Revista de la Facultad de Ciencias
Agrarias. Universidad Nacional de Cuyo. Tomo 55(1). ISSN (en línea) 1853-8665.
Año 2023.
Original article
Quality of Tanzania grass (Panicum maximum)
haylage in relation to plant dry matter content
Calidad del pasto Tanzania (Panicum maximum) almacenada
como henolaje según la materia seca de la planta
Ricardo Loiola Edvan 1
Francigeferson Linhares
dos Santos 2
Romilda Rodrigues do Nascimento
1
Dhiéssica Morgana Alves Barros
1
Edson Mauro Santos 3
Felipe Luênio de Azevedo 2
Lucas de Sousa Barros 1
Keuven dos Santos Nascimento
1
Tairon Pannunzio
Dias-Silva 1*
Marcos Jácome de Araújo 1
1 Federal University of Piauí. Department of Animal
production. Rodovia Municipal Bom
Jesus-Viana. km 01. Planalto Horizonte. 64900000. Bom Jesus. Piauí. Brazil.
2 Federal University of Campina Grande. Health and Technology
Center. Avenida Universitária. s/n - Jatobá. 58708110. Patos. Paraíba. Brazil.
3 Federal University of Paraíba. Department of Animal production.
Avenida
Universitária. s/n
58397000. Areia. Paraiba. Brazil.
Abstract
This study aimed to evaluate the quality of Tanzania grass (Panicum
maximum) haylage with varying contents of dry matter (DM) and stored for 90
days. The quality of this grass was evaluated through the lens of a variety of
physiochemical properties (e.g., chemical composition, aerobic
stability, pH, microbial profile, etc.). A completely randomized design was
used with four treatments (in natura, 400, 500, and 600 g kg-1
DM) and five replicates. Treatment with 600 g kg-1
DM yielded the highest DM haylage (p < 0.01) and
soluble carbohydrate content (p < 0.01). Treatment in natura resulted
in the highest O2 concentration inside the bales
(p < 0.01), whereas treatments with 500 and 600 g kg-1 DM resulted in the highest CO2
values. The highest acetic acid concentrations of 36.4 ± 1.6,
38.2±1.6, and 48.9 ± 1.6 g kg-1 DM (p < 0.01) were
observed post the in natura, 500 g kg-1 DM, and 600 g kg-1
DM treatments, respectively. Treatment with 600 g kg-1
DM yielded the highest pH value at hour zero (p <
0.01). Tanzania grass with 500 and 600 g kg-1 DM produced the highest
quality haylage.
Keywords: Conservation;
Haylage; Grasses; Microbiology; Moisture;
Tanzania grass.
Resumen
El objetivo fue evaluar la
calidad del henolaje del pasto Tanzania (Panicum maximum) con diferentes
contenidos de materia seca y un almacenamiento de 90 días, a través de la
composición química, cuantificación de gases, ácidos grasos volátiles, perfil
microbiológico, estabilidad aeróbica, pH y nitrógeno amoniacal. El diseño
experimental utilizado fue completamente al azar con cuatro tratamientos y
cinco repeticiones. Los tratamientos consistían de cuatro contenidos de materia
seca (MS) de la planta en el momento de la producción del henolaje del pasto
Tanzania siendo: en materia fresca (sin deshidratación), 400, 500 y 600 g kg-1
de MS (deshidratados en pleno sol). El tratamiento con 600 g kg-1
de MS de la planta proporcionó la mayor estimación (p <
0,01) de MS en el henolaje con 581,6 ± 15,4 g/kg, y el mayor (p <
0,01) contenido de carbohidratos solubles con 45,4 ± 1,24 g/kg MS. Después de
90 días de almacenamiento, el tratamiento en materia fresca presentó mayores (p
< 0,01) cantidades de O2 en el interior de los fardos.
En relación al CO2 los
mayores índices fueron observados para los tratamientos con 500 y 600 g kg-1
de MS. También se observó el mayor (p < 0,01) contenido
de ácido acético en los tratamientos materia fresca y con 500 y 600 g kg-1
de MS de la planta, con 36,4 ± 1,6, 38,2 ± 1,6 y 48,9 ± 1,6 g kg-1
de MS, respectivamente. Para el ácido butírico se obtuvo la mejor
(p < 0,01) valoración de 27,0 ± 0,5 g kg-1 de MS en el tratamiento en
materia fresca. El tratamiento con 600 g kg-1 de MS mostró mayor (p <
0,01) valor de pH en la hora cero con 6,36 ± 0,03. El tratamiento en materia
fresca presentó mayor valor de N-NH3 en la hora cero de exposición
al aire con 4,65 ± 0,12. El pasto Tanzania con 500 y 600 g kg-1 de MS, presenta el henolaje de
mejor calidad.
Palabras clave: Conservación; Henolaje; Gramíneas; Microbiología; Humedad; Pasto Tanzania.
Originales: Recepción: 25/10/2020
Aceptación:
13/04/2023
Introduction
The storage of forage plants in the form of haylage is in line with the
sustainable use of leguminous and grass forage species. For example, oats (Avena
sativa) and ryegrass (Lolium multiflorum) are suitable for the
production of haylage in temperate regions, whereas species in the genera Brachiaria,
Cynodon, Panicum, and Pennisetum are better suited for the
production of haylage in tropical regions (27). Tanzania grass (Panicum maximum) has shown great potential
within the context of haylage production, as it has a high yield, a large
number of leaves, and a high nutritional value (9, 54).
Haylage can be defined as stored pre-dried forage with a dry matter (DM)
content of approximately 400 to 800 g/kg (7, 43).
It is stored in the form of bales wrapped in a plastic cover, providing ideal
conditions for the growth of lactic acid bacteria (LAB) that are beneficial for
the conservation and storage of forage. This forage would then be used as
animal feed; this is especially important when resources are scarce (e.g.,
during droughts) (22).
The preservation of grass in the form of haylage is an option for forage
grasses with high moisture content because dehydration of the material
increases DM content, which reduces proteolysis, secondary fermentation, and pH
buffering in the stored material (23). The moisture content of the forage plant is one factor that influences
the microbial profile of the forage mass preserved by fermentation (61). When harvested, tropical grasses have a high moisture content
accompanied with low levels of soluble carbohydrates (CHO) (49), which favors the occurrence of undesirable fermentation as the grass
is preserved.
An alternative to adjusting the DM content in tropical grasses is
dehydration in the field after cutting (10). This process increases the DM content of the forage mass, facilitating
the preparation of the material for undergoing preservation via fermentation.
The DM percentage of haylage influences the quality of the stored material (40).
No exact recommendations are available for the DM
content of tropical grasses for conservation as haylage, and no studies have
been conducted within the context of determining the DM content of Tanzania
grass. Therefore, this study aimed to evaluate the quality of Tanzania grass
haylage stored with different DM contents based on its chemical composition,
gas quantification, volatile fatty acids, microbiological profile, and aerobic
stability.
Material and methods
Study Area
A pasture area established in 2013 was used for haylage production. The
study area is in Alvorada do Gurgueia, Piauí, Brazil, at latitude 08°25’28”
South, longitude 43°46’38” West, and an altitude of 281 m. According to the
Köppen classification (1936), the climate of the region is classified as BSh,
hot semi-arid, with rainy summers and dry winters, as described Medeiros et al. (2013) and Alvares et al.(2013).
