Revista de la Facultad de Ciencias
Agrarias. Universidad Nacional de Cuyo. Tomo 54(2). ISSN (en línea) 1853-8665.
Año 2022.
Original article
Effect of bioslurries
on tomato Solanum lycopersicum L and lettuce Lactuca sativa development
Efecto de bioles en el desarrollo de tomate Solanum
lycopersicum L y lechuga Lactuca sativa
Ernesto Martín Uliarte 3
Andrea Hidalgo 2
1 Consejo Nacional de Investigaciones Científicas y
Técnicas (CONICET). Av. Ruiz Leal s/n - Parque Gral. San Martín (M5500XAD).
Mendoza. Argentina.
2 Universidad Nacional de Cuyo. Facultad de Ciencias
Agrarias. Almirante Brown 500 (M5528AHB). Chacras de Coria.
Mendoza. Argentina.
3
Agrotecnología Sostenible. Estación Experimental Mendoza. Instituto Nacional de
Tecnología Agropecuaria (INTA EEA Mendoza). San Martin 3853 (M5507EVY).
Chacras de Coria. Mendoza. Argentina.
Abstract
Bioslurries,
obtained by anaerobic digestion of fresh organic matter, are emerging as a
cheaper and low-impact alternative for synthetic products in agriculture. The
aim of this study was to evaluate bioslurry obtained from biogas digestion
(Biog), bioslurry for plant nutrition by FAO (Bfao), and lactic fermentation
(Blac) as biostimulant in tomato and lettuce plants. Based on a toxicity test,
a 10% dilution was finally applied to the plants. In lettuce, Bfao and Blac
significantly increased aerial biomass (2.17 ±0.54 and 2.33 ±1.13 g
respectively), regarding water control (1.16 ±0.60 g), while root biomass was
only increased by Bfao (1.60 ±0.44 g) compared to control (0.66 ±0.34 g). All
digestates increased chlorophyll content index (CCI), while yield (Fv/Fm) and
performance index (Plabs) did not show differences with water control. In
tomato, only aerial biomass was significantly increased by Bfao. All digestates
significantly increased CCI, while Fv/Fm was only significantly higher in Bfao
and Blac, related to water control. PIabs showed no differences. In both plant
species, commercial fertilizer showed significantly higher values for all
parameters. In conclusion, all digestates stimulated plant growth, Bfao showed
the highest effect on tomatoes and lettuce biomass followed by Blac and Biog,
being a cheaper, safer and lower-impact alternative for traditional products
for crop growing.
Keywords: Anaerobic digestion; Agroecology; Organic
production; Plant nutrition; Lycopersicum esculentum.
Resumen
Los bioles son bioinsumos obtenidos de la digestión anaeróbica de materia
orgánica, que surgen como una alternativa económica y de bajo impacto para la
agricultura. El objetivo del presente trabajo fue evaluar los bioles obtenidos
de: la producción de biogás (Biog), elaborado para la nutrición vegetal con
base en la FAO (Bfao), y de una fermentación láctica (Blac), para utilizarse
como bioestimulantes en lechuga y tomate. Sobre la base del análisis de
toxicidad, la dilución al 10% fue seleccionada para aplicarse en las plantas.
En lechuga, Bfao y Blac incrementaron significativamente la biomasa aérea (2,17
±0,54 y 2,33 ±1,13 g respectivamente), referente al control (1,16 ±0,60 g),
mientras que la biomasa radical presentó mayores valores solo en Bfao (1,60
±0,44 g), comparado al control (0,66 ±0,34 g). Los bioles incrementaron el
contenido de clorofila (CCI), mientras que los índices de rendimiento (Fv/Fm) y
de desempeño (Plabs) no se diferenciaron del control. En tomate, solo la
biomasa aérea fue significativamente mayor en Bfao, todos los bioinsumos
incrementaron significativamente CCI, mientras que Fv/Fm solo por Bfao y Blac,
y PIabs no mostró diferencias respecto al control. En ambos, lechuga y tomate
el fertilizante comercial fue el tratamiento con mayores valores, sin embargo,
todos los bioinsumos estimularon el crecimiento vegetal. El mayor efecto fue
producido por Bfao, seguido de Blac y Biog, siendo una alternativa adecuada
para una producción vegetal económica, segura y con menor impacto.
Palabras
clave: Digestión anaeróbica; Agroecología; Producción orgánica; Nutrición vegetal; Lycopersicum esculentum.
Originales: Recepción: 23/06/2022
Aceptación:
12/09/2022
Introduction
The Cuyo region
in Argentina is an arid zone with heterogeneous and poorly structured soils
with low fertility and organic matter. Thus, performing management tasks favoring higher biological activity and nutrient availability
turns necessary for sustainable crop growing (1). Moreover, due to the limited information
about organic amendments in these arid soils, several studies suggest an
increase in crop productivity and improvement in soil properties by bio-input
applications (20).
Worldwide,
tomato (Solanum lycopersicum L) and lettuce (Lactuca sativa) are
two important horticultural crops, being tomato the second most consumed in
Argentina. These crops are mainly produced in oases located in arid zones,
being Cuyo region the highest cultivated surface (35), with Mendoza province as the main tomato
producer with 3,757 ha, and 265 ha for lettuce (16). However, the intensive farming of tomatoes
and lettuce requires a large amount of high-cost synthetic agrochemicals to
cover nutrient requirements and control diseases and plagues, thus having
negative environmental impacts (5, 9).
