Revista de la Facultad de Ciencias
Agrarias. Universidad Nacional de Cuyo. Tomo 55(1). ISSN (en línea) 1853-8665.
Año 2023.
Original article
PCR
identification of lactic acid bacteria populations in corn silage inoculated
with lyophilised or activated Lactobacillus buchneri
Identificación
por PCR de poblaciones de bacterias del ácido láctico ensilado a partir de maíz
inoculado con Lactobacillus buchneri liofilizado o activado
Ana Paula Maia dos Santos 1
Edson Mauro Santos 2
Juliana Silva de Oliveira 2
Gherman Garcia Leal de Araújo
3
Anderson de Moura Zanine 4
Ricardo Martins Araújo Pinho
5
Thiago Vinicius Costa do Nascimento
6
Alexandre Fernandes Perazzo
7
Daniele de Jesus Ferreira 4
Alberto Jefferson da Silva
Macedo 8
Francisco Naysson de Sousa
Santos 4*
1 Alagoas State University.
Departament of Animal Science. Rod. BR-316, Km 87.5. s/n.
57500-000. Santana do Ipanema. Alagoas. Brazil.
2 Federal University of
Paraíba. Department of Animal Science. Rod. BR-079, 58397- 000. Areia. Paraíba.
Brazil.
3 Brazilian Agricultural
Research Corporation. Embrapa Tropical Semiarid, Rod. BR- 428. Km 152. s/n, 56302970. Petrolina. Pernambuco. Brazil.
4 Federal University of
Maranhão. Department of Animal Science. Rodovia BR 222. km 4. s/n. 65500-000. Chapadinha. Maranhão. Brazil.
5 Federal Institute of
Education. Science and Technology of Ceara. Department of Animal Science. Rod.
BR-292. s/n. Gisélia Pinheiro. 63155-500. Crato.
Ceará. Brazil.
6 Federal University of
Sergipe. Department of Veterinary. Nossa Senhora da Glória. 49680-000. Sergipe.
Brazil.
7 Federal University of
Piaui. Department of Planning and Agricultural Policy. Campus Universitário
Ministro Petrônio Portella. Bairro Ininga. 64049-550. Teresina. Piauí. Brazil.
8
Federal University of Viçosa. Department of Animal Science. Av. P H Rolfs. s/n - Campus
Universitário. 36570-900. Viçosa. Minas Gerais. Brazil.
* nayssonzootecnista@gmail.com
Abstract
This study aimed to
evaluate the effect of inoculation with lyophilised and/or activated Lactobacillus
buchneri on lactic acid bacteria populations in corn silage. Experimental
treatments consisted of corn silage without additives or silage with the
inoculants of L. buchneri (1 x 105 cfu/g) applied according to
the manufacturer’s recommendations (1 g/tonne fodder) in the forms of the
lyophilised inoculant and pre-activated inoculant. Purified isolates from corn
silage with and without the inoculant were identified, and 93% of the isolates
corresponded to the lactic acid bacteria of the species Lactobacillus
plantarum. Among the isolates, no bacteria of the species L. buchneri
were detected. The application of lyophilised or activated L. buchneri
improved the microbiological profile and reduced ethanol production in corn
silage, even without being identified among the isolates captured 70 days after
ensilage.
Keywords: Fermentation; Inoculant; Lactic acid bacteria; Polymerase chain reaction.
Resumen
Este estudio tuvo como objetivo evaluar el efecto de la
inoculación con L. buchneri liofilizado y/o activado sobre las
poblaciones de bacterias de ácido láctico en ensilajes de maíz. Los
tratamientos experimentales consistieron en ensilaje de maíz sin aditivo y
ensilaje con los inoculantes L. buchneri 1 × 105 ufc/g,
aplicados según las recomendaciones del fabricante (1 g/tonelada de forraje) en
las formas de inoculante liofilizado e inoculante preactivado. Se identificaron
aislados purificados de ensilajes de maíz con y sin inoculación, y el 93% de
los aislados correspondieron a bacterias de ácido láctico de la especie L.
plantarum. Entre los aislados no se detectaron bacterias de la especie L.
buchneri. La aplicación de L. buchneri liofilizado o activado en la
masa ensilada promueve un aumento considerable de la población de bacterias de ácido
lácticas en los ensilajes de maíz durante todo el período de fermentación.
Palabras
clave: Fermentación; Inoculante; Lactobacillus plantarum; Reacción
en cadena de la polimerasa.
Originales: Recepción: 08/11/2021
Aceptación:
23/02/2023
Introduction
Chemical characteristics associated with the roughage used, such
as dry matter content, concentration of water-soluble carbohydrates, buffer
substances and populations of microorganisms present in the forage, and
associated with the silage process bottlenecks, i.e. the silo filling
time, compaction and sealing, are among the main factors that modulate the
silage production process and modify the process and the characteristics of the
silage (23, 34).
The silage process is directly associated with the nature of the
epiphytic microbial community, where bacterial diversity is the determinant
factor of the fermentation pattern of silage. The microbial communities
verified in forage crops before the silage process differ considerably in
number and taxonomy from those quantified in silage (3,
7, 21).