The area of the pasture was determined to be 0.5 hectares, and it had no
artificial irrigation systems. A standardization cut was made 30 cm from the
ground at the beginning of the experimental period for haylage production,
according to the recommendation of Braz et al. (2017). Fertilization
was performed according to the soil analysis and recommendations for highly
demanding species (30).
Experimental design
To assess the chemical composition and volatile fatty acids of the
Tanzania grass haylage, a completely randomized design with four treatments and
five replicates was adopted. The treatments consisted of four groups of haylage
that varied in terms of DM content as follows: in natura plant (not
dehydrated), 400, 500, and 600 g kg-1 DM
(dehydrated in the field until reaching the DM content of the treatment).
A completely randomized design in a 4 × 6 factorial scheme, with five
replications, was adopted for the gas assessment of the Tanzania grass haylage.
The factors were four levels of DM of the plant for haylage production and six
gas evaluation times: 0, 7, 15, 30, 45, and 60 d after wrapping the haylage
bales.
To assess the aerobic stability of Tanzania grass haylage, a completely
randomized design in a 4 × 6 factorial scheme, with five replications was
adopted. There were four levels of plant DM for haylage production and six
evaluation times: 0, 24, 48, 72, 96, and 120 h after opening the bales.
Haylage production
Tanzania grass was harvested right before it flowered; at this point,
the pasture had a height of 90 cm (30 days), as recommended by Euclides et al. (2014). The extracted material was
left in the field for pre-drying until it reached the determined DM content
(400, 500, and 600 g kg-1 DM), except for the material
of the in natura treatment, which was not dehydrated and immediately
baled. For treatments with pre-dried forage, the forage mass
was revolved to standardize dehydration. The forage was collected and
sealed when it reached the predetermined DM level. The DM content was
determined using the microwave method as previously described (55).
The bales were made in manual balers and then manually wrapped in
plastic film (SSFILM SSilage Xtreme®), with eight rounds per bale,
as recommended previously (38), to minimize gas exchange. The haylage bales weighed approximately 3 kg
and were stored for 90 d in a ventilated shed with no sunlight exposure.
To characterize the quality of the haylage, both in natura forage
and haylage were assessed using the following variables: chemical composition,
gas quantification, volatile fatty acids, microbiological profile, aerobic
stability, pH, and ammonia (N-NH3).
The analyses were conducted in the Animal Nutrition Laboratory and Microbiology
Laboratory of the Federal University of Piauí, located in the Bom Jesus, Piauí,
Brazil.
Determination of chemical composition and
gases
The samples used for the chemical composition analysis
of Tanzania grass before the production of the haylage (Table 1) (i.e., after
90 days of storage) were dried in a circulation and air renewal oven, at a
maximum temperature of 55 °C, until they reached a constant weight.
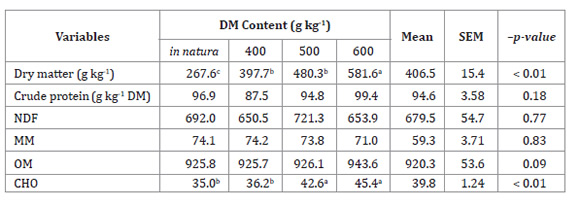
Table 1: Chemical composition of Tanzania grass across varying dry matter (DM)
contents, expressed as g kg-1 DM, prior to the production of
haylage.
Tabla 1: Composición química de la planta de pasto Tanzania de acuerdo con
la deshidratación, expresada como g kg-1 DM antes de la producción de
henolaje.
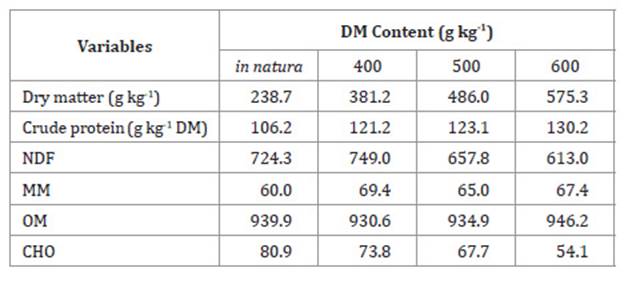
NDF: neutral detergent-insoluble fiber.
MM: Mineral matter. OM: Organic matter. CHO: Soluble
carbohydrate.
NDF:
Fibra insoluble en detergente neutro. MM: Materia mineral. OM: Materia
orgánica. CHO: Carbohidratos solubles.
They were then ground in a Thomas Willey stationary
mill through a 1-mm-mesh sieve. The contents of DM (n°. 934.01), crude protein
(CP n°. 981.10), mineral matter (MM n°. 934.05), and organic matter (OM no. 934.05) were determined
using the methods described previously (4), whereas neutral detergent fiber (NDF) was determined
using the methodology proposed by Van Soest et al. (1991).
The total CHO (TCHO) content was determined using the concentrated
sulfuric acid method described previously (17) with adaptations of Corsato et al. (2008).
The TCHO content was calculated as g 100 ml-1 based on the solution and
subsequently adjusted based on the DM of each sample used.
To evaluate the gases produced in the haylage, the levels of O2
and CO2 were measured. Assessments
were performed on the haylage on days 0, 7, 15, 30, 45, and 90, after it was
wrapped. Haylage was assessed on day 0, immediately after wrapping. The
readings were acquired through two valves (PVC pipes) that were inserted into
each bale and sealed for the duration of the established days’. For the gas
analysis, an O2 meter Instrutherm®
(model MO-900) was used, which also measured the internal
temperature of the bales, while CO2 was measured by a CO2
analyzer Testoryt® (White).
Quantification of volatile fatty acids and
microorganisms
To quantify the contents of volatile fatty acids (i.e., acetic,
propionic, isobutyric, butyric, isovaleric, and valeric acids) of Tanzania
grass haylage after 90 days of storage, only portions of each sample were used
for analysis through the method mentioned by Kung Jr and
Ranjit (2001), where the juice was extracted using a manual press. The
samples were centrifuged, and subsequently, the analysis of organic acids was
performed using high-resolution liquid chromatography using a high-performance
liquid chromatograph (HPLC) detector model SPD-10ª VP, coupled to the
ultraviolet detector (UV), using a wavelength of 210 nm. The boiling alcohol
content was determined using an ebulliometer, as recommended previously (28). Analyses were performed at the Laboratory of the Luís de Queiroz
College of Agriculture.
Microbiological evaluation was performed according to the
recommendations of González et al. (2003) by
collecting 25 g of fresh sample, adding 225 mL of distilled water, and
processing in a blender for approximately 1 min. One milliliter of the mixture
was pipetted at the appropriate dilution (10-1 10-9).
Plating was performed in duplicates for each culture medium. The populations
were determined by the selective technique of culturing in anaerobic media.
Rogosa Agar medium was used for counting lactobacilli (after incubation of 48
hours in an oven at 37°C); BDA Agar medium (Potato Dextrose Agar) acidified
with 1% tartaric acid, for the counting yeasts and molds (after 3-7 days of
incubation at room temperature); and Brilliant Green Bile Agar medium, for
counting the enterobacteria (after incubation of 24 hours at 35°C).
Plates with values between 30 and 300 colony-forming units (CFU) in the
Petri dish were considered acceptable for counting. Plaque averages of the
selected dilutions were considered.
Evaluation of aerobic stability, pH, and
ammonia nitrogen
When the haylage bales were opened, the forage mass was exposed to air
under a controlled room temperature (25°C); this approach was similar to that
applied in evaluations conducted in Johnson et al. (2002).