Therefore,
organic products are becoming a cheaper and low-impact alternative to synthetic
ones. In this sense, biopesticides and biofertilizers developed locally
represent a viable alternative for sustainable crop management, lowering
environmental negative effects and production costs. Bioproducts are
biologically active supplies that promote plant growth by different strategies (10), such as increasing nutrient availability,
producing phytohormones and antibiotics, and/or competing against pathogens (26, 27). Furthermore, natural compounds may quickly
biodegrade, due to microbiologic coevolution and decomposition metabolic
pathways (7).
Bioslurry or
digestate, obtained by anaerobic digestions of fresh organic matter, presents
adequate characteristics for plant growth. Most of the available bibliography
refers to bioslurry as the byproduct or residue of biogas production with
biofertilizer and biocontroller properties against plant diseases (8, 17). The liquid fraction derived from anaerobic
digestions can retain nutrients and microorganisms. It has also been tested as
biofertilizer, soil amendment, and even for bioremediation, with promising
results (29, 30, 36).
By-products of
anaerobic digestions have been also developed as plant biostimulants, with no
biogas generation. The Food and Agriculture Organization (FAO), proposed a
protocol to obtain a bioslurry specifically designed for plant nutrition and
growth promotion,
based on elemental and microbial content in the final product (12). Other authors have reported lactic
fermentation for obtaining adequate biostimulants for plant growth, showing
promising results (22). Nevertheless,
there is a lack of information about the properties of these non-methanogenic
bioslurries in plant nutrition and antipathogen effectiveness.
The study
evaluated three different bioslurries obtained from different raw materials and
elaboration processes, as biofertilizers for tomato and lettuce. The hypothesis
stated that anaerobic digestions generate by-products rich in nutrients and
microorganisms that increase plant development.
Methods
Digestates
elaboration
Three different
digestates or bioslurries were tested as plant biostimulants: bioslurry
obtained from biogas digestion (Biog); bioslurry designed for plant nutrition
by FAO (Bfao); and a lactic fermentation lixiviate (Blac). Biog was obtained
from biogas digestion of a traditional mixture of water: goat manure in a 9:1
ratio. The biodigester was built with two 200 μm polyethylene layers in a 1.25
m3
tubular design intended to be semi-buried in cold climates, for
integrated solar gain systems and insulated enclosure. During Biog production,
the biodigester was operated at 25 °C with a hydraulic retention time of 60
days and fed twice a week with a 7 kg load of goat manure and 60 kg of water.
Bfao was brewed according to FAO protocol (2013): An
anaerobic process was carried out in a 200 L PET recipient, with a screw cap
and a gas trap containing 10 kg of fresh vegetal material, 60 L of fresh goat
manure, 3 kg of wood ashes, 4 kg of bentonite, 500 g of chicken eggshells, 3 kg
of bone ashes, 5 L of cow milk, and free chloride water to a final volume of
170 L. After 3 months of storage, the product was filtered and stored in
darkness at 15-20 °C. Finally, Blac elaboration protocol was based on Quirós et al. (2004): a mixture of 1 L of
commercial rice and 2 L of chlorine-free water was left to settle for 48 h.
Later, the lixiviate obtained was supplemented with 6
L of milk and stored at 30 °C for 3 days. Finally, it was filtered and stored
until use.
Bioslurries
characterization
Toxicity test
Lactuca sativa var Grandrapids
seeds were used for toxicology tests according to US EPA
(1996). Seeds were previously tested for germination power and seed
viability using sterile distilled water. Then, seeds were superficially
sterilized with 70% ethanol and exposed to the biostimulants as follows: 20
seeds were placed on a filter paper in 90 mm Petri dishes, exposed to 0, 10,
25, 50, and 100% dilutions of each product by triplicate, and maintained at 20
°C in darkness for 5 days. Control treatment and dilutions were performed with
well water (H2O),
also used for product brewing. To evaluate the toxic effect, total germination,
and hypocotyl/root elongation, were analyzed.
Physico-chemical
analyses
To perform a
basic characterization, pH, CE, and macronutrients were analyzed in each
bioproduct by duplicate. Total nitrogen (N) was determined in dry samples by
Kjeldahl and steam trawl distillation method (4). Phosphorus (P) was colorimetrically
analyzed by HCl extraction with ammonium methavanadate, ammonium molybdate, and
nitric acid solution. Absorbance at 420 nm was measured with a UV-VIS Milton
Roy spectrophotometer. Finally, K was determined by flame atomic absorption
spectrophotometry (32).
Pot
assays
To evaluate
bioslurries effect on plant growth, seeds of Lactuca sativa var Grand
rapids and Solanum lycopersicum var Platense were germinated and grown
in seedling trays for 15 days, with basal fertilization of 500 mg of commercial
fertilizer (KSC® 2 NPK 23-5-5, Timac Agro USA). Seedlings were transplanted
into a 0.5 L pot containing perlite: peat (1:1) and grown under greenhouse
conditions (23±2 °C, 30% humidity, and natural 16/8 h photoperiod due summer
season). All plants were irrigated with well water every 48 h to maintain field
capacity during the assay. After transplant, homogeneous 10 cm plants were
selected for treatment initiation. A complete randomized block design was
established with 5 treatments for each plant species (n=8): Biog, bioslurry
from biogas production; Bfao, bioslurry designed for plant nutrition by FAO
(2013); Blac, lixiviate of a lactic fermentation; Cont, well water; and Fert,
chemical fertilization. Throughout the assay, 50 mL of each product diluted at
10% were weekly applied.
Aerial and root
dry weight (DWa, DWr, respectively) were determined in 120 days old plants.
