Revista de la Facultad de Ciencias
Agrarias. Universidad Nacional de Cuyo. Tomo 54(2). ISSN (en línea) 1853-8665.
Año 2022.
Original article
Advances
in the etiology of sweet potato (Ipomoea batatas (L.) Lam) yellow curling disease in Argentina
Avances en la etiología de la enfermedad del
encrespamiento amarillo de la batata (Ipomoea batatas (L.) Lam)
en Argentina
Andrés Vicente Luque1
Patricia Elsa Rodríguez Pardina1
Liliana del
Valle Di Feo1
1 Unidad de Fitopatología y Modelización Agrícola (INTA-CONICET). Instituto
de Patología Vegetal (IPAVE). Centro de Investigaciones Agropecuarias (CIAP).
Instituto Nacional de Tecnología Agropecuaria (INTA).
*flamarique.sofia@inta.gob.ar
**vilanova.antonella@inta.gob.ar
Abstract
Sweet potato
yellow curling (YC), the most severe disease of sweet potato detected in
Argentina, causes symptoms and damage to sweet potato crops in all cultivated
regions. Since 2010/11, the presence of four viruses has been detected in
symptomatic cv. Arapey INIA: two potyviruses non-persistently transmitted by Myzus
persicae (sweet potato feathery mottle virus, SPFMV and sweet potato virus
G, SPVG); a closterovirus, sweet potato chlorotic stunt virus (SPCSV)
and a geminivirus, sweet potato leaf curl virus (SPLCV), both transmitted by Bemisia
tabaci in a semi-persistent and persistent manner, respectively. All the
plants were collected from fields in Colonia Caroya, Córdoba province,
Argentina. The objectives of the present work are to isolate and identify the
virus or viruses involved in YC disease of sweet potato, and to elucidate the
viral combination that reproduces YC symptoms. The most severe YC symptoms for
this genotype in the field were only reproduced by a combination of the four
viruses. The symptoms include chlorosis, stunting, mosaic, blistering, leaf
curling, chlorotic spots, chlorotic patterns, leaf area reduction and
distortion, and upward curling of leaf edges. The presence of each virus was
detected by serological (DAS, NCM and TAS-ELISA) and molecular (PCR) tests. It
is concluded that the interaction of SPFMV, SPVG, SPCSV and SPLCV is needed for
the development of YC symptoms.
Keywords: Ipomoea batatas; Sweet potato feathery mottle virus; Sweet potato virus G; Sweet
potato leaf curl virus; Sweet potato
chlorotic stunt virus; Arapey INIA; Koch’s postulates.
Resumen
El encrespamiento amarillo (EA), la enfermedad más severa detectada en
Argentina, causa síntomas y daños en cultivos de batata en toda la región
productora. Desde 2010/11 se ha detectado la presencia de cuatro virus en
plantas sintomáticas del cv. Arapey INIA recolectadas en lotes de Colonia
Caroya, provincia de Córdoba. Los virus son sweet potato feathery mottle virus
(SPFMV) y sweet potato virus G (SPVG), dos potyvirus transmitidos de forma no
persistente por Myzus persicae; un closterovirus: sweet potato chlorotic
stunt virus (SPCSV) y un geminivirus: sweet potato leaf curl virus (SPLCV),
ambos transmitidos por Bemisia tacabi de manera semipersistente y
persistente, respectivamente. Los objetivos de este trabajo fueron aislar e
identificar el o los virus involucrados en la enfermedad EA de la batata y
determinar la combinación de virus que reproduce la sintomatología de EA Solo
la combinación de los cuatro virus permitió reproducir la sintomatología más
severa del encrespamiento amarillo observada a campo en dicho genotipo. Los
síntomas incluyen clorosis, achaparramiento, mosaico, ampollado, enrulado de la
hoja, manchas cloróticas, diseños cloróticos, reducción y distorsión del área
foliar, bordes de la hoja curvados hacia arriba. La presencia de cada uno de
los virus se detectó mediante pruebas serológicas (DAS, NCM y TAS-ELISA) y
moleculares (PCR). Se concluye que la interacción de SPFMV, SPVG, SPCSV y SPLCV
es necesaria para el desarrollo de EA.
Palabras
claves: Ipomoea batatas; Ssweet
feathery mottle virus; Sweet potato virus G; Sweet potato leaf curl virus; Sweet potato chlorotic stunt virus; Arapey INIA; Postulados de Koch.
Originales: Recepción: 17/02/2022
Aceptación:
18/10/2022
Introduction
Sweet potato, Ipomoea batatas (L.) Lam, is a
perennial plant of the Convolvulaceae family, native to northwestern
South America (6, 16). It is ranked
among the 10 most important food crops worldwide, with a production of
89,487,835 tons in an area of 7,400,472 hectares in 2020 (8). The major sweet potato production areas are
located in China and around the Great Lakes of East Africa, but the crop is
also important in many other countries (6, 20). According to the International Potato
Center, developing countries concentrate 95% of the production, but with
low yields. Latin America produces only 1.5 million tons (8).