Room temperature was controlled using an INCOTERM® room thermometer. The internal
temperature of the haylage was measured using an INCOTERM® digital
skewer thermometer, and the surface temperature was measured using a BENETECH®
infrared digital thermometer with laser aim (-50 to 420°C).
Temperature was measured at 0, 24, 48, 72, 96, and 120 h. The aerobic stability
break was defined as an increase of 2°C in the temperature of the haylage in
relation to room temperature after opening the bales (35). During the evaluation period, samples from each treatment were
collected (approximately 100 g) at different time points (0, 24, 48, 72, 96,
and 120 h) to assess pH and ammonia (N-NH3)
levels, as per a previously described methodology (33).
Statistical analysis
The data were subjected to an analysis of variance. Means were compared
using Tukey’s test and linear regression, and all analyses were performed at a
significance level of p < 0.05. The data were analyzed using the
SISVAR software (version 5.0; 19).
Tukey’s test was used to analyze the chemical
composition and volatile fatty acid data. The adopted statistical model was:
![]()
where:
Yij
= record of the DM content i
μ =
general constant
Ti
= effect of the DM content i
with i
= 1-4; εij = random error associated with each DM content Yij
Gas data were analyzed using Tukey’s test for plant DM
and evaluation times. The following statistical model was adopted:
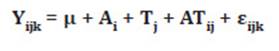
where:
Yijk
= record k, referring to the DM content i evaluated at time j
μ =
general constant
Ai
= effect of DM content i, i = 1-4
Tj
= gas evaluation time j, j = 0-120
ATij
= interaction between DM content i and gas evaluation time j
εijk = random error associated with each Yijk record
To evaluate the aerobic stability data, Tukey’s test was
used for plant DM, and linear regression analysis was used for the evaluation
times. The following statistical model was adopted:
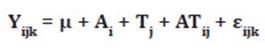
where:
Yijk
= record k, referring to the DM content i, evaluated at time j
μ =
general constant
Ai
= effect of the DM content i, i = 1-4
Tj
= stability evaluation time j, j = 0,...,
120
ATij
= interaction between the DM content i and stability evaluation
time j
εijk = random error associated with each Yijk record
Data referring to the quantification of microbial
groups ( logarithmic units, log 10) were analyzed
descriptively.
Results
Chemical composition and gases
The chemical composition of Tanzania grass haylage
according to plant DM and CHO contents was affected (p < 0.01) after
90 days of storage (Table 2).
Table 2: Chemical composition of Tanzania grass haylage according to the dry
matter content.
Tabla 2: Composición química del henolaje de pasto Tanzania según el
contenido de materia seca (MS) de la planta.
Means followed by different letters in a
row indicate statistical differences according to Tukey’s test at p <0.05;
NDF: Neutral Detergent Insoluble Fiber. MM: Mineral matter. OM: Organic matter.
CHO: Soluble carbohydrate. SEM: standard error of the mean.
Medias
seguidas de letras diferentes en la fila son estadísticamente diferentes según
la prueba de Tukey con p < 0,05. NDF: Fibra insoluble en detergente
neutro. MM: Materia mineral. OM: Materia orgánica. CHO: Carbohidratos
solubles. SEM: error estándar de la media.
The highest DM and CHO contents were observed in the haylage treated
with 600 gDM/kg (581.6 ± 15.4 gDM/kg and 45.4 ± 1.24
gCHO /kg DM, respectively). The other chemical composition variables were not
significantly different among the treatments, yielding mean values of 94.6 ±
3.58 gCP/kg DM, 679.5 ± 54.7 gNDF/kg DM, 59.3 ± 3.71 gMM/kg
DM, and 920 ± 53.6 gOM/kg DM. The desired DM contents after the 400,
500, and 600 gDM/kg treatments were very similar between plants (381.2, 486.0,
and 575.3 g/kg, respectively; Table 1, page 41) and
haylage (397.7, 480.3, and 581.6 g/kg; Table 2).
The quantification of O2 and CO2 gases in the Tanzania grass
haylage revealed a significant interaction effect (p < 0.01) between
the plant DM and number of days during which the gas composition of the grass
was evaluated during storage (Table 3).
Table 3: Gas and temperature quantification in Tanzania grass haylage based on
the plant dry matter (DM) during the various storage times.
Tabla 3: Cuantificación de los gases y la temperatura de henolaje de hierba
de Tanzania según la materia seca de la planta a través de los tiempos de
almacenamiento.
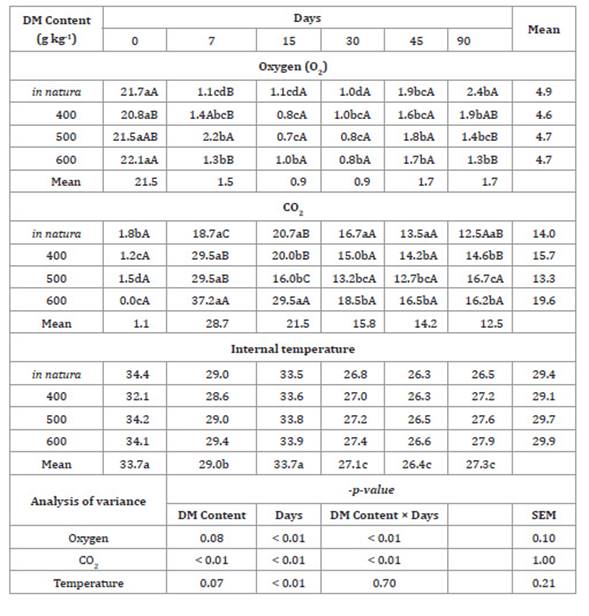
Means followed by different letters in a
column and row indicate statistical differences according to Tukey’s test at p
< 0.05. SEM: Mean Standard error.
Medias
seguidas de letras diferentes en la columna y en la fila son estadísticamente
diferentes según la prueba de Tukey con p <0,05. SEM: Error estándar
de la media.
Treatment with 400 g kg-1 DM resulted in the lowest
value of O2 on day 0, whereas the
treatment in natura resulted in the highest amount of O2 inside the bales when they
were opened after 90 days of storage. There was a reduction in the amount of O2
inside the Tanzania grass haylage bales after 7 days of storage
after all the treatments, and, after 90 days of storage, the O2 content was found to be less
than 2.5% inside all the bales.
The lowest CO2 values were observed on day 0.
CO2
increased between days 7th and 15th days of storage and after 90
days of storage. The treatments 400 and 500 g kg-1 DM resulted in the highest CO2
concentrations, which were 16.7 ± 1.0% and 16.2 ± 1.0%,
respectively. There was a significant effect (p < 0.05) of the
storage period on the internal temperature of the haylage, which reduced to 7.3
± 0.21°C after 45 days, and the highest temperatures were observed on days 0
and 15.
Volatile fatty acids and microorganisms
The highest acetic acid value of 36.4 ± 1.6 g kg-1
DM was obtained for the 600 g kg-1 DM treatment, followed by the
values of 38.2 ± 1.6 and 48.9 ± 1.6 g kg-1 DM for the in natura and
500 g kg-1
DM treatments, respectively. As for butyric acid, the highest
value (27.0 ± 0.5 g kg-1 DM) was observed for the in
natura treatment (Table 4).
Table 4: Concentration of volatile fatty acids in Tanzania
grass haylage based on the plant dry matter (DM) content, expressed as g/kg DM.
