Revista de la Facultad de Ciencias
Agrarias. Universidad Nacional de Cuyo. Tomo 55(1). ISSN (en línea) 1853-8665.
Año 2023.
Original article
Seed treatments with salicylic acid and Azospirillum
brasilense enhance growth and yield of maize plants (Zea mays L.)
under field conditions
Tratamientos de semillas con ácido salicílico y Azospirillum
brasilense aumentan el crecimiento y el rendimiento de las plantas de maíz
(Zea mays L.) en condiciones de campo
Luis Alfredo Rodríguez Larramendi 1*
Miguel Ángel Salas-Marina 1
Vidal Hernández García 1
Rady Alejandra Campos Saldaña 1
Wel Cruz Macías 1
Raúl López Sánchez 2
1 Universidad de Ciencias y Artes de Chiapas. Facultad de
Ingeniería Campus Villa Corzo. Carretera a Monterrey Km 3.0. Villa Corzo CP
30520. Chiapas. México.
2
Universidad de Granma. Carretera a Peralejo Km 18. Bayamo 85100. Granma. Cuba.
*
alfredo.rodriguez@unicach.mx
Abstract
Salicylic acid and Azospirillum brasilense stimulate plant growth
and productivity. In some environments, plant physiology similarly responds to
both bioactive products. Considering this, a field experiment was conducted to
study the physiological effect of Salicilic acid and A. brasilense on
growth and grain yield of maize plants. The experiment involved three
treatments consisting of imbibed seeds in an aqueous solution of SA (0.01 mM),
inoculated seeds with A. brasilense and a control treatment. Seed
imbibition in SA and inoculation with A. brasilense improved vegetative
growth in the early stages of crop ontogeny, increasing leaf growth, plant
height, stem diameter and biomass accumulation. Spikelet length and weight were
greater in plants first inoculated with A. brasilense and then treated
with SA. Results indicated that SA stimulated biomass partitioning towards
leaves, root and stem, while A. brasilense mainly affected leaf growth,
plant height, ear dimensions and grain yield. Such results turn crucial for
biological fertilization strategies aimed at reducing pollutant loads that
accompany chemical fertilizers. Both products can be part of maize management
practices given competitive economic advantages and sustainability.
Keywords:
Bioactive products;
Biofertilizers;
Plant hormones.
Resumen
El ácido salicílico y Azospirillum
brasilense estimulan el crecimiento y la productividad de las plantas y, en
algunos entornos, las respuestas fisiológicas de las plantas a la aplicación
de ambos productos bioactivos parecen ser similares. Con el objetivo de
estudiar su efecto fisiológico en el crecimiento y la producción de granos de
plantas de maíz se diseñó un experimento de campo. Los tratamientos
consistieron en embeber las semillas en una solución acuosa de SA (0,01 mM) e
inocularlas con A. brasilense y un tratamiento control. La imbibición de
las semillas en SA y la inoculación con A. brasilense mejoraron el
crecimiento vegetativo en las primeras etapas de la ontogenia del cultivo,
aumentando el crecimiento foliar, la altura de la planta, el diámetro del tallo
y la acumulación de biomasa. La longitud y el peso de las espigas fueron
mayores en las plantas inoculadas con A. brasilense y tratadas con SA,
en ese orden. Los resultados indicaron que el SA estimuló la acumulación de
biomasa hacia las hojas, la raíz y el tallo, mientras que A. brasilense tuvo
más influencia en el crecimiento de las hojas, la altura de la planta, las
dimensiones de la mazorca y el rendimiento del grano. Tales resultados son
relevantes para el diseño de estrategias de fertilización biológica con la
respectiva reducción de la carga contaminante que acompaña a los fertilizantes
químicos. Ambos productos pueden formar parte de las prácticas agronómicas del
cultivo de maíz por sus ventajas económicas competitivas y por ser ambientalmente
sostenibles.
Palabras clave: Productos bioactivos; Biofertilizantes; Hormonas vegetales.
Originales: Recepción: 24/08/2022
Aceptación: 27/02/2023
Introduction
Maize constitutes an essential nutrient for the human diet. Despite a
low protein content compared to other vegetables, it provides, on average, 39%
of overall protein intake, and 59% of human energy requirements (38).
Mexico is the largest market for maize, consuming 11% of world
production (29). However, the low-yielding domestic
production does not meet the growing demand, and importation reaches
approximately 11.3 and 0.145 million tons of yellow and white maize,
respectively (36). Hence, there is an evident need to
explore technological alternatives for environmentally friendly production and
increasing yields.