Viral diseases
are considered a major constraint to sweet potato production worldwide (22, 38,
40), due to the vegetative
propagation of this species and the consequent cumulative effect of virus
infections (17, 29). The first
report of a sweet potato viral disease in Argentina was published in the 1970s.
This disease, named “batata crespa”, affected cv. Criolla Amarilla and was
caused by the potyvirus sweet potato vein mosaic virus (SPVMV) (30). Later, sweet potato feathery mottle virus
(SPFMV) was detected in the same cultivar (31). In 1978, Argentine farmers adopted a new
cultivar, Morada INTA, tolerant to both potyviruses (SPVMV and SPFMV). However,
during the 1980s, this new cultivar was affected by a severe disease called
sweet potato chlorotic dwarf, caused by a synergistic combination of two
aphid-transmitted potyviruses (SPFMV and sweet potato mild speckling virus,
SPMSV) with a whitefly-transmitted closterovirus serologically related to sweet
potato chlorotic stunt virus (SPCSV) (7). Sweet potato chlorotic dwarf was successfully
controlled using propagation material obtained from disease-free areas.
Nevertheless, in all sweet potato-producing areas of Argentina, cv. Morada INTA
has been progressively replaced by cv. Arapey INIA, an
Uruguayan genotype (7).
In 2010/11, a
new viral disease with severe and variable symptoms was detected in this
cultivar; it was named sweet potato yellow curling disease (YC). YC causes
significant yield losses, with records of up to 90% loss in experimental trials
and commercial plots. In addition, YC affects the quality of the storage roots (23).
Serological tests confirmed the
presence of SPFMV (4) and SPCSV (Unpublished
data) in symptomatic plants. Moreover, in these plants, two pathogens that had
not been described in Argentina at that time, sweet potato virus G (SPVG), a
potyvirus (33) and the geminivirus,
sweet potato leaf curl virus (SPLCV) (34), were detected through
serological tests and PCR, respectively.
Since YC is
presumed to be caused by a viral complex, this work aimed to isolate and identify
the virus or viruses involved in YC disease of sweet potato and to elucidate
the viral combination that reproduces YC symptomatology.
Material and methods
Source of inoculum
More than 100
plants of sweet potato cv. Arapey INIA showing chlorosis, stunting, mosaic,
chlorotic rings, chlorotic patterns, blistering, distortion, reduction of
foliar area, and upward leaf curling were randomly collected from 12 fields in
Colonia Caroya (31°01’16.8’’ S 64°03’42.2’’ W), Colón department, Córdoba
province, Argentina. Plants were transplanted to pots and maintained in the
greenhouse at 25°C under controlled conditions of humidity (70-75% RH) and
photoperiod (16 h light) for further analysis.
Transmission assays
Apterous aphids from a healthy colony of Myzus persicae Sulz. were reared on Raphanus sativus L. and used for
transmission studies. Fasted aphids were allowed to probe briefly (one probe)
on plants of sweet potato cv. Arapey INIA showing typical YC symptoms. Aphids
were transferred to healthy individuals of the indicator plant Ipomoea
setosa (one aphid per each one of 44 plants) and allowed to feed for 12 h;
then they were killed with an aphicide (Pirimicarb) (7, 13). A repetition was performed using sweet
potato cv. Okinawa 100 plants infected with SPVG, SPCSV and SPLCV. The
procedure was similar to the one described previously but involved 46 I.
setosa plants.
To separate
SPCSV from SPLCV (semi-persistent and persistent transmission, respectively),
non-viruliferous Bemisia tabaci Gennadius whiteflies reared on
poinsettia (Euphorbia pulcherrima Willd.), a sweet potato plant cv.
Arapey infected with YC, and 42 healthy seedlings (second unfolded leaf) of I.
setosa were placed in protected cages with anti-aphid mesh, at 25ºC, in a
greenhouse, under controlled conditions of humidity (70-75% RH) and photoperiod
(16h light). The whiteflies were removed from the cages after 1h and up to 48h
of exposure to insect feeding. At 21 days after inoculation, the plants were
checked for the presence of SPCSV and SPLCV by TAS-ELISA and PCR, respectively (25).
Verification
of SPFMV, SPVG, SPCSV and SPLCV presence
Infection by
SPFMV, SPVG and SPCSV was checked by serological tests on all inoculated
plants. A local antiserum was used for SPFMV detection. The SPVGas and SPCSVas
were provided by S. Fuentes (International Potato Center, Lima, Peru) and H. J.
Vetten (Federal Biological Research Center, Institute of Biochemistry, Agriculture,
and Forestry, Braunschweig, Germany), respectively (7, 9). SPLCV was tested by PCR (34). The production of local SPVGas is discussed
below.