Tabla 4: Concentración de ácidos grasos volátiles en henolaje de pasto
Tanzania según la materia seca de la planta, expresada como g/kg MS.
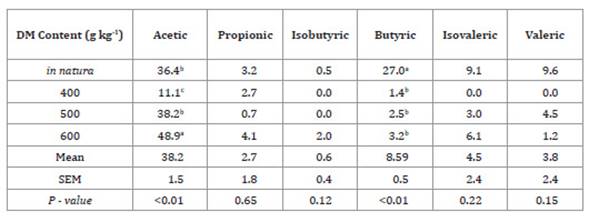
Means followed by different letters in a
column indicate statistical differences according to Tukey’s test at p <
0.05. SEM: Mean Standard error.
Medias
seguidas de letras diferentes en la columna son estadísticamente diferentes
según la prueba de Tukey con p <0,05. SEM: Error estándar de la media.
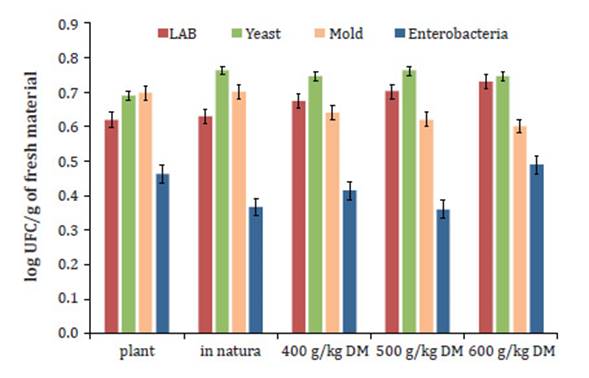
In the assessment of the microbial composition of
Tanzania grass and its resultant haylage (Figure 1), an increase in
the concentration of LAB was shown to be associated with an increase in the DM
content. LAB populations were found in the haylage at 6.9, 7.0 and 7.5 log
CFU/g for the 400, 500 and 600 g kg-1 DM treatments, respectively.
LAB: lactic acid bacteria. FM: fresh
material.
LAB:
bacteria del ácido láctico. FM: material fresco.
Figure 1: Microbial
composition of the plant and haylage of Tanzania grass stored with different
levels of dry matter.
Figura 1: Población de microorganismos de la planta y henolaje de pasto
Tanzania almacenado con diferentes niveles de materia seca.
No difference was found in the yeast population at the different plant
DM contents used for haylage production. Tanzania haylage had the smallest
yeast population of 6.9 log CFU/g. The 400, 500, and
600 g kg-1
DM treatments yielded yeast populations of 7.0, 7.6, and 7.2 log
CFU/g in the haylage, respectively.
The lowest amounts of mold, at 6.2 and 6.0 log
CFU/g, were observed in the haylage for the 500 and 600 g kg-1 DM treatments, respectively.
While the smallest amounts of enterobacteria, at 4.5 and 3.5 log CFU/g, were
found in haylages of the in natura and 500 g kg-1 DM treatments, respectively.
Aerobic stability, pH, and ammonia
nitrogen
Aerobic stability was affected by the interaction between the different
plant DM contents and hours of exposure of the Tanzania grass haylage to air
after opening the bales; this process was largely driven by surface
temperature, internal temperature, pH, and N-NH3 (Table
5).
Table 5: Aerobic stability of Tanzania grass haylage based on the plant dry
matter (DM).
Tabla
5: Estabilidad aeróbica
de henolaje de pasto Tanzania según la materia seca de la planta.
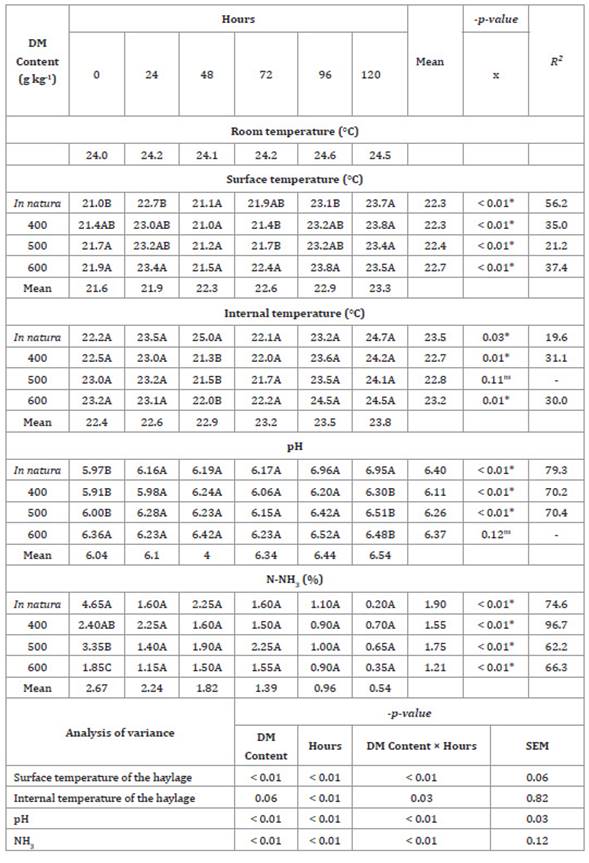
Means followed by different letters in a
column indicate statistical differences according to Tukey’s test at p <
0.05. * Significant at P < 0.05. ns not significant at p >
0.05. x: linear effect; SEM: Mean Standard error.
Medias
seguidas de letras diferentes en la columna son estadísticamente diferentes
según la prueba de Tukey con p < 0,05. * significativo
a p < 0,05. ns no significativo a
pb> 0,05. x: efecto lineal; SEM: Error estándar de
la media.
Haylage surface temperature had a linear relationship (p <
0.01) with the length of time the materials were exposed to air. Specifically,
between 0 and 120 hours of exposure to air, increases of 2.7 ± 0.06°C, 2.4 ±
0.06°C, 1.7 ± 0.06°C, and 1.6 ± 0.06°C were observed for the in natura,
400 g kg-1
DM, 500 g kg-1 DM, and 600 g kg-1
DM treatments, respectively. The 600 g kg-1 DM treatment yielded the
highest surface temperatures of the haylage, which were 21.9, 23.4, 21.5, 22.4,
and 23.8 ± 0.06°C for the exposure to air times of 0, 24, 48, 72, 96, and 120
hours, respectively. The in natura, 400 g kg-1 DM, and 600 treatments had an
increasing linear effect (p < 0.01) on internal temperature over the hours
of haylage exposure to air.
The in natura treatment yielded the highest
temperature of 25 ± 0.82°C in the haylage after 48 hours of exposure to air,
and the highest room temperature recorded was 24.6°C. During exposure to air,
there was no increase of 2% in the surface and internal temperatures of the
haylage compared to the room temperature (Table 5).
The in natura, 400 g kg-1 DM, and 500 g kg-1
DM treatments had an increasing linear effect (p <
0.01) on the pH of haylage during air exposure. In all treatments, the highest
pH values were recorded after 120 hours of exposure of the haylage to air, and
these values were 6.95 ± 0.03, 6.30 ± 0.03, 6.51 ± 0.03, and 6.48 ± 0.03 for
the in natura, 400 g kg-1 DM, 500 g kg-1 DM, and 600 g kg-1
DM treatments, respectively. The 600 g kg-1 DM treatment showed the
highest pH value of 6.36 ± 0.03 at hour 0, while the in natura treatment
showed the highest pH value of 6.95 ± 0.03 after 120 hours of exposure to air.