Additionally, yield (Fv/Fm) as an indicator of photosystem II damage, and
performance index (PIabs) as stress resistance capacity, were measured with a
Chlorophyll Fluorimeter Handy Pocket PEA (Hansatech Instruments Ltd., King’s
Lynn, Norfolk, England). This was carried out with a leaf-clip placed on the
third leaf from the apex for 20 min till dark adaptation (13).
Finally,
chlorophyll content index (CCI) by absorbance was also determined in the third
leaf with a chlorophyll meter (model Clorofilio, Cavadevices, Argentina).
Statistical
analysis
ANOVA was
performed considering the block design, and a LSD Fisher means comparison test
(p<0.05) evaluated the effect of the digestates on seedling biomass.
Shapiro-Wilk normality tests and residues regression were carried out to
confirm ANOVA assumptions. Due to the lack of normality, phytotoxicity test was
analyzed by non-parametric Kruskall Wallis test (p<0.05). InfoStat software
version 2015 performed all statistical analyses (InfoStat Group, FCA, National University of Córdoba, Argentina). All data was
expressed as mean ± standard deviation.
Results
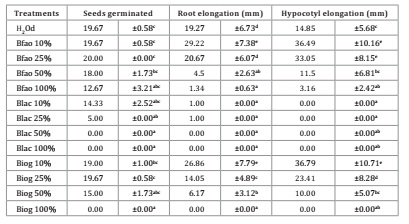
Phytotoxicity
test
Blac treatment
decreased seed germination at concentrations of 100, 50, and 25%, while 10% did
not differentiate from control even though the value was lower. Biog and Bfao
did not show significant differences from control in any dilutions, except for
Biog 100%, with zero germinated seeds.
Root and
hypocotyl elongation was negatively affected by Blac in all concentrations,
whereas Bfao and Biog 10% significantly increased these parameters. At 25%,
both Bfao and Biog significantly increased plant hypocotyl while not affecting
root elongation. At higher concentrations, all products showed toxicity,
reducing plant elongation (Table 1).
Table 1: Phytotoxic
effect of bioslurries on Lactuca sativa var Grand Rapids seeds.
Tabla 1: Efecto de fitotoxicidad de los bioles en semillas de Lactuca
sativa var Grand Rapids.
Kruskal Wallis p= 0.05. Values are expressed as mean ±SD.
Se realizó un análisis de Kruskal Wallis con
una significancia del 0,05. Valores
expresados como media ±DS.
Biog: bioslurry from biogas production; Bfao: bioslurry designed
for plant nutrition by FAO
(2013); Blac: lixiviate of a lactic fermentation; and H2Od:
distilled water used as control.
Biog: biol proveniente de la producción de biogás; Bfao:
biol diseñado para la nutrición vegetal, en base a FAO (2013); Blac: lixiviado de una fermentación láctica; y H2Od: agua destilada usada como
control.
Except for
Blac, no treatment differed from control at 10%, suggesting no phytotoxicity
properties or plant growth stimulation. Such concentration was considered for
further analyses based on these results and the bibliography. Blac 10% was
included in further assays for results verification, considering that in
vitro conclusions may be limited and different results may be expected in
pot treatments.
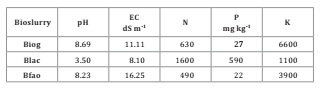
Bioslurries
characterization
Only Bfao
presented an adequate pH value (Resolución 19/2019, Secretaría de Gobierno de
Ambiente y Desarrollo Sustentable, Argentina; Ministry of Environment and
sustainable development of Argentina). All products presented high EC levels,
while Blac also presented high acidity (low pH value). Considering the
phytotoxicity results, all products must be diluted, being 10% the most
adequate dilution (Table 2).
Table 2: Bioslurries physico-chemical characterization. Each
product was analyzed by duplicate.
Tabla 2: Caracterización fisicoquímica de los bioles.
Cada producto fue analizado por duplicado.
Biog: bioslurry from biogas production; Bfao: bioslurry designed
for plant nutrition by FAO
(2013); and Blac: lixiviate of a lactic fermentation.
Biog: biol proveniente de la producción de biogás; Bfao:
biol diseñado para la nutrición vegetal, en base a FAO (2013); Blac: lixiviado de una fermentación láctica.
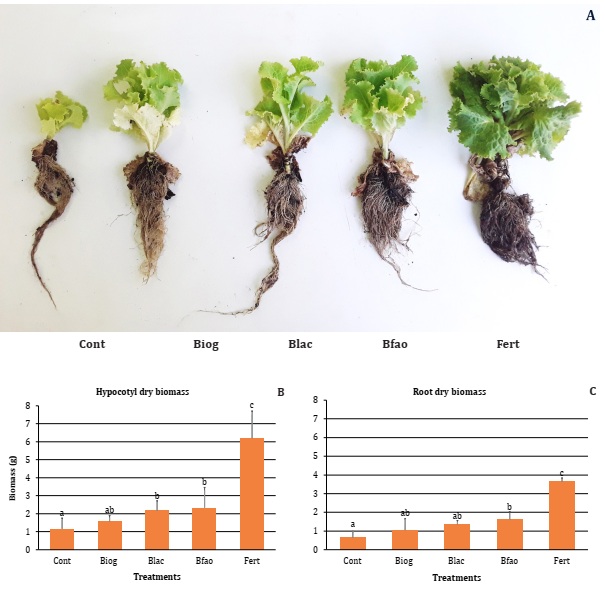
Effect
of bioslurries on lettuce growth
There
were no significant differences among biolsurries treatments. Nevertheless,
Bfao and Blac showed significantly higher aerial biomass (2.17 ±0.54 and 2.33
±1.13 g respectively), related to control (1.16 ±0.60 g; Figure 1B).