Corn (Zea mays L.) has adequate characteristics for good
fermentation in a silo when harvested with the right dry matter content (16). However, due to the high
concentration of water‐soluble
carbohydrates (WSC), DM losses might occur during fermentation and when the
silo is opened to withdraw the silage (16, 26).
Thus, several corn silage inoculants have been researched in
recent years, increasing knowledge about the dynamics of the action of bacteria
in silage mass (31, 32). The main
additives were composed of microbial species of lactic acid bacteria (LAB),
which are gram-positive, non-spore forming, strictly fermentative, anaerobic or
aerobic tolerant, and acidophilic, and classified according to the type of
hexose fermentation (16, 18, 28), which
can be deferred. According to the species of Lactobacillus, Pediococcus,
Leuconostoc, Enterococcus, Lactococcus and Streptococcus,
classified as homolactic fermenters (Lactobacillus plantarum) or
heterolactic fermenters (Lactobacillus buchneri), both producers of
lactic acid and heterolactics can also produce acetic acid (17).
Lactobacillus buchneri has slow growth, with its effects
observed from 45 to 60 days (20, 29),
which increases the aerobic stability of silage (8,
24). The lactic acid and acetic acid produced are inhibitors of
yeasts and moulds (20, 31).
However, the formation of a large amount of acetic acid in silage
does not present an advantage. This is related to the loss of energy and the
reduction of the intensity of the pH drop in the silage (14, 17). The further development of L. buchneri
in silage mass may promote the increased aerobic stability of corn silage (30).
Through commercial products, the
inoculation of silage occurs industrially from the dilution in water of the
inoculant with strains of lyophilised lactic acid bacteria. The previous
activation of the inoculant in reconstituted skim milk (RSM) prior to the ensiling
process may increase the availability of active bacteria in the silage mass.
The activation in RSM favours LAB in competition with other undesirable
microorganisms and redirects the fermentation pattern and aerobic stability of
the silage (27, 31), promoting increased
inoculation efficiency and higher yield and quality of the final product.
Knowledge of epiphytic
microbial diversity can contribute to the understanding of the fermentation
pattern of silage, as well as the phenomena arising from the exposure of this
material to aerobiosis. Molecular biology techniques have been used to
determine changes in the microbial community, favouring the intensity of
responses about microbial diversity in silage and the effects of the
inoculation of different strains of isolated LAB (13,
26).
Accordingly, the aim was
to evaluate the effect of inoculation with L. buchneri lyophilised
or activated in RSM on LAB populations in corn silage.
Material and methods
Location and meteorological data
The experiment was
conducted in the Forage Farming Sector of the Department of Animal Science of
the Center of Agricultural Sciences, Federal University of Paraiba. The climate
in the region is As’ (hot and humid), according to the Köppen classification. According
to data from the Meteorological Station of the Agricultural Sciences Center of
the Federal University of Paraiba, the average annual precipitation is 1400 mm;
the average annual temperature is 24.5°C; and the average relative humidity is
80%.
Corn silage and treatments
The corn harvest was
carried out at 97 days of age, when the grains were in the milky / pasty stage.
The plants were harvested manually with an MS content of 26.2% and chopped in a
stationary forage machine regulated to cut the forage into particles of
approximately 2 cm and homogenised prior to inoculation and silage of the corn
plant (Table
1).
Table 1: Chemical composition and microbial populations of forage prior to
inoculation and silage.
Tabla 1: Composición química y poblaciones microbianas del forraje antes de
la inoculación y ensilaje.
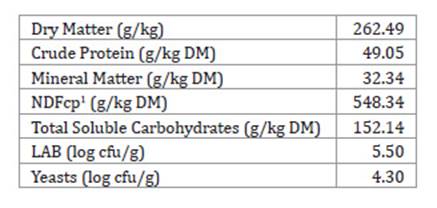
1 Neutral Detergent Fibre. corrected for
ashes and proteins.
1 Fibra
Detergente Neutra corregida para cenizas y proteínas.
Ensiling was carried out
in tubular silos of PVC (15 cm diameter and 40 cm height), according to the
treatments: silage without the inoculant (in natura), silage with the
lyophilised inoculant (SLI) and silage with the activated inoculant (SAI).
The inoculant with strains
of L. buchneri (1 × 105cfu
/ g; Lactobacillus buchneri CNCM I-4323, Lallemand and Animal Nutrition)
was applied according to the manufacturer’s recommendations (1 g / tonne
fodder). The lyophilised inoculant was diluted in 100 ml distilled water and
applied uniformly (2 ml / kg fodder) from a spray and constant mixture.
The inoculant was preactivated in
10% RSM 24 hours prior to ensiling. Skimmed milk powder (10 g) was solubilised
in 100 ml of distilled water, and two grams of sucrose was added as an energy
source for microbial growth, according to the methodology of Santos et al. (2008). After growth, counts of
lactic acid bacteria in the RSM were performed after 24 hours, as well as in
the inoculant dissolved only in water, through the MRS culture medium (5) for Lactobacillus ssp.