There was a decreasing linear effect (p < 0.01) of the hours of
exposure to air on the N-NH3 of Tanzania grass haylage in
all treatments at the time of baling. The in natura treatment showed the
highest N-NH3 value at hour 0 of exposure to
air (4.65 ± 0.12%).
Discussion
Chemical composition and gases
DM increased as the dehydration of the Tanzania grass continued in the
field; thus, the higher DM content of the haylage obtained in the 600 g kg-1
DM treatment, as compared to that obtained after the other
treatments, was due to the grass being dehydrated to a greater extent during
this treatment before it was baled (Table 2). The higher DM content in the stored material optimizes
the fermentation of the forage, shaping its preservation as haylage. Nath et al. (2018) obtained DM values for Tifton 85
grass haylage with different additives and storage times, with an average of
531.10 g/kg. Haylage is a technique that can be used for the storage of grasses
because dehydration reduces the probability of secondary fermentation, which
causes DM loss (31, 44, 58).
The DM values obtained for each treatment through dehydration, both
before and after the production of haylage, were nearly adequate according to
each treatment (400, 500, and 600 g kg-1 DM), demonstrating that the
method used to determine the DM content through a microwave is a viable
alternative to quickly obtain the DM value of forage plants on the farm (55) and can be used to determine the plant DM for haylage production. The
plant DM at harvest directly influences haylage fermentation (26, 40).
The haylage in the 600 g kg-1 DM treatment group had the
highest crude protein (CP) content; however, all haylage groups had a CP content greater than 70 g kg-1 DM, which is suggested by Van Soest (1994) as the ideal amount for the growth of
rumen microorganisms. The high CP content in the haylage was due to the high CP
content of the Tanzania grass before storage (Table
1). Castro et al. (2010)
evaluated the chemical composition of Tanzania grass at day 42 of storage and
obtained a CP content of 97.7 g/kg DM.
The results also indicated that the storage of haylage preserved the CP
content at levels suitable for animal feeding. The high CP content indicates
that when haylage is stored with adequate amounts of DM, it produces conditions
suitable for the growth of LAB (53) and inhibits the growth of undesirable microorganisms (44) that deteriorate CP.
The content of neutral detergent fiber (NDF) was higher than the maximum
limit of 550 g/kg DM recommended for good digestibility of the mass, which
occurs in silages with NDF levels as described previously (58). High NDF content may be related to the loss of cellular content during
the fermentation period (31), which negatively influences feed intake due to rumen filling (8). The NDF content obtained in the haylage of the 500 g kg-1 DM treatment group was lower
than that found by (6) in haylage of Tifton
85 grass, which was 723.6 g/kg DM, indicating the high quality of Tanzania
grass when harvested before flowering. Since NDF constitutes the cell wall of
plants (58), having haylages with NDF content similar to that of the original plant
suggests the adequate preservation of nutrients (Table
2).
Lower contents of mineral matter (MM) were observed in the Tanzania
grass haylages in all treatments (Table 1) than in the material before storage (Table
2). Low MM content is an indicator of better
forage conservation because when inadequate fermentation occurs, the loss of
organic material increases the amount of MM in the DM. The values obtained for
MM in this study were lower than those found by AOAC (1990)
in the haylage of Tifton 85 grass containing a bacterial inoculant.
The higher DM content treatments (500 and 600 g kg-1
DM) also yielded higher CHO content, which is an important
substrate for the fermentation and conservation of forage in the form of
haylage. The increase in dehydration of Tanzania grass increased its CHO
content, indicating that in treatments with higher moisture, there was a
greater use of CHO by microorganisms responsible for driving fermentation (50). In a previous study (14), it was observed that the haylages of two cultivars
of perennial ryegrass (i.e., AberDart and Fennema) fermented better when
the DM had a higher concentration of CHO.
Most tropical forage grasses do not have adequate levels of DM, CHO, or
buffering capacity to allow for fermentation to occur efficiently, resulting in
losses due to secondary fermentation, effluent production, and aerobic
deterioration, which are obstacles in the conservation of tropical grasses (10). Thus, the high levels of CHO in the haylage after 90 days of storage,
as obtained in the 500 and 600 g kg-1 DM treatments, showed that
fermentation was well controlled, resulting in good-quality forage for animal
feeding.
A high amount of oxygen was observed at the time of storage (0 days),
which was subsequently reduced after the 7th day of storage. The presence
of oxygen during storage favors the growth of microorganisms that release
energy in the form of heat, and fermentation by this
microbial mass results in the degradation of the roughage. Therefore,
oxygen must be eliminated before fermentation; in its absence, there is a
decrease in fungal and yeast growth, as anaerobic conditions are not optimal
for the growth of these organisms (20).
The increase in CO2 after the first days of
storage was due to it being released by aerobic microorganisms inside the bales
during fermentation. According to Paula et al. (2016),
the respiration of aerobic microorganisms occurs in the aerobic phase. These
microorganisms use some of the desirable substrates for energy production,
causing DM consumption and CO2 production, which can be
considered as one of the main factors that influence the quality of haylage.
The low levels of oxygen observed in the 500 and 600 g kg-1 DM treatments with 90 d of
storage were indicative of higher levels of anaerobic fermentation inside the
haylage bales, especially if it was associated with high amounts of CO2, as was observed in the
haylage with 600 g kg-1 DM. Low amounts of oxygen and
high amounts of CO2 are desirable parameters that
guarantee adequate anaerobic fermentation and yield products of good
nutritional quality. The activity of certain microorganisms can be controlled
using a controlled atmosphere or packaging in a modified atmosphere (52). According to Müller (2005),
the greater the number of wrapping layers in the haylage bales, the greater the
CO2
concentration. According to Mantilla et
al. (2010), the increase in food conservation time was due to the
inhibitory effect of carbon dioxide (CO2)
on different microbial types and the reduction or removal of oxygen (O2) from inside the bale.
It was observed that during storage, the low CO2 concentration increased from
the 7th
day, and this occurred because the aerobic microorganisms and
optional aerobes began to consume the available CHO, increasing the production
of gases through respiration and fermentation (carbon dioxide and ethanol).
After the 30th day, it was observed that the
microbial activity stabilized, decreasing respiration, and consequently, the
production of gas, as previously noted (60).
The internal temperature of the haylages increased during the first few
days of storage (0, 7, and 15 days), whereas the amount of O2 decreased and that of CO2
increased in this time period. According to Mcdonald
et al. (1991), in the first few days of storage until the end of the
aerobic phase, it is common to observe heating of the material, which can last
from 48 to 144 h.
Volatile fatty acids and microorganisms
A greater amount of acetic acid was observed in the 600 g kg-1
DM treatment as compared to the other treatments, indicating that
the lower moisture content caused an increase in the activity of acetic
acid-producing microorganisms in the haylage of Tanzania grass. The presence of
acetic acid is indicative of the action of heterofermentative LAB and
enterobacteria. High levels of this acid promote greater aerobic stability of
haylage after prolonged storage because it can inhibit yeast growth (1). Low concentrations of strong acids in haylage do not imply poor
fermentation (38), which may be due to the high DM content of the material.
The higher concentration of butyric acid in the in
natura treatment as compared to the other treatments is due to the higher
moisture content of the plant, which favors the growth of bacteria of the genus
Clostridium (49). No difference was observed for the other acids, and
this indicates that the DM content greatly influences the production of haylage
within the context of Tanzania, and its presence is shaped by the action of
heterofermentative LAB, enterobacteria, and clostridia.