The indiscriminate use of ammonia fertilizers on some bedrock types of soil, causes serious acidification problems. Soils in the
Frailesca region of Chiapas are undergoing an acidification process (11) and consequent increasing toxic aluminium contents in maize plants,
particularly at pH levels below 5.0 (6). Faced with such a
situation, finding alternatives entailing reduced agrochemical applications may
contribute to reduced soil degradation. In this sense, several biofertilizers
based on nitrogen-fixing microorganisms have shown positive effects on certain
cultivated plants, such as maize (3,
25, 32), sorghum (19), soybeans (10), and rice (12).
Among nitrogen-fixing bacteria, A. brasilense is considered one
of the most important plant growth-promoting bacteria (8). On one hand, this bacterium promotes growth and increases plant
production, after promoting phytohormone synthesis (5) and biological nitrogen fixation. On the other hand, A. brasilense restricts
certain plant pathogens through antibiosis and siderophores (14). In addition, Azospirillum may associate with more than 100
plant species, 14 of which are grasses (30). As stated by Gavilanes et
al. (2020), A. brasilense (Ab-V5 and
Ab-V6) has been widely used as a commercial inoculant in Brazil (14), with positive effects on grain dry matter and nitrogen accumulation in
plants, especially in grain crops such as maize and wheat (13, 24).
Given the mentioned phytohormone-producing capacity of
this bacterium, inoculation effects could be related to the role played by
salicylic acid. Several studies performed with rhizosphere bacteria show that
most strains produced metabolites of the AIA type, siderophores and salicylic
acid (22). Salicylic acid (SA) is a phenolic secondary
metabolite (20) present in plant tissues (33, 41),
regulating plant growth and increasing crop yield when supplied in low
exogenous concentrations (34). In maize, the application of SA increases grain
production per plant, total dry biomass, and N, P, K contents (41).
Some hypotheses about the effect of SA and A. brasilense consider
the role of SA as a plant hormone and the ability of A. brasilense to
fix atmospheric nitrogen and increase nutrient uptake by promoting increased
stem growth and grain production (15). The effect of both bioactive products
(SA and A. brasilense), effective biomolecules and plant growth
regulators (26), depended on the genotype and
environmental conditions. In this sense, given the agronomic implications on
possible biological fertilization strategies in maize, comparing the effect of
both products turns interesting. Thus, this research aimed to study the
physiological effect of A. brasilense and SA on growth and grain
production of maize plants under field conditions.
Materials and methods
Location
The research was conducted from June 2016 to January 2017 in the
locality of Calzada Larga, municipality of Villaflores, Chiapas, México, at
16°21’08.5” N and 93°18’58.2” W and 713 m a. s. l. (8). The predominant climates are warm and semi-warm, with an average
temperature of 24.5 ℃, an average rainfall of 1200 mm per year, and the following predominant
soil types: lithosols, luvisols, cambisols and vertisols.
Growth conditions and plant material
Seeds of the CLTHW11002 hybrid, yellow maize from the International
Maize and Wheat Improvement Center (CIMMYT), were manually sowed at 0.8 m
between rows and 0.35 m between plants on a luvisol soil with loamy texture and
enough water content.
According to the Mexican Official Standard
(27), the
soil is strongly acidic, free of carbonates and salts, low in potassium,
moderately low in organic matter, medium phosphorus (P-Bray) and nitrogen (N-NO3), moderately high in
magnesium (Mg) and very high in iron (Fe). Both aluminium content and
proportion vs. total soil cations (4.61%) were low, despite the extreme
soil acidity (Table 1).
Table 1: Soil chemical properties.
Tabla 1: Caracterización química del suelo del área experimental.
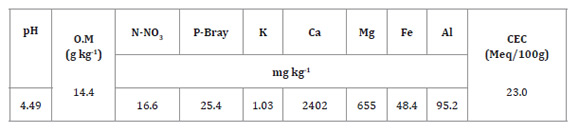
O. M: organic matter, CEC: cation exchange
capacity.
M. O:
Materia Orgánica, CEC: Capacidad de intercambio catiónico.
Two nitrogen fertilizations were applied at 15 days and 30 days after
sowing, both at a rate of 75 kg ha-1 of N, for a total dose of 150
kg ha-1
(46-00-00). Weed chemical control was carried out with Velquat
1.5 L ha-1
at 15 days after sowing, and 1.5 L ha-1 Tacsaquat at 60 days after
sowing. The fall armyworm (Spodoptera frugiperda) was controlled with
Cipermetrine 21.12% EC at a dose of 1.0 L ha-1.