SPVG virus purification and antiserum production
After SPVG
isolation, virus particles were purified from I. setosa-infected leaves
following the method described by Di Feo et al. (2000).
The virus band was collected from a sucrose-CsCl step gradient (0 to 41% CsCl
in borate buffer containing 20% sucrose) after ultracentrifugation (100000 x g
for 5 h at 8°C), and dialyzed against 0.05 M borate buffer. Purified virus
preparation of SPVG (0.02 mg/ml) was injected into a female New Zealand rabbit.
Three inoculations were performed at 20-day intervals. The first inoculation
(1ml of the purified virus + 2 ml of Freund’s complete adjuvant) was
administered by multiple intradermal injections, whereas the other two
inoculations (1 ml of the purified virus + 1 ml of incomplete Freund’s
adjuvant) were administered intramuscularly. Blood sample collection started 20
days after the last injection. The titers were evaluated by Nitrocellulose
membrane enzyme-linked immunosorbent assay (NCM-ELISA) (21, 32), Double-antibody sandwich enzyme-linked
immunosorbent assay (DAS-ELISA) (5) and Immunosorbent electron microscopy plus
decoration (ISEM+D) (26).
To evaluate the
optimal dilution to be used in NCM-ELISA, SPVG antiserum was serially diluted
from 1:500 to 1:1024000 in Tris saline buffer + 2% skimmed milk. Samples were
macerated in extraction buffer (TBS + 0.2% Na2SO3) in a 1/50 (w/v)
proportion and blotted onto a nitrocellulose membrane. Positive controls were I.
setosa plants infected with SPVG, sweet potato cv. Arapey INIA infected
with YC and sweet potato cv. Okinawa infected with the complex (SPVG + SPCSV +
SPLCV). Negative controls were healthy I. setosa and sweet potato
plants. In addition, a sample of I. setosa infected with SPFMV was
included to rule out the possible cross-reaction between the antiserum and this
potyvirus.
For the
DAS-ELISA test, IgG was purified from the SPVG antiserum. Enzyme conjugate was
prepared with the purified IgG and Phosphatase Alkaline VII S (SIGMA -Aldrich)
(10 mg protein/ml suspension of SO4 (NH)4;
30000 enzyme units/ml). DAS-ELISA test was performed using a NUNC polystyrene
plate with 96 flat-bottomed wells as support. Three dilutions of IgG (1:500,
1:1000 and 1:2000) were tested against three dilutions of enzyme conjugate
(1:500. 1:1000 and 1:1500). Samples were macerated in extraction buffer (PBS +
0.05% Tween 20 + 2% PVP+ 2% skimmed milk + 0.2% SO2Na2) in a 1/10 and a 1/1000
proportion, respectively. Positive and negative controls were sweet potato
plants infected with SPVG and healthy plants, respectively. Absorbance was
measured using a spectrophotometer (Dynatech Laboratories, Model MRX) at 405
nm.
ISEM+D was performed according to the protocol described by Milne & Lesemann (1978). Copper grids were first
covered with the SPVG-specific antiserum, diluted 1:1000 in TBS (20 mM Tris base,
500 mM NaCl, pH 7.5) and incubated for 30 min. Samples co-infected with SPVG +
SPCSV were macerated in TBS containing 0.2% sodium sulfite (1/10 w/v). For
decoration of virions, antiserum was diluted at 1:50 and incubated on grids for
15 min. Moreover, the modal length of virions obtained from purified
preparations was determined. Observations were made under an electron
microscope (JEOL JEM EX II 1220®).
Inoculation of healthy sweet potato plants with four viruses in
different combinations
This experiment
was designed to determine the viral combination(s) that reproduces YC in sweet
potato. Scions of I. setosa plants infected individually with each of
the four isolated viruses (SPFMV, SPVG, SPCSV and SPLCV) were double, triple
and quadruple side-grafted onto 10 healthy plants of cv. Arapey INIA (obtained
by “in vitro’’ meristem culture) and I. setosa, respectively. Six
healthy plants of cv. Arapey INIA grafted with scions from YC-affected plants
were used as controls.
Grafted plants
were kept in protected cages with anti-aphid mesh in a greenhouse at 25°C under
controlled conditions of humidity and photoperiod.
The presence of
SPFMV was determined by NCM-ELISA using local antiserum (7). To detect SPCSV, a triple-antibody sandwich
enzyme-linked immunosorbent assay (TAS-ELISA) was performed, as described by Gibson et al. (1998), using antisera provided by H.
J. Vetten (Federal Biological Research Center, Institute of Biochemistry,
Agriculture, and Forestry, Braunschweig, Germany). Infection with SPVG was
confirmed by NCM-ELISA using the local antisera produced in this study. The
presence of SPLCV was verified by PCR using the primers SPG1 and SPG2 designed
to bind to conserved regions in open reading frames C2 and C1, and to amplify a
912-bp fragment (18).