LAB concentrations increased according to increasing DM content of the
plants used for haylage production (Figure 1). The larger population of homofermentative LAB tends to
reduce pH more quickly, reducing the action of undesirable microorganisms and
preserving a greater amount of carbohydrates, with the increase in the DM
content (36); this phenomenon
was not observed in this experiment that obtained high pH values. The haylages
produced had LAB populations greater than the minimum limit of 5 log CFU g-1
recommended by Pahlow (1986) and Muck et al. (1991) and t required for a good fermentation process.
The increased presence of yeast populations is concerning because of
their potential to rapidly multiply, but no difference was observed in the
yeast population with respect to the different DM contents of the plant used
for haylage-making. Notably, several types of yeasts may predominate during the
haylage-making process, and the yeast species present are not necessarily
aerobic. This may explain the absence of a difference in the counts of these
microorganisms between treatments. Low yeast populations are desirable for
preserving the material during fermentation and after bales opening (45). The yeast count of the Tanzania grass haylages was higher than that
found by Müller and Johansen (2020) in reallocated
haylage (5.31 log CFU g-1)
and that observed by Müller et al. (2011) in the haylages of horse farms (4.57 log CFU
g-1).
A lower count of molds was observed in the haylage of the 500 and 600 g
kg-1
DM treatments, which is related to the plant DM during storage.
Generally, the population of microorganisms is strongly affected by the
moisture content and temperature recorded during storage (37). The presence of fungi causes a reduction in nutritional value and
palatability due to the associated protein degradation (16). Some mycotoxin-producing molds were observed in the haylages, but they
only occurred at low concentrations; this was in line with the results of a
study by Müller et al. (2011). The presence of such
molds can be reduced through the use of additives (38).
The 600 g kg-1 DM treatment yielded the
highest amount of enterobacteria in the haylage of Tanzania grass. The large
number and prevalence of these microorganisms are undesirable, as they cause
protein degradation by performing secondary fermentation and producing compounds
such as acetic and butyric acids, impairing conservation (51). However, these bacteria produce acetic acid, which, in the absence of
lactic acid, can help conserve the material and increase its aerobic stability.
Enterobacteria compete for water-soluble carbohydrates with LAB, and the
component with the highest concentration at the end of this process is acetic
acid, which has a positive effect on aerobic stability. Haylage with poor
aerobic stability has high levels of residual sugar and lactic acid (14).
Aerobic stability, pH, and ammonia
nitrogen
The temperature increase observed in Tanzania grass haylages with
different DM levels throughout exposure to air was not sufficient to negatively
impact aerobic stability across the evaluation period of 120 h. The aerobic
stability of the haylage, regardless of treatment, was likely maintained
because of the high acetic acid concentration in the 600 g kg-1 DM treatment and the low
amount of CHO in the other treatments. These characteristics inhibit the growth
of deteriorating microorganisms (16). Müller (2009) did not observe a change in the
aerobic stability of the haylage of plants harvested at different times.
Haylage treated with 600 g/kg DM had the highest surface temperature,
probably because of the high amounts of CHO. Better haylage fermentation
patterns with higher DM content provide a greater number of available
substrates for the consumption of microorganisms in the aerobic phase (61).
The variation in the internal temperature of the
Tanzania grass haylage was not enough to overcome the room temperature at 2°C
in all treatments and times. Neres et al. (2013)
assessed the aerobic stability of Tifton 85 grass silage and observed that the
room temperature was lower than the ensiled mass temperature during the seven
days of aeration, which contributed to good preservation of the roughage and
inhibition of the growth of undesirable microorganisms. The aerobic stability
of haylage can also be influenced by the production of acetic acid, which
varies according to pH and temperature increases in the respective pre-dried
forage masses (56).
Haylage stored with 600 g kg-1 DM showed no difference in pH
during exposure to air. This occurred because of the lower moisture content,
which provided greater resistance to the pH drop because of the lower activity
of microorganisms. Belém et al. (2016) reported
that the limited activity of bacteria owing to moisture has a direct effect on
aerobic fermentation.
The pH values observed in the Tanzania grass haylages were higher than
those observed by Coblentz et al. (2016) in the
alfalfa haylage (5.1), which was almost similar to those observed by Nath et al. (2018) in the haylage of Tifton 85
grass (5.72) and lower than those obtained by Weirich et
al. (2018) in the Tifton 85 haylage
(7.38). The high pH values observed in the haylage
of tropical grasses may be due to the low concentration of organic acids in the
masses of these species (3). Müller et al. (2007) compared silage to
haylage and observed that haylage had a higher pH owing to lower concentrations
of fermentative products.
The N-NH3 content of Tanzania grass
haylage decreased as its exposure to air progressed. As the times of aerobic
exposure advanced, the Tanzania grass haylages showed a reduction in the
average levels of ammoniacal nitrogen, probably due to evaporative processes and
a decrease in the enterobacteria population (48).
The in natura treatment produced the highest amount of N-NH3, indicating the high
intensity of proteolysis during the fermentation process. However, it is
important to note that all haylages were classified as those of good quality. Monteiro et al. (2011) classified haylages as good
quality haylages when the fermented materials had levels of N-NH3 below 12%. This was also
indicative of low proteolysis intensity during fermentation (57).
Conclusions
Higher plant DM yields Tanzania grass haylage of high quality. Tanzania
grass with 500 and 600 g kg-1 DM for haylage production had
a high content of CHO, a better concentration of gases, and a greater amount of
volatile fatty acids and beneficial microorganisms that facilitate
preservation. Additionally, these haylages showed sustained aerobic stabilities.
It is
necessary to conduct further studies on plant DM using other tropical grasses
to produce high-quality haylage.
Acknowledgments
Tanks CNPq (Grant No.424941/2016-3) for
financial support.
1.
Agarussi, M. C. N.; Pereira, O. G.; Silva, V. P.; Leandro, E. S.; Ribeiro, K.
G.; Santos, S. A. 2019. Fermentative
profile and lactic acid bacterial dynamics in non-wilted and wilted alfalfa
silage in tropical conditions. Mol Biol Rep. 46:
451˗460.
2. Alvares, C. A.; Stape, J. L.; Sentelhas,
P. C.; Moraes, G.; Leonardo, J.; Sparovek, G. 2013. Köppen’s
climate classification map for Brazil. Rev Bras Meteorol. 22: 711˗728.
3.
Amorim, D. S.; Sousa, C. M. S.; Nascimento, R. R.; Silva, A. L. 2017.
Pré-secado: uma alternativa para aumentar a segurança alimentar dos rebanhos no
período de escassez de forragem. Rev.
electrón. vet. 18: 1-13.
4. AOAC. 1990. Association of official,
chemists, official methods of analysis. 15th Edition,
Washington DC. U.S.A.
5. Belém, C. D. S.; Souza, A. M. D.; Lima,
P. R. D.; Carvalho, F. A. L. D.; Queiroz, M. A. Á.; Costa, M. M. D. 2016. Digestibility, fermentation and microbiological characteristics of
Calotropis procera silage with different quantities of grape pomace. Cienc. e Agrotec. 40: 698˗705.
6. Bernard, J. K.; Castro, J. J.; Mullis,
N. A.; Adesogan, A. T.; West, J. W.; Morantes, G. 2010. Effect
of feeding alfalfa hay or Tifton 85 bermudagrass haylage with or without a
cellulase enzyme on performance of Holstein cows. J Dairy Sci. 93:
5280-5285.