Populations of 2.1 × 108 and 4.5 × 106cfu
/ ml were grown in RSM growth medium and diluted in water, respectively. In 10
ml of RSM with reactivated L. buchneri, which was rediluted in 90
ml of distilled water, 2 ml / kg of natural material of this mixture was
applied by spray.
The additives were mixed homogeneously to fill the experimental
silos. Immediately, forage compaction was carried out in the silos, aiming to
reach a specific mass of 600 kg / m3 of natural matter. The silos
were stored for 70 days at a mean temperature of 24°C before opening. Openings
were performed at 1, 3, 7, 14 and 70 days after the silos were closed (Table 2).
Table 2: Mean pH and concentrations of organic acids, ethanol and yeast of
corn silage without inoculants and with lyophilised or activated microbial
inoculants.
Tabla 2: Valores medios de pH, concentraciones de ácidos orgánicos, etanol
y levaduras de ensilajes de maíz sin inoculantes y con inoculante microbiano
liofilizado o activado.
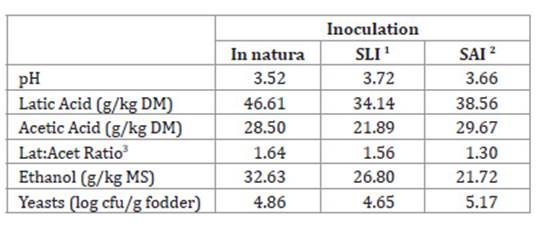
1 SLI = Corn silage with lyophilised microbial inoculant; 2
SAI = Corn silage with activated microbial inoculant; 3
Lat: Acet Ratio = Latic acid and Acetic acid ratio.
1 Ensilaje
de maíz con inoculante microbiano liofilizado; 2 SAI = Ensilaje de maíz con
inoculante microbiano activado; 3 Relación Lat: Acet = Relación
de ácido láctico y ácido acético.
Quantification of lactic bacteria populations
The population count of
lactic acid bacteria was performed according to the recommendations of González and Rodrigues (2003). Twenty-five grams of
fresh silage samples were collected according to the defined opening periods,
and 225 ml of sterile ringer solution was added and processed in a blender for
approximately 1 minute. One millilitre of these mixtures was removed and
pipetted to an appropriate dilution (10-1 to 10-9).
Plating was performed in
duplicate for each culture medium. The populations were determined by the
selective culture technique in anaerobic medium, where the culture medium MRS
was used and incubated for 48 hours in an oven at 37°C, according to the
methodology of De Man et al. (1960).
The plaques considered
susceptible to counting were those in which there were values between 30 and
300 cfu in a Petri dish. The plate means of the selected dilutions were then
considered.
Lactic bacteria culture technique
After quantification of
lactic acid bacteria populations using the pour plate technique in agar MRS
culture (5), cultures were purified
and cultured in a Falcon tube with 5 mL of MRS broth for 24 h at 37°C, and 10
cfu of each treatment was randomly selected 70 days after ensiling. The
cultures were centrifuged for 10 minutes at 3600 rpm (rotation per minute) to
obtain the cell pellet. The supernatant was removed, and 1 mL of saline
(Ringer’s solution) was added and vortexed. Using a pipette, the entire volume
of the pellet was transferred to the microtube and centrifuged at 6000 rpm for
3 minutes. The microtubes were stored in a freezer until DNA extraction was
performed.
DNA extraction
DNA extraction from the
isolates was performed using the Wizard Genomic DNA Purification Kit (Promega).
The pellet was resuspended in 480 μl of 50 mM EDTA (Ethylic DiaminoTetracyclic
Acid) in vortex. Fifty microlitres of lysozyme (50 mg / ml concentration) were
added. The samples were incubated in a water bath at 37°C for 60 minutes and
centrifuged for 2 minutes at 12,000 RPM, and the supernatant was removed.
After DNA extraction, DNA
quantification procedures were carried out using Nanovue equipment (Nanodrop)
at the Animal Biotechnology Laboratory of the Animal Science Department of the
Federal University of Paraiba.
On average, the extraction
resulted in a concentration of 1161.37 ng of DNA with 1.9 degrees of purity.
After quantification, it was diluted to a concentration of 20 ng of DNA per μL.
Polymerase chain reaction (PCR)
Amplification of the 16S
rDNA fragment of the isolates occurred with Primer 1492R (TAG G(C/T)A CCT TGT TAC GAC TT) and Primer p027F (GAG AGT TGA TCC TGG
CTC AG) (Heuer et al., 1997). The PCR reaction was performed in 0.2 mL
tubes containing 50 μL of the reaction mixture: DNA (80 ng), 5X buffer solution
(0.1 mol/L Tris-HCl, pH 8.0, 0.5 mol/L KCl), 1.5 mmol/L MgCl2,
pH 8.0; 0.2 mmol/L dNTP mix (Promega), Taq polymerase 2U (Promega), 0.12 μmol/L
primer p027F 0.12 μmol/L, and 1429R 0.12 μmol/L (IDT Síntese Biotecnologia).
