water status
Leaf relative water content (RWC) was calculated according to Weatherley’s method (1950), using the following equation:
RWC (%) = (FW-DW) / (TW-DW) x 100
where:
FW = fresh weight,
TW = turgid weight,
DW = dry weight of the tissue, respectively.
This determination was performed on expanded young leaves collected at noon. Foliar succulence was measured according to Atzori et al. (2017) as the ratio of leaf FW to leaf area.
Photosynthetic pigments
Foliar photosynthetic pigment concentration was determined in young, recently expanded leaves collected at noon, as described in Sesták et al. (1971). Samples (20 mg FW) were placed in 5 ml of 96% ethanol at 80° C for 10 minutes to extract the pigments. The absorbance of the extracts was spectrophotometrically measured and the equations reported by Lichtenthaler (1987) were used to calculate chlorophyll and carotenoid concentrations.
Mineral analysis
Leaf samples (0.5 g DW) were dry-ashed and dissolved in HCl in accordance with Duque (1971). Phosphorus, potassium, calcium, magnesium, manganese, zinc, iron and sodium concentrations were determined using a Perkin Elmer Optima 4300 inductively coupled plasma optical emission spectroscopy (ICP-OES) (Perkin Elmer, USA). The ICP-OES operating parameters were: radio frequency power 1300 W, nebulizer flow 0.85 L min-1, nebulizer pressure 206.84 kPa, auxiliary gas flow 0.2 L min-1, sample introduction 1 mL min-1 and three replicates per sample. Total nitrogen and carbon were quantified after leaf DW combustion (950°C) with pure oxygen using an elemental analyser with a thermal conductivity detector (TruSpec CN, Leco, USA).
Statistics
To compare all three treatments, a one-way analysis of variance (ANOVA) (SPSS v.26, IBM Corp., USA) was conducted. The means ± standard deviation (SD) were calculated. When the F-ratio was significant (p<0.05), Duncan’s multiple range test was applied. When only two treatments were compared, Student’s t-test was performed. Significance levels were always set at 5%.
Results
Growth parameters of Mesembryanthemum crystallinum showed significant differences according to the culture medium (Table 1).
Table 1: Growth parameters in ice plants grown under different mode of cultivation.
Tabla 1: Parámetros de crecimiento de lechuga glacial crecida bajo diferentes modos de cultivo.
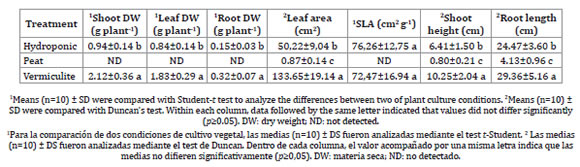
Plants in vermiculite presented greater shoot growth and leaf DW than those under hydroponic conditions, but the difference was especially notable compared to peat-grown plants. Vermiculite treatment also enhanced ice plant root length and biomass, while peat growth presented significantly low values. In fact, peat pot culture did not seem to be an adequate substrate to cultivate M. crystallinum. Despite the same environmental conditions and age, plants did not grow properly. Therefore, it was not possible to obtain sufficient plant matter from the peat treatment to perform further determinations such as: RWC, succulence, photosynthetic pigments or mineral analysis. On the other hand, vermiculite-grown plants exhibited a significant increase in leaf area compared to the hydroponic culture, despite their similar SLA values.
Regarding leaf water status, ice plant foliar RWC did not differ according to the cultivation mode (Table 2).
Table 2: Leaf fresh weight (FW), relative water content (RWC) and succulence in ice plants grown under different mode of cultivation.
Tabla 2: Materia fresca foliar, contenido hídrico relativo y suculencia de hojas de lechuga glacial crecida bajo diferentes modos de cultivo.
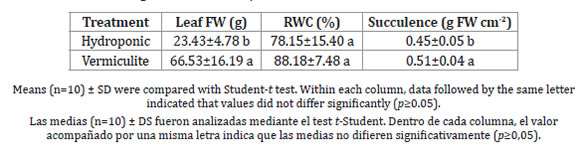
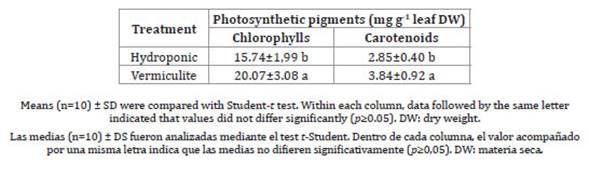
Vermiculite-grown plants, however, presented the highest edible leaf FW production values together with greater succulence. As for photosynthetic pigments (Table 3), both chlorophyll and carotenoid concentrations increased in vermiculite-grown plants.