Results
Transmission assay
Only SPFMV was
transmitted to healthy I. setosa plants from sweet potato cv. Arapey
INIA infected with the YC complex. However, the frequency of isolation using
single aphids (M. persicae) was 77% for SPFMV and 13% for SPVG when the
inoculum source was sweet potato cv. Okinawa 100 infected with SPVG, SPCSV and
SPLCV. Virus sources were maintained by grafting onto I. setosa and
sweet potato cv. Arapey INIA in insect-proof cages in the greenhouse.
Transmission of SPCSV and SPLCV
from sweet potato plants infected with YC showed that one hour after the I.
setosa seedlings were removed from the cage, 100% of the plants were
infected with SPCSV and 0% with SPLCV. On the other hand, at 48 h, 100% of the
plants were infected with SPLCV and 33% with SPCSV. Those plants negative for
closterovirus were selected for further testing.
Virus purification and serology
SPVG was
purified from single-infected I. setosa plants. The A260/A280
ratio and viral concentration of the band extracted from the CsCl
gradient were 1.16 and 0.161 mg/ml, respectively. According to the NCM-ELISA
test, the optimal dilution of the antiserum was 1:512000, without
cross-reaction with SPFMV.
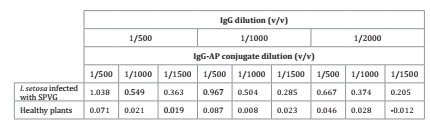
In DAS-ELISA,
optimal values of A405 were obtained after 90 minutes
of reaction. The most suitable dilutions of IgG and enzyme conjugate for virus
diagnosis were 1:1000 and 1:500, respectively (Table 1), whereas 1/10 w/v was the most appropriate dilution of
sweet potato plant tissue.
Table
1: Absorbance values (A405)
in DAS-ELISA using SPVGas.
Tabla 1: Valores de absorbancia (A405) en DAS-ELISA
utilizando SPVGas.
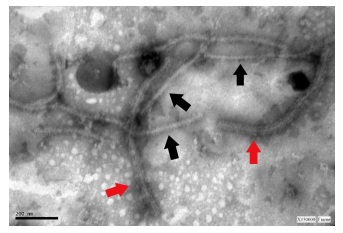
When the
SPVG antiserum was used for ISEM + Decoration tests, differentially decorated
virions were observed in samples of tissue infected with SPVG +SPCSV (Figure 1).
Observations were made under a transmission electron microscope
(JEOL JEM EX II 1220®) (X150000).
Observaciones realizadas al Microscopio electrónico de
transmisión (JEOL JEM EX II 1220®) (X150000).
Figure 1: Virus particles from tissue samples co-infected with SPVG + SPCSV.
ISEM+D coated 1/2000 with SPVG antiserum, decoration 1/50 (v/v), and contrasted
with 2% uranyl acetate (red arrow).
Figura 1: Viriones de SPVG de muestras de tejido coinfectado
con SPCSV, ISEM + D sensibilizado con antisuero SPVG diluido 1/2000, decorado
con antisuero diluido 1/50 (v/v), y contrastado negativamente con acetato de
uranilo al 2% (flecha roja).
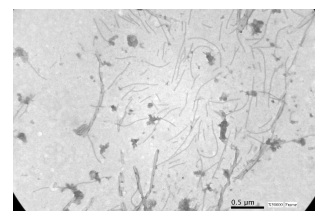
A modal length of 850-900 nm (150
virions) from purified suspensions was determined (Figure
2), which corresponds to the range of length
established for potyviruses (37).
The modal length (850- 900 nm) corresponds to the range of length
established for potyviruses.
La longitud modal (850- 900 nm) corresponde al rango de
longitud establecido para los potyvirus.
Figure 2: SPVG viral
particles from purified suspensions, observed under a transmission electron
microscope JEOL JEM EX II 1220® (X50000).
Figura 2: Partículas virales SPVG observadas al microscopio
electrónico de transmisión JEOL JEM EX II 1220® a partir de suspensiones
purificadas (X50000).
Inoculation
Symptoms caused
by each virus combination are shown in Table 2.
Table
2: Symptoms induced by
different combinations of sweet potato feathery mottle virus (SPFMV), sweet
potato virus G (SPVG), sweet potato chlorotic stunt virus (SPCSV) and sweet
potato leaf curl virus (SPLCV) in graft-inoculated Ipomoea setosa and
sweet potato (Ipomoea batatas) cv. Arapey INIA.
Tabla
2: Síntomas
inducidos por diferentes combinaciones de sweet potato feathery mottle virus
(SPFMV), sweet potato virus G (SPVG), sweet potato chlorotic stunt virus
(SPCSV) y sweet potato leaf curl virus (SPLCV) en plantas de Ipomoea setosa y
batata (Ipomoea batatas) cv. Arapey INIA inoculadas mediante injerto.