The volume of the reaction mixture was filled to 50 μL with sterile ultrapure
water. PCR was performed in a thermocycler 3Prime (Techne), and the reaction
conditions employed in the PCR were: 94°C/5 min; 30 cycles (denaturation: 94°C
for 30 seconds; 60°C for 30 seconds); polymerisation: 72°C/2 min; final
extension: 72°C/5 min. An aliquot of 3 μl of the PCR product was mixed in 3 μl
of the mixture: 1 μL Gel Red (Biotium) and 2 μL of pigment 6X Gel Loading
(Promega) and analysed using agarose gel electrophoresis (1.2%) in buffer
solution Tris-Borate-EDTA (TBE 1X). The gel was visualised under ultraviolet
light, and images were captured using a gel system for photo documentation
(MBS). The PCR product, a fragment of approximately 1500 bp, was sent to the
Macrogen Company, Korea, for purification and sequencing.
Sequence analysis of the isolates
The sequences of the
isolates were compared with those available in the GenBank database and aligned
using the BLASTn algorithm (Basic Local Alignment Search Tool) (http://www.ncbi.nlm.nih.gov/BLAST) for nucleotides. Sequences of the 16S rRNA gene that
presented similarity equal to or greater than 95% were considered to belong to
the same Operational Taxonomic Unit (OTU) (1).
Experimental design and statistical analysis
The experiment was carried
out in a completely randomised design, with 3 treatments and 5 replicates per
treatment, in each opening period (1, 3, 7, 14 and 70 days). The microbial
counts were transformed into log10. Variance analysis and multiple comparisons
of data were performed using the GLM procedures in SAS. The means were compared
using the Kruskal-Wallis test.
Data on culture, DNA extraction,
PCR and sequence analysis were performed and discussed through descriptive
statistical analysis.
Results
Quantification of lactic acid bacteria in silage
Quantification of lactic
acid bacteria populations was estimated at the different opening periods of the
silos (1, 3, 7, 14 and 70 days). The lactic acid bacteria showed a rapid
multiplication speed, reaching their highest development at 7 days of the
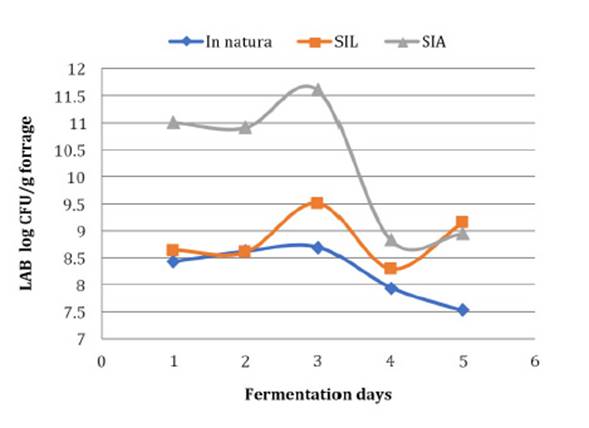
fermentation process (Figure 1).
SLI = Silage treated with the lyophilised inoculant; SAI = Silage
treated with the activated inoculant.
SLI =
Ensilado tratado con inoculante liofilizado; SAI = Ensilaje tratado con
inoculante activado.
Figure 1: Mean growth values of
lactic acid bacteria in corn silage in natura and inoculated with Lactobacillus
buchneri during different fermentation periods.
Figura 1: Valores medios de
crecimiento de bacterias lácticas en ensilaje de maíz in natura e
inoculado con Lactobacillus buchneri durante diferentes períodos de
fermentación.
Identification of lactic acid bacteria populations in silage
It was observed in this study
that a greater number of the isolates (93.34%) were formed by lactic
acid-producing strains of the species L. plantarum (Table 3).
Table 3: Molecular identification of samples of bacteria of the genus Lactobacillus
isolated 70 days after ensilage.
Tabla 3: Identificación molecular de muestras de bacterias del género
Lactobacillus aisladas a los 70 días del ensilaje.
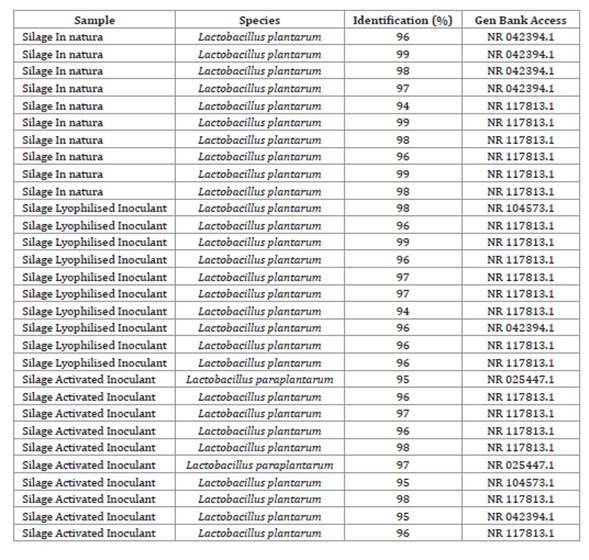
With the sequencing of the 16S
rDNA fragment produced by PCR using Primers 1492R and p027F, the isolates were
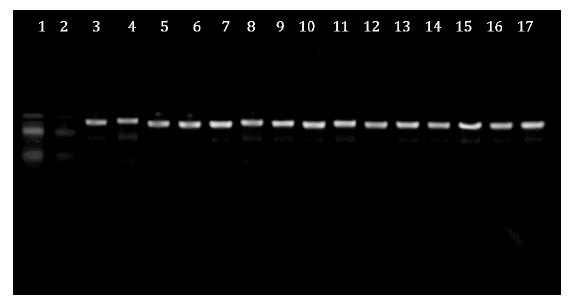
closely related to L. plantarum. Figure 2, shows that the PCR products were visualised using agarose
gel electrophoresis.