Experimental design and treatments
The randomized block experimental design with three
treatments and three replicates consisted of T1: salicylic acid (SA), T2: A.
brasilense (5 x 108 CFU), T3: control treatment without SA and not
inoculated. Each experimental unit measured 25 m2 (5 x 5 m), leaving one border
plant on each side of the plot.
Seed inoculation with A. brasilense
The commercial product tested was Azofer®, composed of A. brasilense (50%),
peat (37.5%) and calcium carbonate (12.5%). Calcium carbonate allows achieving
the ideal pH (6.8-7.0) for the inoculant (6), while not affecting plant growth or
development. The concentration of A. brasilense was 5 x 108 CFU’s per
gram of commercial product.
For seed inoculation, an inert solution was prepared with 60 g of
carbosil-methyl-cellulose adhesive powder dissolved in 1.5 L of distilled water
and left to stand for two hours. Subsequently, mointened seeds with the
adherent powder were homogeneously covered with the biofertilizer and left to
dry in the shade, at room temperature, before sowing.
Imbibition of seeds with salicylic acid
A solution with distilled water and salicylic acid at a concentration of
0.01 mM, was prepared according to Hayat and Ahmad (2007), Gordillo-Curiel et al.
(2020) and Rodríguez-Larramendi et al. (2017). The seeds were soaked for 2 hours and left to dry at room temperature
before sowing.
Analytical evaluations
For growth measurements at 30 and 60 days after sowing, five plants per
replicate were selected, number of leaves per plant (LP) was counted, and plant
leaf area was determined in cm2 (LA) with a CI-202 (Bioscience
®) portable leaf area meter. Plant height (PH) was measured with a millimetre
ruler. Leaves, stem, roots of the five selected plants
were separated, and oven dried at 80°C for 72 hours. Finally, leaves dry weight
(LDW), shoot dry weight (SDW) and root dry weight (RDW) were obtained using a
Sartorius® analytical balance. Plant dry weight was calculated.
Growth rates
Based on leaf area and dry weight, the leaf area index (LAI) was calculated
by dividing plant leaf area (LA) by soil coverage. Root weight fraction (RWF, g
g-1) was calculated
as the root dry weight vs. plant dry weight ratio. This indicator
expresses the “root’s investment” or biomass gain (4). Leaf weight fraction (LWF, g g-1)
was calculated as leaf dry weight vs. plant dry weight, estimating leaf
partitioning (42). The root mass/leaf mass ratio
(dimensionless) estimated phenotypic plasticity in biomass allocation (4). Specific leaf area (SLA, cm2 g-1)
was calculated as leaf area/leaf dry weight, reflecting functional traits of
leaf morphology such as thickness and density (31) and vegetative vigor (23). Leaf area ratio (LAR, cm2 g-1),
one important determinant of plant relative growth rate (42), was calculated as leaf area by plant dry weight.
Yield components
When the crop reached physiological maturity, five plants per treatment
were selected in each replicate and ear length (EL, cm), ear diameter (ED, cm),
100-grain dry weight, and dry grain yield per plant (Yield, g plant-1) at 14% grain
moisture, were assessed.
Statistical analysis
ANOVA with a randomized block design followed by LSD mean comparison
test was conducted considering p ≤ 0.05. ANOVA assumptions were verified by the
Cochran and Barttlet tests. A multivariate Principal Component Analysis (PCA)
searched for a possible relationship between plant growth, yield, ear weight and ear size. All analyses were performed with
the statistical package STATISTICA® release 8.0 (39).
Results
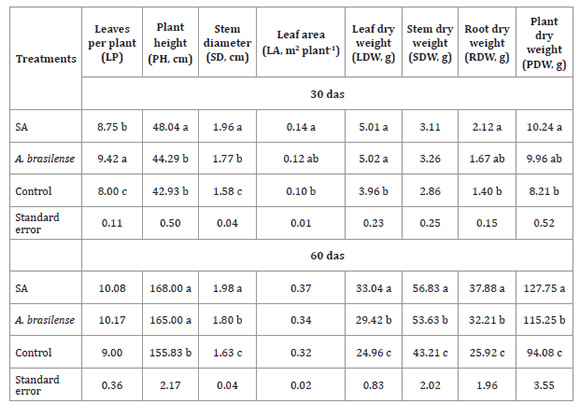
The number of leaves per plant was significantly
higher in inoculated plants, compared to those treated with SA and control, the
latter of which produced fewer leaves. Despite a tendency towards more leaves
per plant on both sampling dates (at 30 and 60 das), at 60 das, this difference
was not significant (Table 2).