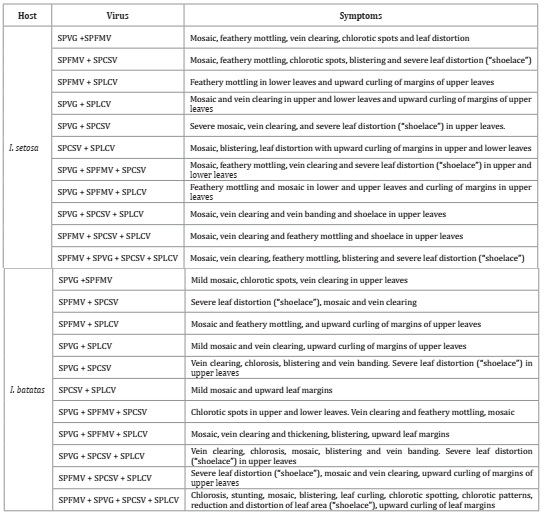
The mixed infections in which SPCSV was
present caused different degrees of severity.
The combination
of the four viruses induced the most severe symptomatology, resembling that of
YC-affected plants in the field (Figure 3):
chlorosis, stunting, mosaic, blistering, leaf curling, chlorotic spotting,
chlorotic patterns, reduction and distortion of leaf area, and upward curling
of leaf margins.
(A) Asymptomatic leaf (healthy plant); (B) SPFMV + SPVG, feathery
mottle and chlorotic spots; (C) SPFMV + SPVG + SPCSV, interveinal chlorosis,
chlorotic spots and upward curling of leaf margins; (E-H) SPFMV + SPVG + SPCSV
+ SPLCV, feathery mottle, mosaic, vein clearing, chlorotic spots, blistering,
upward curling of leaf margins, leaf area reduction and distortion.
(A) Hoja asintomática (planta sana); (B) SPFMV + SPVG,
moteado plumoso y punteado clorótico; (C) SPFMV + SPVG + SPCSV, clorosis
internerval, punteado clorótico y curvado de hojas; (E-H) SPFMV + SPVG + SPCSV
+ SPLCV, moteado plumoso, mosaico, aclaramiento de nervaduras, punteado
clorótico, ampollado, curvado de hojas, reducción y distorsión del área foliar.
Figure 3: Symptoms induced in cv. Arapey INIA by
different combinations of the viruses involved in sweet potato yellow curling
disease.
Figura 3: Síntomas observados en diferentes
combinaciones de los virus involucrados en encrespamiento amarillo cv. Arapey INIA.
Young plants of cv. Arapey INIA developed this
symptomatology 15 days after grafting with the combination of the four viruses.
On the
other hand, I. setosa became symptomatic 10 days after inoculation.
Symptoms varied from mosaic, vein clearing, feathery mottling and blistering to
severe leaf distortion (“shoelace”).
Discussion
Vegetative
propagation is a cultural practice that facilitates efficient virus
perpetuation and dissemination way between cropping seasons or growing areas (14). While several viral diseases have been
reported in sweet potato, YC is the most severe one reported in Argentina so
far. It is caused by at least four viruses and produces severe symptoms and
significant damage in all growing areas in the country (23).
The new disease
differs from the ones previously described in Argentina. Both “batata crespa”
and “sweet potato chlorotic dwarf disease” affected only sweet potato plots
from Santiago del Estero and Córdoba provinces. The former was caused by SPVMV,
and the latter, by three viruses: SPCSV + SPFMV + SPMSV (7, 30). Two viruses already present in Argentina
(SPFMV and SPCSV) and two new viruses (SPVG and SPLCV) are involved in YC.
Mixed viral
infections and synergistic complexes are frequent in sweet potato (36). SPFMV is the most common virus infecting
sweet potato and occurs in all sweet potato growing regions (27). SPCSV interacts synergistically with SPFMV
to cause sweet potato virus disease (SPVD), the most serious disease of sweet
potato (1, 12, 19, 29). The ubiquitous presence of SPFMV has often
masked the presence of other viruses in sweet potato, especially those
belonging to the same family, such as SPVG; thus, detecting or isolating them
is very difficult (35). It is widely
known that suitable management of viral diseases that affect the sweet potato
crop requires rapid and accurate detection (6). However, in previous works, it has been
shown that low titers of the viral agents and the high concentration of
inhibitors in the sweet potato plant hinder serological and molecular diagnosis
(11, 14). The antiserum
obtained allowed us to detect SPVG in single and mixed infections. In the
latter case, the detection was evident, without cross-reaction with SPFMV, a
ubiquitous virus in sweet potato crops (39). Detection reagents are available for local
species involved in YC, such as SPVGas, which is used for routine diagnosis in
IPAVE.