Cont: control,
irrigated with water; Biog: bioslurry from biogas production; Bfao: bioslurry
designed for plant nutrition by FAO (2013);
Blac: lixiviate of a lactic
fermentation; and Fert: inorganic fertilizer (KSC® 2 NPK 23-5-5, Timac Agro
USA).
Cont: control, regado con agua; Biog: biol proveniente de la producción
de biogás; Bfao: biol diseñado para la nutrición vegetal, en base a FAO
(2013); Blac: lixiviado de
una fermentación láctica; y Fert: suplementado con fertilizante inorgánico
(KSC® 2 NPK 23-5-5, Timac Agro USA).
Values are
expressed as mean ±SD. ANOVA (LSD Fischer, p<0.05).
Valores expresados como media ±DS. ANOVA (LSD Fischer, p<0,05)
Figure 1: Effect of
bioslurries on Lactuca sativa total (A), aerial
(B) and root biomass (C).
Figura 1: Efecto de los bioles en la biomasa total (A),
aérea (B) y radical (Ca) de Lactuca sativa.
Root
biomass was only increased by Bfao (1.60 ±0.44 g) (0.66 ±0.34 g; Figure 1C). As expected, Fert was the
treatment with significantly higher values of plant biomass (3.64 ±1.21 and 6.2
±1.50 g, respectively for root and aerial dry weight, Figure
1A).
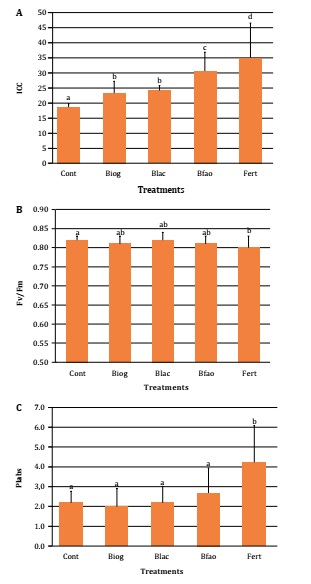
CCI in lettuce
plants was significantly increased by all digestates with respect to control
(18.61 ±3.35), with the maximum value reached by Fert treatment (35.06 ±2.00),
and followed by Bfao (30.59 ±3.70). Biog (23.33 ±1.27) and Blac (24.17 ±2.12)
showed lower values and did not differentiate from each other (Figure 2A).
A: CCI: chlorophyll content index; B: Fv/Fm: yield index,
indicators of photo system II damage; and C: PIabs: performance index,
indicator stress resistance capacity. Cont: control, irrigated with water;
Biog: bioslurry from biogas production; Bfao: bioslurry designed for plant
nutrition by FAO (2013); Blac: lixiviate of a lactic fermentation; and Fert:
inorganic fertilizer (KSC® 2 NPK 23-5-5, Timac Agro USA).
A: CCI: índice de contenido de clorofila; B:
Fv/Fm: índice de rendimiento, indicador de daño del fotosistema II; y C: PIabs:
índice de desempeño, indicador de la capacidad de resistir estrés. Cont:
control, regado con agua; Biog: biol proveniente de la producción de biogás;
Bfao: biol diseñado para la nutrición vegetal, en base a FAO (2013); Blac:
lixiviado de una fermentación láctica; y Fert: suplementado con fertilizante
inorgánico (KSC® 2 NPK 23-5-5, Timac Agro USA).
Values are expressed as mean ±SD. ANOVA (LSD Fischer, p<0.05).
Valores expresados como media ±DS. ANOVA (LSD Fischer, p<0,05).
Figure 2: Effect of bioslurries on nutritional and stress indicators in Lactuca
sativa.
Figura 2: Efecto de los bioinsumos en indicadores
nutricionales y de estrés en Lactuca sativa.
As stress
indicator, damage in photosystem II was significantly higher in Fert (0.80
±0.03), regarding Cont (0.82 ±0.01), reaching the lowest Fv/Fm value, while
Fert and Cont digestates did not differentiate (Figure 2B). Oppositely, PIabs were significantly increased by Fert
(4.23 ±1.85), and digestates did not differentiate from Cont (2.19 ±0.58), with
Bfao showing the highest value (2.68 ±1.26, Figure 2C).
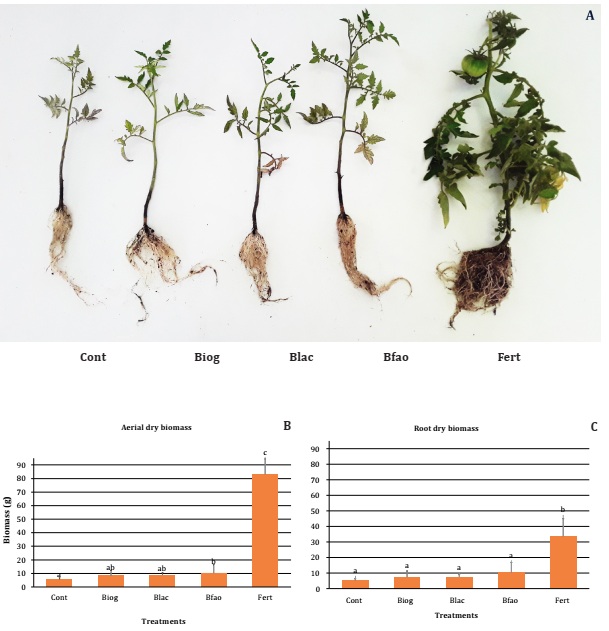
Effect
of bioslurries on tomato growth
Bioslurries
did not significantly increase root dry biomass in tomato plants, and were significantly
lower than Fert treatment. As in lettuce, Fert was the treatment with the
highest root and aerial biomass (33.53 ±11.45 and 83.09 ±5.39 g for) while
control showed the lowest biomass values (Figure
3).