Table 3: Leaf photosynthetic pigment concentration in ice plants grown under different mode of cultivation.
Tabla 3: Concentración foliar de pigmentos fotosintéticos de lechuga glacial crecida bajo diferentes modos de cultivo.
Foliar mineral concentration is summarised in Tables 4 and 5.
Table 4: Foliar concentrations of macronutrients in ice plants grown under different mode of cultivation.
Tabla 4: Concentración foliar de macronutrientes de lechuga glacial crecida bajo diferentes modos de cultivo.
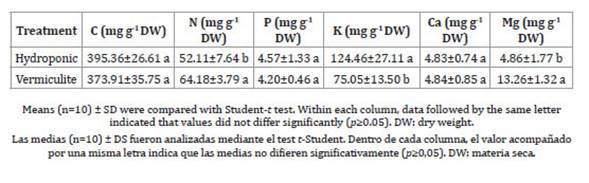
Table 5: Foliar concentrations of micronutrients and sodium in ice plants grown under different mode of cultivation.
Tabla 5: Concentración foliar de micronutrientes de lechuga glacial crecida bajo diferentes modos de cultivo.
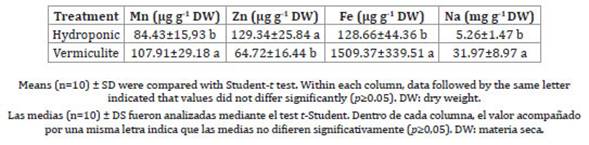
While C concentration did not differ among the plants grown in different media, foliar N concentration was higher in the ice plant cultivated in vermiculite (Table 4). Moreover, vermiculite-grown plants had significantly higher Mg (Table 4), Mn, Fe and Na (Table 5) concentrations compared to the hydroponic treatment. Nevertheless, K and Zn concentrations were greater in plants grown in hydroponics, while P and Ca contents were similar across all treatments.
Discussion
Peat is a widely used substrate for plant cultivation. In fact, Mesembryanthemum crystallinum has been successfully grown in peat-based substrate or mixed with vermiculite (3, 4, 8). However, our results showed that peat was not a suitable medium for ice plant pot-growing. Plant growth was very poor, making it impossible to determine many parameters and leading to significantly reduced shoot length, root length and leaf area. These results could be given by the fact that peat-grown plants were irrigated with demineralised water, while those grown in vermiculite and hydroponics received Hoagland nutrient solution. This may have generated an essential nutrient deficit in peat-grown plants, since the limited 1 L potting space increases nutrient needs (28). Moreover, according to Morales and Casanova (2015), peat is poorly drained. This probably leads to insufficient root oxygenation, causing growth limitations.
On the other hand, hydroponic and vermiculite cultures led to optimum M. crystallinum growth. In addition, vermiculite-grown plants irrigated with Hoagland nutrient solution presented a significantly greater yield. Vermiculite has a high cation exchange capacity (19), while in hydroponics, the nutrients are very accessible (22). It is worth noting that the hydroponic culture’s EC levels at the end of the experiment were more than two times greater than that of the other two substrates; despite this, EC levels were within an optimum range regarding root water and nutrient absorption (15). In addition, the nutrient solution was replaced every week in order to ensure good nutrient balance in the hydroponic culture.
Vermiculite-grown plants presented higher values across all growth parameters, except for SLA. The reason may be that the nutrient intake of vermiculite-grown ice plants is greater due to longer roots and higher root DM, which would improve water and nutrient acquisition and/or use efficiency (26). In fact, plants grown in vermiculite presented higher leaf N, Mg, Mn and Fe concentrations.
Regarding the water status, cultivation mode was not found to influence foliar RWC in any way. On the contrary, leaf succulence improved in vermiculite-grown plants, probably due to greater nutrient intake than in hydroponics. Increases in succulence, that is, water content per unit area, allows osmotic adjustment (14). According to our results, vermiculite-grown Mesembryanthemum leaves also exhibited higher Na concentrations. The latter increases the salty taste of leaves and is highly appreciated by consumers. In fact, leaf succulence and glistening bladder cells, in particular, provide the edible leaves with a taste, consistency and appearance that make ice plants particularly valued by consumers (6). In addition, Agarie et al. (2007) worked on an ice plant mutant lacking epidermal bladder cells (EBCs). The authors concluded that EBCs contribute to succulence by serving as a water storage reservoir. They also contribute to salt tolerance by maintaining ion sequestration and homeostasis within photosynthetically active tissues of M. crystallinum.