Table 2: Effect of salicylic acid (SA) and A. brasilense on
growth of maize plants at 30 and 60 days after sowing (das).
Tabla 2: Efecto del ácido salicílico (AS) y A. brasilense en el
crecimiento de plantas de maíz a los 30 y 60 días después de la siembra (dds).
ns: Not significant; * Statistically
significant for p ≤ 0.05, das: days after sowing.
Ns: sin
diferencias significativas, * Diferencias estadísticas significativas para p ≤
0,05, dds: días después de la siembra.
However, plant height and stem diameter were significantly higher in
plants grown from SA-treated seed, and even higher than those inoculated with
A. brasilense at 30 das. At 60 das, no significant difference in plant height
was observed between SA and A. brasilense-treated plants (Table
2).
Leaf area was larger in SA-treated plants at 30 das than control plants.
The latter exhibited no significant differences concerning those inoculated
with A. brasilense. At 60 das, no difference in leaf area was observed among
treatments (Table 2).
Leaf dry weight was statistically higher in plants treated with SA and
A. brasilense than in control plants at 30 das. At 60 das this difference was
even higher for SA-treated plants followed by A. brasilense, compared to
the control. Similar results were observed for root and stem dry weight and,
therefore, for total plant dry weight. However, stem dry weight was
statistically different at 60 das (Table 2).
Table 3 shows the effect of bioactive
products on growth ratios, only significant at 30 das, when an increasing leaf
area index was observed for the SA treatment.
Table 3: Effect of the application of salicylic acid (SA) and A. brasilense on
growth rates of maize plants at 30 and 60 days after sowing (das).
Tabla 3: Efecto de la aplicación de ácido salicílico (AS) y A.
brasilense en los índices de crecimiento de plantas de maíz a los 30 y 60
días despues de la siembra (dds).
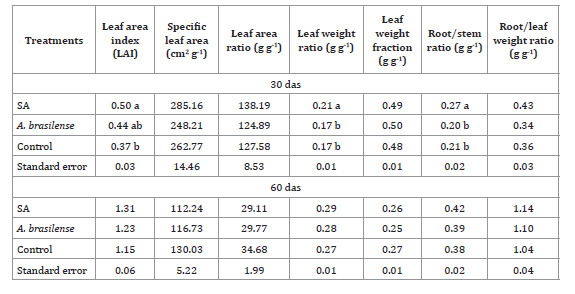
Ns: Not significant; * Statistically
significant for p ≤ 0.05.
Ns: sin
diferencias significativas, * Diferencias estadísticas significativas para p ≤
0,05 dds: días después de la siembra.
No difference was observed between A. brasilense and control. At 60 das,
such growth-stimulating effects were not significant (Table
3).
The higher leaf growth values observed with SA and A.
brasilense at early stages of maize plant development (Table 2) was similar to the
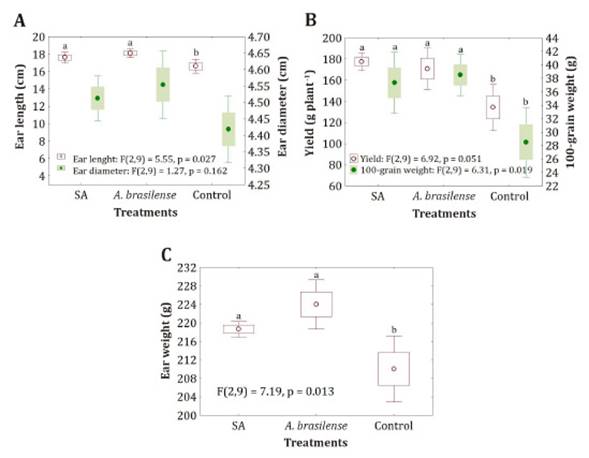
increase detected in ear length and weight, and yield per plant (Figure 1).
Box and vertical lines indicate the
standard error and standard deviation, respectively.
Los
cuadros y las líneas verticales indican el error etándar y la desviación
estándar de la media respectivamente.
Figure 1: Effect of salicylic acid and A.
brasilense on A) ear length and diameter, B) yield and 100-grain weight and
C) ear weight.