Figure 2: PCR amplification products
for Lactobacilli. Line 1, Molecular marker (100 bp DNA ladder); Line 2, White;
Lines 3 to 17, Lactobacillus plantarum.
Figura 2: Productos de amplificación por PCR para Lactobacilli. Línea 1.
Marcador molecular (escalera de DNA de 100 pb); Línea 2. Blanco; Líneas 3 a 17:
Lactobacillus plantarum.
Discussion
The silage inoculated with
preactivated L. buchneri had the highest values of lactic
bacteria populations until the 14th day of the fermentation
period, reaching 11 log CFU / g in the forage at 7 days of fermentation (Figure 1). These results corroborate those observed by de Santos et al. (2008) since there was the
addition of a larger number of active bacteria, thus facilitating their
multiplication. An indication of the presence of heterofermentative bacteria
was the reduction of lactic acid concentration, the increase in acetic acid
concentration and the reduction of the proportion of lactic and acetic acids.
Consequently, the ethanol levels were low at the end of the 70 days of silage (6).
However, the addition of a
group of microorganisms as additives at the end of the silage process may not
be present. These microorganisms promote changes in the pH value, and the
presence of several organic compounds, such as acetic, lactic, butyric,
propionic, ethanol, CO2,
and antimicrobial and/or bacteriostatic compounds, also affect the interaction
between the various groups of microorganisms (14).
These modifications were
desirable considering that the pre-silage material had a dry matter content
(26.2%) below that recommended for good-quality silage (16). According to Danner et al. (2003),
lactic acid formation is fundamental to reducing the pH of the silage, causing
the selection of microorganisms in the silage mass. However, acetic acid also
needs to be formed to ensure that the silage mass has an adequate pH of 3.8-4.2
(16), thus providing control
of moulds and yeasts. However, when the pH value is reduced too much, it may favour
the development of these microorganisms, which causes a decrease in aerobic
stability (20).
Although there was no reduction
in yeast in the corn silage with lyophilised or activated microbial inoculant,
the presence of acetic acid has antifungal action, which reduces the production
of other compounds, such as ethanol (22, 36),
as observed in this study. According to Sadiq et al. (2019),
there is a consensus from the scientific community that organic acids are the
main antifungal metabolites of LAB.
Organic acids in their
dissociated or undissociated form are lipophilic in nature and thus readily
diffuse across the fungal cell membrane and accumulate in the cytoplasm,
reducing activity or leading to yeast death. However, acetic acid has higher
inhibitory activity against fungal growth compared to lactic acid. Although the
LAB populations of the silages treated with the lyophilised inoculant showed
lower growth compared with the activated inoculation, they stabilised at 70
days with populations above 9.0 log cfu/g forage. The values were similar in
silage with the inoculant and higher than in silage without the inoculants (Figure 1). This difference may be due to a peak in LAB development
at 7 days of silage, when there may have been intensified substrate
competition. Probably, the activation of the lyophilised LAB with RSM caused
this difference in the population on the 7th day of silage, which remained
until the 70th.
The fermentative process
of silage is complex and involves many species of LAB and their interactions.
The use of specific inoculants is indicated to dominate or overcome the number
of epiphytic lactic bacteria present in the forage, either to improve the
fermentation process (19, 32) or to
increase the aerobic stability of silage (37). However, the increase in LAB in the ensiled mass promotes the
greater availability of specific microorganisms. The greater or lesser degree
of development of these bacteria depends on the conditions of the medium (9).
Inoculation of the silage
with activated heterofermentative LAB (L. buchneri), facultative
aerobes, produces lactic acid and acetic acid. These characteristics cause a
rapid proliferation of the same in the ensiled mass and cause an adequate
reduction of the silage pH, controlling the growth of yeasts and moulds (Table 2). Thus, the ensiled mass can provide greater development
of epiphytic bacteria, such as L. plantarum, which is
homofermentative (15, 28).
The pH of the ensiled mass
rapidly decreased due to the homofermentative LAB multiplying rapidly,
producing more lactic acid. Lactobacillus buchneri bacteria limit the
growth and metabolism of lactic acid degradation as a strategy to maintain cell
viability (Table
2) (20, 29).
The activity of L. buchneri was evidenced by higher acetic acid
production. In the 70-day fermentation period, the selected isolates of
inoculated corn silage were not identified (Table 3). Possibly due to the greater fermentative capacity in the
corn silages, L. plantarum dominated the fermentation of the
silage. Guo et al. (2018) observed similar
behaviour in alfalfa silage, including L. buchneri, which is
tolerant to acidic environments and uncompetitive compared to other LAB
species.