Cont:
control, irrigated with water; Biog: bioslurry from biogas production; Bfao:
bioslurry designed for plant nutrition by FAO
(2013);
Blac: lixiviate of a lactic fermentation; and Fert: inorganic fertilizer (KSC®
2 NPK 23-5-5, Timac Agro USA).
Cont: control, regado con agua; Biog: biol proveniente
de la producción de biogás; Bfao: biol diseñado para la nutrición vegetal, en
base a FAO
(2013); Blac: lixiviado de una fermentación láctica; Fert: suplementado con
fertilizante inorgánico (KSC® 2 NPK 23-5-5, Timac Agro USA).
Values are expressed as mean ±SD. ANOVA (LSD
Fischer, p<0.05).
Valores expresados como
media ±DS. ANOVA (LSD Fischer, p<0,05).
Figure 3: Effect of
bioslurries on Solanum lycopersicum total (A), aerial (B) and root
biomass (C).
Figura 3: Efecto de los bioles en la biomasa total (A)
aérea (B) y radical (C) de Solanum lycopersicum.
Aerial
biomass was significantly increased only by Bfao (10.21 ±3.05 g), in relation
to Cont (5.47 ±1.38 g), while Blac and Biog did not show significant effects.
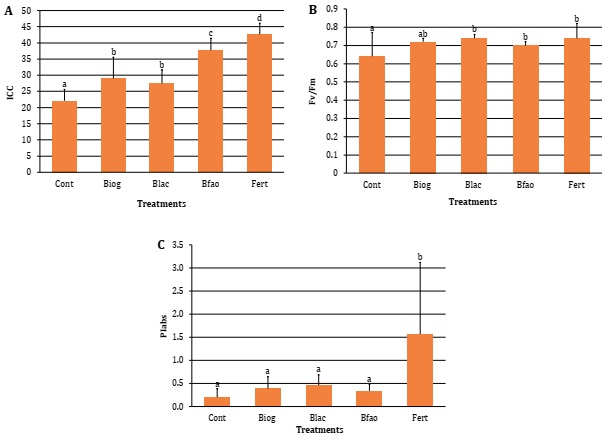
All digestates
significantly increased CCI in tomato plants compared to control (22.1 ±3.56).
The maximum value was reached by Fert treatment (42.66 ±3.44), followed by Bfao
(37.69 ±3.71). As in lettuce plants, Biog (29.04 ±6.55) and Blac (27.5 ±4.16) did
not differentiate from each other and presented lower values (Figure 4A).
A:
CCI: chlorophyll content index; B: Fv/Fm: yield index, indicators of
photosystem II damage; and C: PIabs: performance index, indicator
stress resistance capacity. Cont: control,
irrigated with water; Biog: bioslurry from biogas production; Bfao: bioslurry
designed for plant
nutrition by FAO (2013); Blac: lixiviate of a lactic
fermentation; and Fert: inorganic fertilizer (KSC® 2 NPK 23-5-5, Timac Agro
USA).
A: CCI: índice de contenido de clorofila; B: Fv/Fm:
índice de rendimiento, indicador de daño del fotosistema II; y C: PIabs: índice
de
desempeño, indicador de la capacidad
de resistir estrés. Cont: control, regado con agua; Biog: biol proveniente de
la producción de biogás;
Bfao: biol diseñado para la nutrición
vegetal, en base a FAO (2013); Blac: lixiviado de una fermentación láctica; y Fert:
suplementado con
fertilizante inorgánico (KSC® 2 NPK
23-5-5, Timac Agro USA).
Values are expressed as mean ±SD. ANOVA (LSD
Fischer, p<0.05).
Valores expresados como media ±DS.
ANOVA (LSD Fischer, p<0,05).
Figure 4: Effect of bioslurries on nutritional and stress indicators in Solanum
lycopersicum.
Figura 4: Efecto de los bioinsumos en indicadores
nutricionales y de estrés en Solanum lycopersicum.
Yield
index Fv/Fm was significantly higher in Bfao (0.70 ±0.02), Blac (0.74 ±0.02)
and Fert (0.74 ±0.08), with respect to Cont (0.64 ±0.13), and with no
differences among them (Figure 4B). PIabs was significantly increased only by Fert (1.57
±1.55). Nevertheless, no differences were detected among digestates and Cont
treatments (Figure 4C).
Discussion
In the present
study, we demonstrated the ability of different bioslurries or digestates
obtained from organic waste as biofertilizers, resulting in high-quality inputs
for agricultural production. Generally, digestates are the result of anaerobic
digestion of organic residues for energetic generation, suggesting the
potentiality of anaerobic processes to reduce negative environmental impacts.
However,
digestates may have high EC, exceeding the 3 dS m-1,
limit established for irrigation water, and potentially toxic for agriculture
production (24). It may also
contain high ammonia concentrations causing decreased oxygen concentration in
the root system (6). According to
our results, the retention time of each elaboration process seems independent
to element solubilization. Bfao, with a retention time of 90 days, did not show
higher NPK values than a 60 day Biog, while Blac showed the highest N and P
content, with 5 days of brewing time.