Significant differences were found regarding pigment concentrations, with enhanced levels in total chlorophylls and carotenoids of vermiculite-grown leaves. This fact could imply that ice plants grown in vermiculite have a greater photosynthetic capacity, together with a greater allocation of energy in thermal dissipation over photochemistry (6).
Conclusions
Clear evidence was found that vermiculite pot culture irrigated with nutrient solution allows obtaining M. crystallinum yield for edible purposes. Under greenhouse conditions, this cultivation mode leads to greater biomass - including the production of edible fresh leaves - than hydroponics or peat culture. In addition, increased leaf succulence and Na concentration of vermiculite-grown plants offer consumers a more interesting taste, consistency and appearance.
Funding
This work was financed by the Valencian Community, Spain (GV/2018/068).
Acknowledgements
The authors wish to thank to Susana Carrión and Mar Benavent for their help with biomass analysis and laboratory determinations.
References
1. Abdalla, M. A.; Hanan, S.; Maksoud, A.; Shanan, N. T. 2015. Evaluation of the growth behavior of ice plant (Mesembryanthemum crystallinum L.) grown under different habitats in Egypt. World Journal of Agricultural Sciences. 11(6): 391-400.
2. Abd El-Gawad, A. M.; Shehata, H. S. 2014. Ecology and development of Mesembryanthemum crystallinum L. in the deltaic Mediterranean coast of Egypt. Egyptian Journal of Basic and Applied Science. 1(1): 29-37.
3. Agarie, S.; Shimoda, T.; Shimizu, Y.; Baumann, K.; Sunagawa, H.; Kondo, A.; Ueno, O.; Nakahara, T.; Nose, A.; Cushman, J. 2007. Salt tolerance, salt accumulation, and ionic homeostasis in an epidermal bladder-cell-less mutant of the common ice plant Mesembryanthemum crystallinum. Journal of Experimental Botany. 58: 1957-1967.
4. Agarie, S.; Kawaguchi, A.; Kodera, A.; Sunagawa, H.; Kojima, H; Nose, A.; Nakahara, T. 2009. Potential of the common ice plant, Mesembryanthemum crystallinum as a new high-functional food as evaluated by polyol accumulation. Plant Production Science. 12(1): 37- 46.
5. Amari, A.; Ghnayaa, T.; Debez, A.; Taamali, M.; Youssef, N.; Lucchini, G.; Sacchi, G.; Abdelly, C. 2014. Comparative Ni tolerance and accumulation potentials between Mesembryanthemum crystallinum (halophyte) and Brassica juncea: Metal accumulation, nutrient status and photosynthetic activity. Journal of Plant Physiology. 171: 1634-1644.
6. Atzori, G.; De Vos, A. C.; van Rijsselbergue, M.; Vignolini, P.; Rozema, J.; Mancuso, S.; van Bodegom, P. M. 2017. Effects of increased seawater salinity irrigation on growth and quality of the edible halophyte Mesembryanthemum crystallinum L. under field conditions. Agricultural Water Management. 187: 37-46.
7. Barkla, B. J.; Vera-Estrella, R.; Pantoja, O. 2012. Protein profiling of epidermal bladder cells from the halophyte Mesembryanthemum crystallinum. Proteomics. 12(18): 2862-2865.
8. Barkla, B. J.; Vera-Estrella, R. 2015. Single cell-type comparative metabolomics of epidermal bladder cells from the halophyte Mesembryanthemum crystallinum. Frontiers in Plant Science. 6: 435.
9. CSIRO. 2004. Mesembryanthemum crystallinum L., Australia: CSIRO. http://www.cpbr.gov.au/cpbr/WfHC/Mesembryanthemum/index.html (April 2020).
10. D’Antonio, C.; Meyerson, L. A. 2002. Exotic plant species as problems and solutions in ecological restoration: a synthesis. Restoration Ecology. 10(4): 703-713.
11. Duque, F. 1971. Joint determination of phosphorus, potassium, calcium, iron, manganese, copper and zinc in plants. Anales de Edafología y Agrobiología. 30: 207-229.