The high frequency of L.
plantarum in corn silage promoted the effective fermentation of lactic
acid, rapidly reducing the pH of the ensiled mass and preventing the
development of microorganisms deleterious to silage, such as Clostridia and
enterobacteria. In addition to decreasing the pH from lactic acid production, L.
plantarum can also inhibit the growth of filamentous moulds through the
production of antifungal activity (2). However, this characteristic is more likely for
heterofermentative LAB species (25).
In this study, strains of Lactobacillus
paraplantarum (6.6% of isolates) were also identified in corn silage
inoculated with RSM pre-activated bacteria (Table 3). Zhang et al. (2017) observed the presence and
predominance of L. paraplantarum in LAB populations in corn
silage with high humidity.
Wang et
al. (2017), characterising isolated lactic acid bacteria and their
effects on the fermentation of silage, verified the growth of L. paraplantarum
limited to 10°C and pH 3.0. According to the authors, the growth of L.
paraplantarum strains in low pH and temperature environments confirmed
the resistant and acidic nature of this species. At the end of the 70-day
silage period, the dynamics of the microbial population provided the prevalence
of the largest population of L. lantarum among LAB. These
findings corroborate the findings of Guo et al. (2018),
who reported that alfalfa silage inoculated with L. buchneri changed
the population profile of the epiphytic LAB species on the 60th day after
silage. The same authors stated that the L. plantarum population
predominated by more than 90%, but they failed to confirm the cause of this
change with certainty and suggested more studies related to the metabolomics of
LAB.
Conclusions
The application of
lyophilised or activated L. buchneri improved the microbiological
profile and reduced ethanol production in corn silages, even without being
identified among the isolates isolated 70 days after ensilage.
Acknowledgments
This research was supported by
the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil
(CAPES) finance code 001.
1.
Altschul, S. F.; Gish, W.; Miller, W.; Myers, E. W.; Lipman, D. J. 1990. Basic
local alignment search tool. Journal of Molecular Biology. 215(3): 403-410.
2.
Arasu, M. V.; Jung, M. W.; Ilavenil, S.; Jane, M.; Kim, D. H.; Lee, K. D.;
Park, H. S.; Huh, T. Y.; Choi, G. J.; Lim, Y. C.; Al-Dhabi, N. A.; Al-Dhabi, K.
C. 2013. Isolation and characterization of antifungal compound from Lactobacillus
plantarum KCC-10 from forage silage with potential beneficial properties.
Journal Applied Microbiology. 115(8): 1172-1185. https://doi.
org/10.1111/jam.12319
3.
Brito, G. S. M. S.; Santos, E. M.; Araújo, G. G. L.; Oliveira, J. S.; Zanine,
A. M.; Perazzo, A. F.; Campos, F. S.; Oliveira, J. S.; Lima, A. G. V. O.;
Cavalcanti, H. S. 2020. Silagens mistas de pêra de cacto e gliricídia:
composição química, características de fermentação, população microbiana e
estabilidade aeróbica. Scientific Reports. 10(1): 6834-6844.
https://doi.org/10.1038/s41598-020-63905-9
4.
Danner, H.; Holzer, M.; Mayrhuber, E.; Braun, R. 2003. Acetic acid increases
stability of silage under aerobic conditions. Applied and Environmental
Microbiology. 69(1): 562-567. https:// dx.doi.org/10.1128/AEM.69.1.562-567.2003
5.
De Man, J. C.; Rogosa, D.; Sharpe, M. E. 1960. A medium for the cultivation of
lactobacilli. Journal of Applied Bacteriology. 23(4): 130-135.
https://doi.org/10.1111/j.1365-2672.1960.tb00188.x
6.
dos Santos, A. P. M.; Santos, E. M.; Silva de Oliveira, J.; Pinto de Carvalho,
G. G.; Garcia Leal de Araújo, G.; Moura Zanine, A.; Martins Araújo Pinho, R.;
Ferreira, D. de J.; da Silva Macedo, A. J.; Pereira Alves, J. 2021. Effect of
urea on gas and effluent losses, microbial populations, aerobic stability and
chemical composition of corn (Zea mays L.) silage. Revista de la
Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza.
Argentina. 53(1): 309-319.
7.
Ferreira, D. J.; Zanine, A. M.; Lana, R. P.; Souza, A. L.; Negrão, F. M.;
Geron, L. J. V.; Parente, H. N.; Parente, M. O. M.; Pinho, R. M. A. 2019.
Fermentation and chemical composition of Marandu grass ensiled with dehydrated
brewery residue. Grassland Science. 65(8): 69-72. https://doi.
org/10.1111/grs.12210
8.
Gandra, J. R.; Oliveira, R. R.; Gandra, E. R. S.; Takiya, C. S.; Goes, R. H. T.
B.; Oliveira, K. M. P.; Silveira, K. A.; Araki, H. M. C.; Orbach, N. D.;
Vasquez, D. N. 2017. Inoculation of Lactobacillus buchneri alone or with
bacillus subtilis and total losses, aerobic stability, and microbiological
quality of sunflower silage. Journal Applied Animal Research. 45(10): 609-614.
https:// doi.org/10.1080/09712119.2016.1249874
9.