In line with
other studies, for plant growth, dilutions are commonly needed to reduce
phytotoxic effects of pure products. In agriculture, there is no agreement
among authors on the optimal dilution of digestates for maximum stimulation and
minimum toxicity. Song et al. (2021) reported
20% as digestate optimal concentration for biostimulant usage in several
horticultural crops. Nonetheless, Díaz Montoya (2017)
suggested negative effects on lettuce germination at concentrations above 2-4%;
and Silva et al. (2011) reported possible
phytotoxicity at concentrations higher than 10%. Our results indicated
dilutions lower than 25% for Bfao and Biog, and 10% for Blac. This suggests a
high influence of the raw material used for bio-inputs brewing, determining the
quality and variety of the nutrient and metabolites, more than the EC itself.
Despite the higher EC of Bfao and Biog, lettuce plants showed low toxicity at a
higher digestate concentration (25%), while in Blac, a
10% dilution was necessary, avoiding negative effects.
Tomato and
lettuce biomass increased with the bioproducts, mainly by Bfao, due to its
specific design for plant growth. Biog seemed to stimulate growth but not
significantly from control, while Blac significantly increased lettuce biomass.
Regardless of the high N and P content of Blac, its elaboration process is focused
on lactic bacteria content and their effect on plant growth. These
microorganisms serve as biofertilizers, biocontrollers, biostimulants, and
bioelicitors (19, 33), probably
explaining the increase in lettuce biomass. On the other hand, Biog process is
the only one not trying to increase plant nutrition and beneficial
microorganisms, but producing energetic compounds. However, several studies
have reported this product´s biostimulant quality (3, 15). Accordingly, our results displayed an
increase in plant biomass, showing no toxic effects at dilutions below 25%.
Inhibition of
plant growth in early stages has been reported by the application of
digestates, suggesting dilutions below 5% in these periods (Medina
et al., 2015), probably explaining the differences in plant biomass
as regards Fert treatments. Consequently, the use of these products as
stimulants in the initial stages of plant growth and development may be
counterproductive, thus higher dilutions are needed. Another important factor
to be considered is the substrate used. Previous studies demonstrated that
compost combined with digestate is the best treatment for plant growth, even at
a similar level or above commercial substrate and chemical fertilizers (14). All this suggests complex interactions
among biostimulants, substrates, and plants, being important for phenologic
stage, concentration, and frequency of product application.
Chlorophyll
content is highly correlated to N content in leaves and may be used as a
nutritional indicator (23). All digestates significantly increased this
parameter, suggesting the nutritional beneficial effect, mainly N intake in
lettuce and tomato plants. Although bioproduct composition presents macro and
micronutrients, they also contribute with microorganisms that promote plant
growth (18) by mechanisms
like hormone production, nutrient solubilization, and N2 fixation. This may explain the
increase in plant biomass, despite the lower nutrient content with respect to
inorganic fertilizer, indicating that in a bioproduct, microbial content may be
more important than nutrient concentration.
Stress
indicators Fv/Fm and PIabs, suggested no bioslurries negative effect on plant
growth. In lettuce, those plants treated with inorganic fertilizer presented
the lowest values, suggesting higher photosystem II damage with respect to
digestates and water control. According to the manufacturer (Hansatech
Instruments Ltd.), Fv/Fm near 0.85 indicates healthy tissues. Therefore, tomato
showed greater damage than lettuce since the values were lower, being control
the most affected and suggesting nutritional limitations. PIabs represents
plant capacity to respond to stress, being more sensitive than Fv/Fm for stress
determinations (2). Despite the
high variability between lettuce and tomato, PIabs was significantly increased
by Fert, possibly given to higher nutrient intake. Despite the lack of
significance, all bioslurries increased these parameters, suggesting an
improvement in plant coping ability under biotic and abiotic stress.
Our results
demonstrated that digestates can be used as biostimulants for plant growth,
with different properties depending on the source and brewing method. Dilutions
are needed, due to the toxicity of the pure product, especially for seedling
production, which may require even lower concentrations. Further studies are
needed to determine dilutions, appropriate moment and frequency of application,
and the possibility of combining the different digestates for optimal plant
growth, allowing for reduced synthetic products, with lower negative impacts
and safer production strategies.
Conclusion
Digestates are
valuable by-products, rich in nutrients and microorganisms for high-quality
plant production. Each product presented different characteristics and effects
on plant biomass, suggesting complex interactions, thus consequent possible
complementation in their use. All digestates stimulated plant growth. Bfao
showed the highest effect on tomatoes and lettuce biomass followed by Blac and
Biog, constituting an adequate alternative for a cheaper, safer and low-impact
strategy for crop growth. The biostimulants presented high nutrient content and
no phytotoxic effects at concentrations of 10%, being an excellent strategy to
treat organic residues while high-quality by-products are obtained. Further
studies are needed to determine optimal brewing conditions, dilutions, raw
materials and application techniques for producers. Moreover, Liquid
biofertilizers should be used and evaluated, not only as an isolated practice
but also within a set of sustainable crop management strategies.
Funding
Research was
funded by the Universidad Nacional de Cuyo (SIIP A092, UNCuyo 2019-2021 to
Andrea Hidalgo and Iván Funes-Pinter) and Fondo para la Investigación
Científica y Tecnológica (FONCYT, PICT 2019-2193 to Iván Funes-Pinter).
Acknowledgment
The
authors are thankful to INTA EEA Mendoza for making use of the facilities to
perform the experiments, and to Natalia Giancaglini and Inés Hugalde for
language revision.
1. Abril, A.; Villagra, P.; Noe, L. 2009. Spatiotemporal
heterogeneity of soil fertility in the Central Monte desert (Argentina).