On the other
hand, geminiviruses that infect sweet potato are widely distributed throughout
the world. Twelve viruses belonging to the Geminiviridae family have
been reported (6, 15, 28). They can
cause significant losses in the production and quality of certain sweet potato
genotypes, without noticeable symptom expression (41). SPLCV has been detected in several
locations worldwide, including Taiwan, Japan, Israel, and the United States.
The geographical range of this virus, however, is still mostly unknown (14). Leaf curl disease associated with SPLCV was
first reported in Argentina in 2012 (34). Sweet potato leaf curl Georgia virus
(SPLCGV) was also reported in Argentine sweet potatoes (24). The presence of both pathogens is
associated with an increase in whitefly populations (3). Therefore, it is necessary to study the
dispersion of the new viral species, since climate change causes the
geographical expansion of vectors such as whiteflies (2, 3, 10).
The global
exchange of sweet potato germplasm contributes to the wide distribution of the
viruses in the sweet potato production regions (19). One effective way to prevent the spread of
viruses and, therefore, control viral diseases is to use virus-tested planting
material.
The new
disease, called yellow curling, is a serious threat to sweet potato crops in
Argentina. We are conducting studies to demonstrate its detrimental effects on
the production and quality of roots.
Conclusion
In this work,
the first antiserum for the fast, safe and efficient diagnosis of SPVG in the
country was obtained.
The four
viruses involved in YC disease were isolated and identified: SPFMV, SPVG, SPCSV
and SPLCV.
The interaction of SPFMV, SPVG,
SPCSV and SPLCV is needed for the development of yellow curling symptoms in
sweet potato in Argentina.
Acknowledgements
Daniela Martinelli contributed to
SPVG biological characterization. Consejo
Nacional de Investigaciones Científicas y Técnicas (CONICET) and Instituto
Nacional de Tecnología Agropecuaria (INTA) supported this research.
1. Adikini, S.; Mukasa, S. B.; Mwanga, R. O.; Gibson, R. W. 2016. Effects
of sweet potato feathery mottle virus and sweet potato chlorotic stunt virus on
the yield of sweetpotato in Uganda. Journal of
Phytopathology. 164: 242-254. https://doi.org/10.1111/jph.12451
2. Aregbesola, O. Z.; Legg, J. P.; Sigsgaard, L.; Lund, O. S.;
Rapisarda, C. 2019. Potential impact of climate change on
whiteflies and implications for the spread of vectored viruses. Journal of Pest Science. 92: 381-392.
https://doi.org/10.1007/s10340-018-1059-9
3. Bejerman, N. 2019. Geminivirus-Vector
Relationship. In: Kumar, R. (Ed). Geminiviruses: Impact, Challenges and
Approaches. Springer. Cham.
137-145. https://doi.org/10.1007/978-3- 030-18248-9_8
4. Bejerman, N.; Zanini, A.; Rodríguez Pardina, P.; Di Feo, L.
2016. Use of 454-Pyrosequencing for the characterization of
sweet potato virus C and sweet potato feathery mottle virus isolates from
Argentina and development of a multiplex one-step RT-PCR for their simultaneous
detection. Journal of Phytopathology. 164:
386-394. https://doi.org/10.1111/jph.12466
5. Clark, M. F.; Adams, A. N. 1977. Characteristics of the
microplate method of enzyme-linked immunosorbent assay for the detection of
plant viruses. Journal of General Virology. 34: 475-483.
https://doi.org/10.1099/0022-1317-34-3-475
6. Clark, C. A.; Davis, J. A.; Abad, J. A.; Cuellar, W. J.;
Fuentes, S.; Kreuze, J. F.; Gibson, R. W.; Mukasa, S. B.; Tugume, A. K.; Tairo,
F. D.; Valkonen, J. T. P. 2012. Sweetpotato viruses: 15 years of progress on
understanding and managing complex diseases. Plant Disease.
96: 168-185. https:// doi.org/10.1094/PDIS-07-11-0550
7. Di Feo, L.; Nome, S. F.; Biderbost, E.; Fuentes, S.; Salazar,
L. F. 2000. Etiology of sweet potato chlorotic dwarf disease
in Argentina. Plant Disease .
84: 35-39. https://doi.org/10.1094/ PDIS.2000.84.1.35
8. FAOSTAT. 2022. Food and Agriculture Data. https://www.fao.org/faostat/es/#data/QCL. Accessed February 11 2022
9. Gibson, R. W.; Mpembe, I.; Alicai, T.; Carey, E. E.; Mwanga, R.
O. M.; Seal, S. E.; Vetten, H. J. 2002. Symptoms, aetiology
and serological analysis of sweet potato virus disease in Uganda. Plant Pathology. 47: 95-102.
https://doi.org/10.1046/j.1365-3059.1998.00196.x
10. Jones, R. A. C. 2016. Chapter Three - Future Scenarios for
plant virus pathogens as climate change progresses. In: Kielian, M.;
Maramorosch, K.; Mettenleiter, T. C. (Eds.). Advances in
Virus Research. Academic Press. p. 87-147.
https://doi.org/10.1016/bs.aivir.2016.02.004
11. Karyeija, R. F.; Kreuze, J. F.; Gibson, R. W.; Valkonen, J. P.
2000. Synergistic interactions of a potyvirus and a
phloem-limited closterovirus in sweet potato plants. Virology.