7. Bernardes, T. F.; Rêgo, A. C. 2014.
Study on the practices of silage production and utilization on Brazilian dairy
farms. J
Dairy Sci. 97: 1852˗1861.
8.
Bonfá, C. S.; Castro, G. H. F.; Villela, S. D. J.; Santos, R. A.; Evangelista,
A. R.; Jayme, C. G.; Barbosa, J. A. S. 2015. Elephant
grass silage added to passion fruit’s peel. Arq Bras Med Vet
Zootec. 67:
801˗808.
9.
Braz, T. G. D. S.; Martuscello, J. A.; Santos, M. E. R.; Pereira, V. V. 2017. Partial correlation analysis in the study
of morphogenesis and herbage accumulation in Panicum maximum cv. ‘Tanzânia’. Ciênc Rural. 47: 238˗256.
10. Carvalho, C. B. D. M.; Silva, S. F.; Carneiro, M. S. D. S.;
Edvan, R. L.; Pereira, E. S. 2017. Composição química de silagem e feno de
Gliricídia sepium em diferentes alturas de resíduo. Rev bras saúde prod Anim.
18: 239˗248.
11.
Castro, G. H. F.; Rodriguez, N. M.; Gonçalves, L. C.; Maurício, R. M. 2010.
Características produtivas e nutricionais do capim-Tanzânia em diferentes
idades de corte. Arq Bras Med Vet
Zootec. 62: 654˗666.
12. Coblentz, W. K.; Coffey, K. P.; Chow,
E. A. 2016. Storage characteristics, nutritive value, and fermentation
characteristics of alfalfa packaged in large-round bales and wrapped in stretch
film after extended time delays. J Dairy Sci. 99: 3497˗3511.
13. Collins, B. M.; Stevens, J. T.;
Miller, J. D.; Stephens, S. L.; Brown, P. M.; North, M. P. 2017. Alternative
characterization of forest fire regimes: incorporating spatial patterns. Landsc
Ecol. 32: 1543˗1552.
14. Conaghan, P.; Kiely, P. O.; Mara, F.
P. O. 2010. Conservation characteristics of wilted perennial ryegrass silage
made using biological or chemical additives. J Dairy Sci. 93: 628˗643.
15. Corsato, C. E.; Scarpare Filho, J. A.;
Sales, E. C. J. 2008. Teores de carboidratos em órgãos lenhosos do caquizeiro em clima
tropical. Rev Bras Frutic. 30: 414-418.
16.
Dordević, S.; Mandić, V.; Stanojević, D.; Jovanović-Ljesković, N. 2017. Effects of Lactobacillus plantarum inoculants
on maize silage quality. Biotechnol Anim Husban. 33: 115˗122.
17. Dubois, M.; Gilles, K. A.; Hamilton,
J. K.; Rebers, P. A.; Smith, F. 1956. Colorimetric method for
determination of sugars and related substances. Anal.
Biochem. 28:
350˗356.
18.
Euclides, V. P. B.; Montagner, D. B.; Barbosa, R. A.; Nantes, N. N. 2014.
Manejo do pastejo de cultivares de Brachiaria brizantha (Hochst) Stapf e de
Panicum maximum Jacq. Rev Ceres. 61: 808˗818.
19. Ferreira, D. F. 2011. Sisvar: computer
statistical analysis system. Cienc. e Agrotec. 35: 1039˗1042.
20.
França, A. M. S.; Ferreira, I. C.; Hermisdorff, Í. C.; Mendonça, E. P.;
Fernandes, E. A.; Rossi, D. A. 2015. Dinâmica química, microbiológica e física
da silagem de farelo úmido de glúten de milho. Cienc. Rural. 45:
684˗689.
21. González, G.; Rodríguez, A. A. 2003. Effect of storage method on fermentation characteristics, aerobic
stability, and forage intake of tropical grasses ensiled in round bales.
J Dairy Sci. 86: 926˗933.
22. Harris, P. A.; Ellis, A. D.; Fradinho,
M. J.; Jansson, A.; Julliand, V.; Luthersson, N.; Vervuert, I. 2017. Feeding
conserved forage to horses: recent advances and recommendations. Animal. 11: 958˗967.
23. Horst, E. H.; Neumann, M.; Mareze, J.;
Leão, G. F. M.; Dochwat, A. 2016. Silagem pré-secada de cereais de inverno em estádio de
pré-florescimento: Revisão. Pubvet. 11: 415˗423.
24. Johnson, L. M.; Harrison, J. H.;
Davidson, D.; Mahanna, W. C.; Shinners, K.; Linder, D. 2002. Corn silage
management: effects of maturity, inoculation, and mechanical processing on pack
density and aerobic stability. J Dairy Sci. 85: 434˗444.
25. Kung Jr., J. R. L.; Ranjit, N. K.
2001. The effect of Lactobacillus buchneri and other
additives on the fermentation and aerobic stability of barley silage. J Dairy Sci . 84: 1149˗1155.
26. Leão, G. F. M.; Jobim, C. C.; Neumann,
M.; Horst, E. H.; Santos, S. K. D.; Venancio, B. J.; Santos, L. C. 2017. Nutritional composition and aerobic stability of winter cereal
silage at different storage times. Acta Sci Anim Sci. 39: 131˗136.
27.
Lehmen, R. I.; Fontaneli, R. S.; Fontaneli, R. S.; Santos, H. P. D. 2014.
Rendimento, valor nutritivo e características fermentativas de silagens de
cereais de inverno. Cienc. Rural . 44: 1180˗1185.
28.
Maia, A. B. R. A.; Campelo, E. A. P. 2005. Tecnologia da cachaça de alambique.
Belo Horizonte: SEBRAE-MG/SINDBEBIDAS. p 129.
29.
Mantilla, S. P. S.; Mano, S. B.; Vital, H. C.; Franco, R. M. 2010. Atmosfera
modificada na conservação de alimentos. Rev Acad: Ciênc Agrár. 8: 437-448.
30.
Martha Jr., G. B.; Vilela, L.; Sousa, D. M. G. 2007. Adubação nitrogenada.
Cerrado: uso eficiente de corretivos e fertilizantes em cerrados. Planaltina:
Embrapa Cerrados. 117˗144.
31.
Mcdonald, P.; Henderson, A. R.; Heron, S. J. E. 1991. The biochemistry of silage.
2. New York: Chalcombe Publications. p 339.
32.
Medeiros, R. M.; Santos, D. C.; Sousa, F. A. S. 2013. Análise Climatológica,
Classificação Climática e Variabilidade do Balanço Hídrico Climatológico na
Bacia do Rio Uruçui Preto, PI. Rev Bras Geogr Fís. 6: 652˗664.
33.
Mizubuti, I. Y.; Pinto, A. P.; Pereira, E. S.; Ramos, B. M. O. 2009. Métodos
laboratoriais de avaliação de alimentos para animais. 507, Londrina: EDUEL. p
228.
34.
Monteiro, I. J. G.; Abreu, J. G.; Cabral, L. S.; Ribeiro, M. D.; Reis, R. H. P.
2011. Elephant grass silage additives with
alternative products. Acta Sci Anim Sci. 33: 347˗352.
35. Moran, J. P.; Weinberg, Z. G.;
Ashbell, G.; Hen, Y.; Owen, T. R. 1996. A comparison of two
methods for the evaluation of the aerobic stability of whole crop wheat silage.