12. Easlon, H. M.; Bloom, A. J. 2014. Easy Leaf Area: Automated digital image analysis for rapid and accurate measurement of leaf area. Applications in Plant Sciences. 2(7): 1400033.
13. FloraBase. 2015. The Western Australian Flora. Western Australia, Australia: Department of Environment and Conservation. http://florabase.dec.wa.gov.au/ (Accessed April 2020)
14. Flowers, T. J.; Colmer, T. D. 2008. Salinity tolerance in halophytes. New Phytologist. 179: 945-963.
15. Gruda, N. 2009. Do soilless culture systems have an influence on product quality of vegetables? Journal of Applied Botany and Food Quality. 82: 141-147.
16. Herppich, W. B.; Huyskens-Keil, S. M.; Schreiner, M. 2008. Effects of saline irrigation on growth, physiology and quality of Mesembryanthemum crystallinum L., a rare vegetable crop. Journal of Applied Botany and Food Quality. 82: 47-54.
17. Hoagland, D. R.; Arnon, D. I. 1938. The water culture method for growing plants without soil. University of California. 31 p.
18. Ibtissem, B.; Abdelly, C.; Sfar, S. 2012. Antioxidant and antibacterial properties of Mesembryanthemum crystallinum and Carpobrotus edulis extracts. Advances in Chemical Engineering and Science. 2: 359e65.
19. Lara, A. 1999. Manejo de la solución nutritiva en la producción de tomate en hidroponía. Terra Latinoameriacana. 17(3): 221-229.
20. Libik, M.; Pater, B.; Elliot, S.; Slesak, I.; Miszalski, Z. 2004. Malate accumulation in different organs of Mesembryanthemum crystallinum L. following age-dependent or salinity-triggered CAM metabolism. Zeitschrift für Naturforschung. Section C. Biosciences. 59(3/4): 223-228.
21. Lichtenthaler, H. K. 1987. Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods in Enzymology. 148: 350-382.
22. López, P. P.; Cano, A.; Rodríguez, G. S.; Torres, N.; Rodríguez, S.; Rodríguez, R. 2011. Efecto de diferentes concentraciones de potasio y nitrógeno en la productividad de tomate en cultivo hidropónico. Tecnociencia. 5(2): 98-104.
23. Morales, E. R.; Casanova, F. 2015. Mezclas de sustratos orgánicos e inorgánicos, tamaño de partícula y proporción. Agronomía Mesoamericana. 26(2): 365-372.
24. Popova, O.; Ismailov, S. 2002. Salt-induced expression of NADP-dependent isocitrate dehydrogenase and ferredoxin-dependent glutamate synthase in Mesembryanthemum crystallinum. Planta. 215: 906-913.
25. Rahman, S. M. A.; Abd-Ellatif, S. A.; Deraz, S. F.; Khalil, A. A. 2011. Antibacterial activity of some wild medicinal plants collected western Mediterranean coast, Egypt: natural alternatives for infectious disease treatment. African Journal of Biotechnology. 10: 10733e43.
26. Rich, S. M.; Ludwig, M.; Pedersen, O.; Colmer, T. D. 2011. Aquatic adventitious roots of the wetland plant Meionectes brownie can photosynthesize: implications for root function during flooding. New Phytologist. 190(2): 311-319.
27. Sesták, Z.; Catský, J.; Jarvis, P. 1971. Plant Photosynthetic Production: Manual of Methods. Dr. W. Junk Publishers. The Hague, Netherlands. 818 p.
28. Terés, V. 2000. Riego en sustratos de cultivo. Horticultura. 147: 16-30.
29. Thomas, J.; Malick, F.; Endreszl, C.; Davies, E.; Murray, K. 1998. Distinct responses to copper stress in the halophyte Mesembryanthemum crystallinum. Physiologia plantarum. 102: 360-368.
30. Vogel, G. 1996. Handbuch des speziellen Gemüsebaues. Verlag Eugen Ulmer. Stuttgart. Germany.
31. Weatherley, P. E. 1950. Studies in the water relations of the cotton plant: The field measurements of water deficits in leaves. New Phytologist. 49: 81-89.
32. Zavaleta, E. S.; Hobbs, R. J.; Mooney, H. A. 2001. Viewing invasive species removal in a wholeecosystem context. Trends in Ecology & Evolution. 16(8): 454-459.