269: 26-36. https:// doi.org/10.1006/viro.1999.0169
12. Karyeija, R. F.; Gibson, R. W.; Valkonen, J. P. 2008. Resistance to sweet potato virus disease (SPVD) in wild East
African Ipomoea. Annals of Applied Biology.
133: 39-44. https://doi. org/10.1111/j.1744-7348.1998.tb05800.x
13. Kennedy, G. G.; Moyer, J. W. 1982. Aphid transmission and
separation of two strains of Sweet potato feathery mottle virus from sweet
potato. Journal of Economic Entomology. 75: 130-133.
https://doi.org/10.1093/jee/75.1.130
14. Kokkinos, C. D.; Clark, C. A. 2006. Real-Time PCR Assays for
detection and quantification of sweetpotato viruses. Plant Disease . 90: 783-788.
https://doi.org/10.1094/PD-90-0783
15. Kwak, H. R.; Kim, M. K.; Shin, J. C.; Lee, Y. J.; Seo, J. K.;
Lee, H. U.; Jung, M. N.; Kim, S. H.; Choi, H. S. 2014. The current incidence of
viral disease in Korean sweet potatoes and development of multiplex RT-PCR
assays for simultaneous detection of eight sweet potato viruses. The Plant Pathology Journal. 30: 416-424.
10.5423/PPJ.OA.04.2014.0029
16. Lan, P.; Li, F.; Abad, J.; Pu, L.; Li, R. 2018. Simultaneous
detection and differentiation of three Potyviridae viruses and sweet potato by
a multiplex TaqMan real time RT-PCR assay. Journal of
Virological Methods. 252: 24-31. https://doi.org/10.1016/j.jviromet.2017.09.006
17. Lewthwaite, S. L.; Fletcher, P. J.; Fletcher, J. D.; Triggs,
C. M. 2011. Cultivar decline in sweetpotato (Ipomoea
batatas). New Zealand Plant Protection. 64:
160-167. https://doi.org/10.30843/ nzpp.2011.64.5976
18. Li, R.; Salih, S.; Hurtt, S. 2004. Detection
of Geminiviruses in sweetpotato by Polymerase Chain Reaction. Plant Disease . 88: 1347-1351.
https://doi.org/10.1094/PDIS.2004.88.12.1347
19. Li, F.; Xu, D.; Abad, J.; Li, R. 2012. Phylogenetic
relationships of closely related potyviruses infection sweet potato determined
by genomic characterization of Sweet potato virus G and Sweet potato virus 2. Virus Genes. 45: 118-125.
https://doi.org/10.1007/s11262-012-0749-2
20. Liu, Q.; Wang, Y.; Zhang, Z.; Lv, H.; Qiao, Q.; Qin, Y.;
Zhang, D.; Tian, Y.; Wang, S.; Li, J. 2017. Diversity of
sweepoviruses infecting sweet potato in China. Plant Disease . 101: 2098-2103. https://
doi.org/10.1094/PDIS-04-17-0524-RE
21. Lizarraga, C.; Fernandez-Northcote, E. N. 1989. Detection of
potato viruses X and Y in sap extracts by a modified indirect enzyme-linked
immunosorbent assay on nitrocellulose membranes (NCM-ELISA). Plant Disease . 73: 11-14.
22. Loebenstein, G. 2015. Control of sweet potato virus diseases. In: Loebestein, G.;
Katis, N. I. (Eds.). Advances in virus research. Control of plant virus diseases. Academic
Press. p. 33-45. https://doi.org/10.1016/bs.aivir.2014.10
23. López Colomba, E.; Rodríguez Pardina, P.; Luque, A.; Nome, C.;
Di Feo, L. 2011. Estimación de daños potenciales causados por
virus involucrados en el encrespamiento amarillo de la batata. Horticultura Argentina. 30: 55.
24. Martino, J. M.; Fontele, R. S.; Ferreira, F. A.; Ribeiro, S.
G.; Di Feo, L. D. V. 2017. First report of sweet potato leaf
curl Georgia virus in sweet potato in Argentina. Plant
Pathology. 101: 513.
https://doi.org/10.1094/PDIS-08-16-1215-PDN
25. Martino, J. A. 2019. Etiología e importancia
de las virosis en cultivos de batata (Ipomoea batatas (L.) Lam) de
regiones productoras de Argentina. Rol de los insectos vectores y efecto final
sobre la calidad de raíces. Ph. D. Tesis. Facultad de Ciencias Exactas Físicas
y Naturales. Universidad Nacional de Córdoba. Argentina.