Giraffa, G.; Chanishvili, N.; Widyastuti, Y. 2010. Importance of lactobacilli in
food and feed biotechnology. Research in Microbiology. 161(3): 480-487.
http://dx.doi.org/10.1016/j.resmic.2010.03.001
10.
González, G.; Rodríguez, A. A. 2003. Effect of storage method on fermentation
characteristics, aerobic stability and forage intake of tropical grasses
ensiled in round bales. Journal of Dairy Science. 86(8): 926-933.
https://doi.org/10.3168/jds.S0022-0302(03)73675-3
11.
Guo, X. S.; Ke, W. C.; Ding, W. R.; Ding, L. M.; Xu, D. M.; Wan, W. W.; Zhang,
P.; Yang, F. Y. 2018. Profiling of metabolome and bacterial community dynamics
in ensiled Medicago sativa inoculated without or with Lactobacillus
plantarum or Lactobacillus buchneri. Scientific Reports. 8(1):
357-365. https://doi.org/10.1038/s41598-017-18348-0
12.
Heuer, H.; Krsek, M.; Baker, P.; Smalla, K.; Wellington, E. M. 1997. Analysis
of actinomycete communities by specific amplification of genes encoding 16S
rRNA and gel-electrophoretic separation in denaturing gradients. Applied and
Environmental Microbiology. 63(11): 3233-3241.
13.
Kermanshahi, R. K.; Peymanfar, S. 2012. Isolation and identification of
lactobacilli from cheese, yoghurt and silage by 16S rDNA gene and study of
bacteriocin and biosurfactant production. Jundishapur Journal of Microbiology.
5(1): 528-532. https://doi.org/10.5812/JJM.3444
14.
Li, D.; Nia, K.; Pang, H.; Wang, Y.; Cail, Y.; Jin, Q. 2015. Identification and
antimicrobial activity detection of lactic acid bacteria isolated from corn
stover silage. Asian-Australasian Journal Animal Sciences. 28(9): 620-631.
https://dx.doi.org/10.5713/ajas.14.0439
15.
Li, Y.; Nishino, N. 2011. Effects of inoculation of Lactobacillus rhamnosus and
Lactobacillus buchneri on fermentation, aerobic stability and microbial
communities in whole crop corn silage. Grassland Science. 57(5): 184-191.
https://doi.org/10.1111/j.1744-697X.2011.00226.x
16. McDonald, P.; Henderson, A. R.; Heron, S. 1991. The
biochemistry of silage. Marlow: Chalcombe.
17.
Muck, R. E. 2010. Silage microbiology and its control through additives.
Revista Brasileira de Zootecnia. 39(2): 183-191.
https://doi.org/10.1590/S1516-35982010001300021
18.
Nishida, S.; Ishii, M.; Nishiyama, Y.; Abe, S.; Ono, Y.; Sekimizu, K. 2017. Lactobacillus
paraplantarum 11-1 isolated from rice bran pickes activated innate immunity
and improved survival in a silkworm bacterial infection model. Frontiers in
Microbiology Food Microbiology. 8(4): 1-8.
https://doi.org/10.3389/fmicb.2017.00436
19.
Oliveira, A. S.; Weinberg, A. G.; Cervantes, A. A. P.; Arriola, K. G.; Jiang,
Y.; Kim, D.; Li, X.; Gonçalves, M. C. M.; Vyas, D.; Adesogan, A. T. 2017.
Meta-analysis of effects of inoculation with homofermentative and facultative
heterofermentative lactic acid bacteria on silage fermentation, aerobic
stability and the performance of dairy cows. Journal of Dairy Science. 100(12):
1-17. https://doi.org/10.3168/jds.2016-11815
20.
Oude, F.; Elferink, S. J. W. H.; Krooneman, J.; Gottschal, J. C.; Spoelstra, S.
F.; Faber, F.; Driehuis, F. 2001. Anaerobic conversion of lactic acid to acetic
acid and 1,2-propanediol by Lactobacillus buchneri. Applied and
Environmental Microbiology. 67(3): 125-132. http://
dx.doi.org/10.1128/AEM.67.1.125-132.2001
21.
Pahlow, G. R. E.; Muck, F.; Oude Elferink, S. J. W. H.; Spoesltra, S. F. 2003.
Microbiology of ensiling. In D. R. Buxton, R. E. Muck & J. H. Harrison
(Eds.). Silage Science and Technology (p. 31-94). Madison: American Society of
Agronomy. Crop Science Society of America. Soil Science Society of America.
22.
Pedroso, A. F.; Nussio, L. G.; Paziani, S. F. 2005. Fermentation and epiphytic
microflora dynamics in sugar cane silage. Scientia Agricola. 62(10): 427-432.
https://doi.org/10.1590/S0103-90162005000500003
23.