In: International Silage Conference, 11. Aberystwyth. Proceedings. Aberystwyth: University of Wales Aberystwyth. 162˗163.
36.
Muck, K. A.; Albrecht, R. E. 1991. Proteólise em leguminosas forrageiras
ensiladas que variam na concentração de taninos. Crop
Sci. 31: 464˗469.
37. Mufatto, L. M.; Neres, M. A.; Nath, C.
D.; Stangarlin, J. R.; Scheidt, K. C.; Casarotto, L.; Sunahara, S. M. M. 2016. Caracterização e
quantificação da população de fungos em área de produção de feno de capim
Tifton 85, adubado com biofertilizante suíno. Cienc. Rural . 46:
486˗491.
38. Müller, C. E. 2005. Fermentation
patterns of small-bale silage and haylage produced as a feed for horses. Grass
Forage Sci. 60: 109˗118.
39. Müller, C. E. 2009. Influence of
harvest date of primary growth on microbial flora of grass herbages and
haylage, and on fermentation and aerobic stability of haylage conserved in
laboratory silos. Grass Forage Sci . 64: 328˗338.
40. Müller, C. E. 2018. Silage
and haylage to horses. Grass Forage Sci . 73: 815˗827.39.
41. Müller, C. E.; Pauly, T. M.; Udén, P.
2007. Storage of small bale silage and haylage - influence of
storage period on fermentation variables and microbial composition. Grass Forage Sci . 62: 274˗283.
42. Müller, C. E.; Hultén, C.; Gröndahl,
G. 2011. Assessment of hygienic quality of haylage fed to healthy horses. Grass Forage Sci . 66: 453˗463.
43. Müller, C. E.; Johansen, A. 2020. Rebaling of silage and haylage and its effects on forage microbial
and chemical composition - A pilot study. Grass Forage Sci .
75: 216˗226.
44. Nath, C. D.; Neres, M. A.; Scheidt, K.
C.; Santos, B. L.; Sunahara, S. M. M.; Sarto, J. R. W.; Perin, A. P. 2018. Characterization of Tifton 85 bermudagrass haylage with different
layers of polyethylene film and storage time. Asian-Australas J Anim
Sci. 31: 1197.
45.
Neres, M. A.; Zambom, M. A.; Fernandes, T.; Castagnara, D. D.; Rodrigues, J. F.
H.; Taffarel, L. E.; Javorski, C. R.; Pozza, M. S. D. S. 2013. Perfil
microbiológico e estabilidade aeróbica da silagem de capim-tifton 85 com
diferentes aditivos. R
Bras Zootec. 42: 381˗387.
46. Pahlow, G. 1986. Microbiology
of inoculants, crops and silages. In: Proceedings of the Eurobac
Conference, 12-16 August 1986, Uppsala, Sweden. Grass and Forage Reports, 3,
1990 (Special Issue), p. 45-59. Uppsala, Sweden: Swedish University of
Agricultural Sciences.
47. Paula, F. L. M.; Menezes, L. F. G.;
Paris, W.; Ronsani, R.; Hoppen, S. M.; Ciesc, J. 2016. Silage
production and the chemical composition of corn and Grass-tanzania
intercropping. Semina: Ciênc Agrár. 37: 1607˗1616.
48.
Possenti, R. A.; Arantes, A. M.; Brás, P.; Andrade, J. B.; Júnior, E. F. 2016.
Avaliação nutritiva da silagem de cártamo, produção de biomassa, grãos e óleo.
B Ind Anim. 73: 236˗243.
49.
Santos, K. C.; Magalhães, A. L. R.; Silva, D. K. A.; Araújo, G. G. L. D.;
Fagundes, G. M.; Ybarra, N. G.; Abdalla, A. L. 2017. Nutritional
potential of forage species found in Brazilian Semiarid region. Livest Sci.
195: 118˗124.
50. Santos, K. C.; Magalhães, A. L. R.;
Conceição, M. G.; Carvalho, F. F. R.; Ferreira, G. D. G. 2018. Common bean residue as additive in sugarcane silage. Rev
Cienc Agron. 49: 159˗166.
51. Sariçiçek, B. Z.; Yildirim, B.;
Kocabaş, Z. B. Z.; Demir, E. Ö. 2016. The effects of storage time on nutrient
composition and silage quality parameters of corn silage made in plastic mini
silo in laboratory conditions. J Inst Sci Tech. 6: 177˗183.
52.
Silva, J. L.; Costa, F. B.; Nascimento, A. M.; Araújo, C. R.; Santos, K. P.;
Nobre, M. A. F.; Sales, G. N. B. 2020. Influence
of storage under controlled temperature the physical characteristics of jujube
fruits (Ziziphus joazeiro Mart.). Res Society Develop. 9: 1859˗1867.
53. Soundharrajan, I.; Kim, D. H.;
Srisesharam, S.; Kuppusamy, P.; Park, H. S.; Yoon, Y. H.; Kim, W. H.; Song, Y.
G.; Choi, K. C. 2017. Application of customised bacterial
inoculants for grass haylage production and its effectiveness on nutrient
composition and fermentation quality of haylage. Biotech.
7: 321.
54. Sousa, C. M.; Bittar, D. Y.; Diniz
Buso, W. H. 2022. Responses of Panicum and Brachiaria
to irrigation during winter in the Goiás’ Cerrado-Brazil. Revista de la Facultad de
Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina. 54(2):
117-125.
55.
Souza, G. B.; Nogueira, A. R. A.; Rassini, J. B. 2002. Determinação de matéria
seca e umidade em solos e plantas com forno de microondas doméstico. Circular
Técnica. São Carlos: Embrapa Pecuária Sudeste. p 9.
56.
Twarużek, M.; Dorszewski, P.; Grabowicz, M.; Szterc, P.; Grajewski, J.;
Kaszkowiak, J. 2016. Effect
of additives on the fermentation profile, microbial colonization and oxygen
stress of alfalfa silages. J Elem. 21:
1161˗1172.
57. Tyrolová, Y.; Bartoň, L.; Loučka, R.
2017. Effects of biological and chemical additives on fermentation progress in
maize silage. Czech J Anim Sci. 62: 306˗312.
58. Van Soest, P. J. 1994. Nutritional ecology of ruminant. 2° ed. Ithaca: Cornell
University Press. p. 476.
59. Van Soest, P. J.; Robertson, J. B.;
Lewis, B. A. 1991. Methods for dietary
fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to
animal nutrition. J Dairy Sci . 74: 3583˗3597.
60.
Valença, R. L.; Ferreira, A. C. D.; Santos, A. C. P.; Silva, B. C. D.; Santos,
G. R. A.; Oliveira, E. S. 2016. Composição química e perdas em silagem de
bagaço de laranja pré-seco. B Ind Anim. 73: 206˗211.
61. Weirich, D. T.; Neres, M. A.; Hunoff,
C. A.; Ströher, S. M.; Nath, C. D.; Sunahara, S. M. M.; Oldoni, T. 2018. Microbiological profile and aerobic stability of Tifton 85
bermudagrass silage with or without vacuum and microbial inoculants. Biosci J. 34: 151-162.
62.
Zamarchi, G.; Pavinato, P. S.; Menezes, L. F. G.; Martin, T. N. 2014. Silagem
de aveia branca em função da adubação nitrogenada e pré-murchamento. Semina: Ciênc Agrár. 35: 2185˗2195.
Disclosure statement
The authors
declare no conflicts of interest associated with this paper. The authors alone are
responsible for the content and writing of this manuscript.