26. Milne, R. G.; Lesemann, D. E. 1978. An
immunoelectron microscopic investigation of oat sterile dwarf and related
viruses. Virology. 90: 299-304.
https://doi.org/10.1016/0042- 6822(78)90314-8
27. Moyer, J. W.; Cali, B. B.; Kennedy, G. G.; Abou-Ghadir, M. F.
1980. Identification of two Sweet potato feathery mottle
virus strains in North Carolina. Plant Disease . 64: 762-764.
28. Mulabisana, M. J.; Cloete, M.; Laurie, S. M.; Mhela, W.;
Maserumule, M. M.; Nhlapo, T. F.; Cochrane, N. M.; Oelofse, D.; Rey, M. E. C.
2019. Yield evaluation of multiple and co-infection of geminiviruses and potyviruses
on sweet potato varieties under field conditions and confirmation of multiple infection by NGS. Crop Protection.
119: 102-112. https://doi. org/10.1016/j.
cropro.2019.01.009
29. Nhalpo, T. F.; Rees, D. J. G.; Odeny, D. A.; Mulabisana, J.
M.; Rey, M. E. C. 2018. Viral metagenomics reveals sweet potato virus diversity
in the Eastern and Western Cape provinces of South Africa. South
Africa Journal of Botany. 117: 256-267. https://doi.org/10.1016/j.
sajb.2018.05.024
30. Nome, S. F. 1973. Sweet potato vein mosaic
virus in Argentina. Journal of Phytopathology . 77: 44- 54.
https://doi.org/10.1111/j.1439-0434.1973.tb04110.x
31. Nome, S. F.; Giorda, L. M.; Vázquez, A. 1980. El virus de moteado plumoso de la batata (Sweet potato feathery mottle
virus) en Argentina. Revista de Investigaciones Agropecuarias. 15: 625-634.
32. Parent, J. G.; Bélanger, F.; Desjardins, S.; Brisson, J. D.
1985. Dot-immunobinding for detection of Tomato mosaic virus and Potato virus X
infecting greenhouse tomatoes. Phytoprotection. 66:
53-57.
33. Rodríguez Pardina, P.; Bejerman, N.; Luque, A. V.; Di Feo, L.
2012a. Complete nucleotide sequence of an Argentinean isolate of Sweet potato
virus G. Virus Genes. 45: 593-595. https://doi. org/10.1007/s11262-012-0784-z
34. Rodríguez Pardina, P.; Luque, A.; Nome, C.; López Colomba, E.;
Fuentes Delgado, S.; Di Feo, L. 2012b. First report of Sweet
potato leaf curl virus infecting sweet potato in Argentina. Australasian Plant Disease Notes.
7: 157-160. https://doi.org/10.1007/s13314-012-0073-7
35. Souto, E. R.; Sim, J.; Chen, J.; Valverde, R. A.; Clark, C. A.
2003. Properties of strains of Sweet potato feathery mottle virus and two newly
recognized Potyviruses infecting sweet potato in the United States. Plant Disease . 87: 1226-1232.
https://doi.org/10.1094/ PDIS.2003.87.10.1226
36. Untiveros, M.; Fuentes, S.; Salazar, L. F. 2007. Synergistic interaction of Sweet potato chlorotic stunt virus
(Closterovirus) with carla-, cucumo-, ipomo-, and potyviruses infection sweet
potato. Plant Disease .
91: 669-676. https://doi.org/10.1094/PDIS-91-6-0669
37. Urcuqui-Inchima, S.; Haenni, A. L.; Bernardi, F. 2001.
Potyvirus proteins: a wealth of functions. Virus Research.
74: 157-175. 10.1016/S0168-1702(01)00220-9
38. Valverde, R. A.; Sim, J.; Lotrakul, P. 2004. Whitefly transmission of sweet potato viruses. Virus Research. 100: 123-128.
https://doi.org/10.1016/j.virusres.2003.12.020
39. Valverde, R. A.; Clark, C. A.; Valkonen, J. P. 2007. Viruses and virus disease complexes of sweetpotato. Plant Viruses. 1: 116-126.
40. Wang, Q.; Zhang, L.; Wang, B.; Yin, Z.; Feng, C.; Wang, Q.
2010. Sweetpotato viruses in China. Crop
Protection. 29: 110-114. https://doi.org/10.1016/j.cropro.2009.11.002
41. Wanjala, B. W.; Ateka, E. M.; Miano, D. W.; Low, J. W.;
Kreuze, J. F. 2019. Storage root yield of sweetpotato as influenced by
Sweetpotato leaf curl virus and its interaction with Sweetpotato feathery
mottle virus and Sweetpotato chlorotic stunt virus in Kenya. Plant Disease . 104: 1477-1486. https://doi.org/10.1094/PDIS-06-19-1196-RE .
Both authors contributed equally to
this study.