Journal of Arid Environments. 73: 901-906. https://doi.org/10.1016/j. jaridenv.2009.04.019
2. Banks, J. M. 2018. Chlorophyll fluorescence
as a tool to identify drought stress in acer GENOTYPES. Environmental and Experimental Botany. 155: 118-127. https://doi.org/10.1016/j. envexpbot.2018.06.022
3. Baştabak, B.; Koçar, G. 2020. A review of the
biogas digestate in agricultural framework. J Mater
Cycles Waste Manag. 22: 1318-1327.
https://doi.org/10.1007/s10163-020-01056-9
4. Bremner, J. M.; Mulvaney, C. S. 1982. Methods of soil analysis,
Part 2: Chemical and Microbiological properties. In: Norman, A. G. (Ed.),
Agron. Monogr. 9.2. ASA. SSSA.
p. 1149-1178. https:// doi.org/10.2134/agronmonogr9.2.c32
5. Campos, E. V. R.; Proença, P.; Oliveira, J.; Bakshi, M.;
Abhilash, P. C.; Fraceto, L. 2018. Use of botanical insecticides for
sustainable agriculture: Future perspectives. Ecological
Indicators. IN PRESS.
https://doi.org/10.1016/j.ecolind.2018.04.038
6. Cheong, J. C.; Lee, J. T. E.; Lim, J. W.; Song, S.; Tan, J. K.
N.; Chiam, Z. Y.; Yap, K. Y.; Lim, E. Y.; Zhang, J.; Tan, H. T. W.; Tong, Y. W.
2020. Closing the food waste loop: Food waste anaerobic digestate as fertilizer
for the cultivation of the leafy vegetable, xiao bai cai
(Brassica rapa). Sci. Total Environ. 715: 136789
7. Couillerot, O.; Loqman, S.; Toribio, A.; Hubert, J.; Gandner,
L.; Nuzillard, J.; Renault, J. 2014. Purification of
antibiotics from the biocontrol agent Streptomyces anulatus S37 by centrifugal
partition chromatography. J Chromatogr B. 944: 30-34. https://doi.org/10.1016/j.
jchromb.2013.11.008
8. Dahlin, J.; Herbes, C.; Nelles, M. 2015. Biogas digestate marketing:
Qualitative insights into the supply side. Resour Conserv Recy. 104: 152-161. https://doi.org/10.1016/j. resconrec.2015.08.013
9. Damalas, C. A.; Eleftherohorinos, I. G. 2011. Pesticide
exposure, safety issues, and risk assessment indicators. Int J Environ Res
Public Health. 8(5): 1402-19. https://doi. org/10.3390/ijerph8051402
10. de Andrade Filho, F. C.; Queiroga de Oliveira, E.; Silva de
Lima, J. S.; Moreira, J. N.; Silva, I. N.; Anizio Lins, H.; Cecílio Filho, A.
B.; Paes Barros Júnior, A.; Bezerra Neto, F. 2020. Agro-economic viability from
two croppings of broadleaf vegetables intercropped with beet fertilized with
roostertree in different population densities. oot-shoot
growth and time to transplant of different lettuce (Lactuca sativa L.),
genotypes during nursery. Revista de la Facultad de Ciencias Agrarias.
Universidad Nacional de Cuyo. Mendoza. Argentina. 52(1): 210-224.
11. Díaz Montoya, A. J. 2017. Características
fisicoquímicas y microbiológicas del proceso de elaboración de biol y su efecto
en germinación de semillas (Tesis de maestría). Universidad
Nacional Agraria La Molina. Lima, Perú. 129 p.
12. FAO. 2013. Los biopreparados para la producción de
hortalizas en la agricultura urbana y periurbana. Supervisión técnica de
Alberto Pantoja, Ph.D Oficial de Producción y Protección Vegetal. http://www.fao.org/3/a-i3360s.pdf
13. Funes Pinter, I.; Salomon, M. V.; Berli, F.; Gil, R.; Bottini,
R.; Piccoli, P. 2018. Plant growth promoting rhizobacteria alleviate stress by
AsIII in grapevine. Agriculture, Ecosystems and Environment.
267: 100-108. https://doi.org/10.1016/j.agee.2018.08.015
14. Funes-Pinter, I.; Pisi, G.; Aroca, M.; Uliarte, M. 2020. Plant
Bioestimulants: Compost Tea and Bioslurry Characterization. PREPRINT (Version
1) available at Research Square. https:// doi.org/10.21203/rs.3.rs-56311/v1
15. Guilayn, F.; Rouez, M.; Crest, M.; Patureau, D.; Jimenez, J.
2020. Valorization of digestates from urban or centralized biogas plants: a
critical review. Rev Environ Sci Biotechnol. 19: 419- 462.
https://doi.org/10.1007/s11157-020-09531-3
16. IDR (Instituto de Desarrollo Rural). 2022.
Estimación de la superficie cultivada con hortalizas de verano en Mendoza. Temporada
2021-2022. https://www.idr.org.ar/wpcontent/uploads/2022/04/estimacion_hortalizas_verano_2021_2022.pdf (Accessed Abril 2022)
17. Koszel, M.; Lorencowicz, E. 2015. Agricultural Use of Biogas
Digestate as a Replacement Fertilizers. Agriculture and Agricultural Science Procedia. 7: 119-124. https://doi.org/10.1016/j. aaspro.2015.12.004
18. Kumar, A.; Verma, L. M.; Sharma, S.; Neetu, S. 2022. Overview
on agricultural potentials of biogas slurry (BGS): applications, challenges,
and solutions. Biomass Conv. Bioref. Jan. 4: 1-41.
https://doi.org/10.1007/s13399-021-02215-0
19. Lamont, J. R.; Wilkins, O.; Bywater-Ekegärd, M.; Smith, D. L.
2017. From yogurt to yield: Potential applications of lactic acid bacteria in
plant production. Soil Biology and Biochemistry. 111:
1-9. https://doi.org/10.1016/j.soilbio.2017.03.015
20. Martínez, L. E.; Vallone, R. C.; Piccoli, P. N.; Ratto, S. E.
2018. Assessment of soil properties, plant yield and
composition, after different type and applications mode of organic amendment in
a vineyard of Mendoza, Argentina. Revista de la Facultad
de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina. 50(1):
17-32.