Pereira, G.; Santos, E.; Araújo, G.; Oliveira, J.; Pinho, R.; Zanine, A. M.;
Souza, A. L.; Macedo, J. M. C.; Nascimento, T. V. C. 2020. Isolation and
identification of lactic acid bacteria in fresh plants and in silage from Opuntia
and their effects on the fermentation and aerobic stability of silage.
Journal of Agricultural Science. 3(1): 1-9.
https://doi.org/10.1017/S0021859620000143
24.
Queiroz, O. C. M.; Arriola, K. G.; Daniel, J. L. P.; Adesogan, A. T. 2013.
Effects of 8 chemical and bacterial additives on the quality of corn silage.
Journal of Dairy Science. 96(8): 5836-5843. https://
doi.org/10.3168/jds.2013-6691
25.
Sadiq, F. A.; Yan, B.; Tian, F.; Zhao, J.; Zhang, H.; Chen, W. 2019. Lactic
acid bacteria as antifungal and anti‐mycotoxigenic agents:
Comprehensive reviews in food science and food safety. Food Sci. Food Saf.
18(5): 1403-1436. https://doi.org/10.1111/1541-4337.12481
26.
Santos, A. O.; Ávila, C. L. S.; Pinto, J. C.; Carvalho, B. F.; Dias, D. R.;
Schwan, R. F. 2016. Fermentative profile and bacterial diversity of corn
silages inoculated with new tropical lactic acid bacteria. Journal of Applied
Microbiology. 120(2): 266-279. https://doi.org/10.1111/jam.12980
27.
Santos, E. M.; Zanine, A. M.; Ferreira, D. J.; Oliveira, J. S.; Penteado, D. C.
S.; Pereira, O. G. 2008. Inoculante ativado melhora a ilage de capim-tanzânia
(Panicum maximum). Archivos de Zootecnia. 57(7): 35-42.
28.
Santos, E. M.; Silva, T. C.; Macedo, C. H. O.; Campos, F. S. 2013. Lactic acid
bactéria in tropical Grass silages. In Lactic Acid Bacteria - R & D for
Food, Health and Livestock Purpose. IntechOpen.
29.
Schmidt, R. J.; Hu, W.; Mills, J. A.; Kung Jr, L. 2009. The development of
lactic acid bacteria and Lactobacillus buchneri and their effects on the
fermentation on alfalfa silage. Journal of Dairy Science. 92(1): 5005-5010.
https://doi.org/10.3168/jds.2008-1701
30.
Schmidt, R. J.; Kung Jr., L. 2010. The effects of Lactobacillus buchneri with
or without a homolactic bacterium on the fermentation and aerobic stability of
corn silages made at different locations. Journal of Dairy Science.
93(9): 1616-1624. https://doi.org/10.3168/jds.2009-2555
31.
Silva, M. A.; Edvan, R. L.; Parente, H. N.; Zanine, A. M.; Pereira Filho, J.
M.; Santos, E. M.; Oliveira, J. P. F.; Araujo, M. J.; Bezerra, L. R. 2020.
Addition of fermented corn juice as bioinoculant improved quality of Saccharum
officinarum silage. International Journal of Agriculture and Biology.
23(1): 349-356. http://dx.doi.org/10.17957/IJAB/15.1295
32.
Zanine, A. M.; Bonelli, E. A.; Ferreira, D. J.; Santos, E. M.; Pinho, R. M. A.;
Parente, H. N.; Parente, M. O. M. 2018. Fermentation and chemical composition
of guinea grass silage added with wheat meal and Streptococcus bovis.
New Zealand Journal of Agricultural Research. 1(12): 1-8.
https://doi.org/10.1080/00288233.2018.1445651
33.
Zhang, M.; Wang, Y.; Tan, Z.; Li, Z.; Li, Y.; Lv, H.; Zhang, B.; Jin, O. 2017.
Microorganism profile, fermentation quality and rumen digestibility in vitro of
maize-stalk silages produced at different maturity stages. Crop Pasture
Science. 68: 225-233. https://doi.org/10.1071/CP16324
34.
Yitbarek, M. B.; Tamir, B. 2014. Silage additives: Review. Open Journal of
Applied Sciences. 4(05): 258-274. http://dx.doi.org/10.4236/ojapps.2014.45026
35.
Wang, S.; Yuan, X.; Dong, Z.; Li, J.; Guo, G.; Bai, Y.; Zhang, J.; Shao, T.
2017. Characteristics of isolated lactic acid bacteria and their effects on the
silage quality. Asian-Australasian Journal of Animal Science. 30(5): 819-827.
https://doi.org/10.5713/ajas.16.0589
36. Weinberg, Z. G.; Khanal, P.; Yildiz, C.; Chen, Y.; Arieli, A.
2011. Ensiling fermentation products and aerobic stability of corn and sorghum
silages. Grassland Science. 57(2): 46-50.
37.
Weiss, K.; Kroschewski, B.; Auerbach, H. 2016. Effects of air exposure,
temperature and additives on fermentation characteristics, yeast count, aerobic
stability and volatile organic compounds in corn. Journal of Dairy Science.
99(10): 8053-8069. https://doi.org/10.3168/jds.2015-10323 .
Conflict
of interest statement
The authors had no conflict of interest. .