21. Medina, A.; Quipuzco, L.; Juscamaita, J. 2015. Evaluación de la calidad de biol de segunda generación de estiércol de ovino
producido a través de biodigestores. Anales Científicos. 76(1): 116-124.
https://doi.org/10.21704/ac.v76i1.772
22. Mohd Zaini, N. S.; Idris, H.; Yaacob, J. S.; Wan-Mohtar, W. A.
A. Q. I.; Putra Samsudin, N. I.; Abdul Sukor, A. S.; Lim, E. J.; Abd Rahim, M.
H. 2022. The Potential of Fermented Food from Southeast Asia
as Biofertiliser. Horticulturae. 8: 102. https://doi.org/10.3390/ horticulturae8020102
23. Muchecheti, F.; Madakadze, C.; Soundy Puf. 2016.
Leafchlorophyll readings as an indicator of nitrogen status and yield of
spinach (Spinacia oleracea L.) grown in soils amended with Luecaena
leucocephala prunings. Journal of Plant Nutrition.
39(4): 539-561. https://dx.doi. org/10.1080/01904167.2016.1143488
24. Olías, M.; Cerón, J. C.; Fernández, I. 2005. About the use of the U.S. Salinity Laboratory (USLS) classification
of irrigation water. Geogaceta. 37: 111-113.
25. Quirós, P. A.; Albertin, B. A.; Blázquez, S. M. 2004. Elabore sus propios abonos, insecticidas y repelentes orgánicos.
Organización para Estudios Tropicales. 36 p.
26. Salomon, M. V.; Funes Pinter, I.; Piccoli, P.; Bottini, R.
2017. Use of plant growth-promoting Rhizobacteria as Biocontrol Agents: Induced
systemic resistance against biotic stress in plants. In: Kalia, V. C. (Ed.). Microbial Applications. Vol. 2: Biomedicine, Agriculture and
Industry. Springer International Publishing. Cham. p. 133-152. https://doi.
org/10.1007/978-3-319-52669-0_7
27. Saritha, M.; Prasad Tollamadugu, N. V. K. V. 2019. Chapter 15. The status of research and application of
biofertilizers and biopesticides: Global Scenario. Editor(s): Viswanath
Buddolla. Recent Developments in Applied Microbiology and
Biochemistry. Academic Press. 195-207. https://doi.org/10.1016/B978-0-12-816328-3.00015-5
28. Silva, M.; Azevedo, M.; Silva, F.; Fernandes, J.; Monteiro, A.
2011. Análise da crescimento não destrutiva das plantas de
feijão branco adubadas com biofertilizante supermagro. Resumos
do VII Congresso Brasileiro de Agroecologia. Fortaleza. Brasil. 6(2): 5.
29. Smith, J.; Assefa Abegaz, A.; Matthews, R. B.; Subedi, M.;
Orskov, E. R.; Tumwesige, V.; Smith, P. 2014. What is the potential for biogas
digesters to improve soil fertility and crop production in Sub-Saharan Africa? Biomass and Bioenergy. 70: 58-72. http://dx.doi.org/10.1016/j.
biombioe.2014.02.030
30. Smith, E.; Thavamani, P.; Ramadass, K.; Naidu, R.; Srivastava,
P.; Megharaj, M. 2015. Remediation trials for hydrocarbon-contaminated soils in
arid environments: Evaluation of bioslurry and biopiling techniques. International Biodeterioration & Biodegradation. 101:
56-65. https://doi.org/10.1016/j.ibiod.2015.03.029
31. Song, S.; Lim, J. W.; Lee, J. T. E.; Cheong, J. C.; Hoy, S.
H.; Hu, Q.; Tan, J. K. N.; Chiam, Z.; Arora, S.; Lum, T. Q. H.; Lim, E. Y.;
Wang, C.; Tan, H. T. W.; Tong, Y. W. 2021. Food-waste anaerobic digestate as a
fertilizer: The agronomic properties of untreated digestate and
biochar-filtered digestate residue. Waste Management.
143-152. https://doi.org/10.1016/j.wasman.2021.10.011
32. TMECC (Test Methods for the Examination of Composting and
Compost). 2002. US Composting Council.
33. Trakselyte-Rupsiene, K.; Juodeikiene, G.; Alzbergaite, G.;
Zadeike, D.; Bartkiene, E.; Ozogul, F.; Rueller, L.; Robert, J.; MRocha, J. M.
F. 2022. Bio-refinery of plant drinks press cake permeate
using ultrafiltration and lactobacillus fermentation into antimicrobials and
its effect on the growth of wheatgrass in vivo. Food
Bioscience. 46: 101427. https://doi.org/10.1016/j.
fbio.2021.101427
34. US EPA (US Environmental Protection Agency). 1996. Ecological
effects test guidelines. Seed germination/root elongation toxicity
test. OPPTS 850.4200
35. Viteri, M. L.; Ghezán, G.; Iglesias, D. 2013. Tomate y lechuga: producción, comercialización y consumo. Estudio
socioeconómico de los sistemas agroalimentarios y agroindustriales. 165 p.
36. Warnars, L.; Oppenoorth, H. 2014. Bioslurry: A supreme
fertilizer. Ed: Kelly Atkinson. Netherlands: Deltahage.