Figura 1:
Efecto del tratamento con ácido
salicílico y A. brasilense en: A) longitud y diámetro de la mazorca, B)
rendimiento y peso de 100 granos y C) peso de la mazorca.
Plants treated with SA and A. brasilense developed longer ears,
consistently with the higher 100-grain weight (Figure
1 B), ear weight and yield per plant (Figure 1 B, C).
Considering vegetative growth, ear size and grain
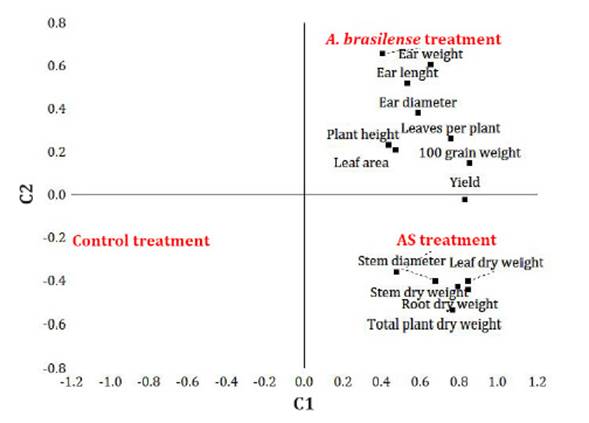
production, a PCA allowed establishing the effects of both bioactive products
as a function of the evaluated variables (Figure 2).
Figure 2: PCA
biplot of growth variables, ear dimensions and grain yield of corn plants
treated with SA and A. brasilense, according to components C1 (48.) % and C2 (16.20%).
Figura 2: Biplot de las variables de crecimiento, dimensiones de la mazorca
y producción de granos de plantas de maíz tratadas con AS y A. brasilense,
en el plano formado por las componetes C1 (48.) % y C2 (16.20%).
Component 1
(48.58%) discriminates control plants from both bioactive treatments, and
component 2 (16.20%) discriminates both bioactive treatments. Variables related
to biomass accumulation and stem diameter, discriminate in favour of SA
treatment, while variables related to leaf growth, plant height, ear dimensions
and grain yield, separate the A. brasilense treatment (Figure 2).
Discussion
Maize growth response to seed inoculation with A. brasilense and
imbibition with SA on two sampling dates suggests a possible relationship with
plant physiological stage and ontogeny, something to be further addressed in
future research. The effect of both bioactive products resulted organ
dependent.
The results further demonstrate that SA and A. brasilense have
similar effects, promoting vegetative growth of maize plants at the early
stages. However, these effects depend on sampling date (34) and particularly, on plant organ. This effect is probably given by SA
and A. brasilense sharing an analogous property, stimulating hormone
synthesis and interacting with cytokinins. SA constitutes a plant hormone
exceeding plant immunity and abiotic stress (34). In coordination with cytokinins, ethylene, auxins, gibberellins,
jasmonic acid and abscisic acid, SA significantly contributes to growth and
development regulation, although through unknown mechanisms (34). In this sense, it has been shown that the growth-promoting effects of
SA could be related to hormone modulation (1,
37)
or gas exchange improvement (39).
The PCA showed correlations between leaf growth vs. plant height,
and ear size vs. increased grain production. These aspects are related
to the A. brasilense enhanced nitrogen absorption, even in fertile soils
(15), and enhanced hormone synthesis,
including auxin, gibberellin, and cytokinin (2, 17).
The effect of SA on grain production in maize plants is also supported
by other studies. Low doses of SA induce increased ear length and maize grain
yield (41). However, in our research, SA was applied
to leaves (41) and not seeds, opening a new field of
research.
Positive plant responses to inoculation with A. brasilense might
be given by biological nitrogen fixation, probably overshadowed by the nitrogen
fertilization carried out in the experiment, and by plant hormonal production.
In this regard, inoculations with A. brasilense had improved root growth
and development of Setaria viridis grass after increased CO2
fixation and reduced accumulation of photo-assimilated carbon in
leaves, resulting in greater canopy growth, increased water content in plant
tissues, and reduced stress (24,
28).
In addition, increased production of indoleacetic acid may improve nutrient
uptake by augmenting root growth (24,
28). Zeffa et al. (2019)
studied A. brasilense inoculated seeds, finding intensified plant
growth, improved biochemical traits and raised NUE under nitrogen deficit.
Other authors have found that regardless of nitrogen source and dosage, A.
brasilense increased maize grain yield (16).
SA effects on plant dry weight are probably related to SA ability to
increase N, P2O5
and K2O contents in
plant tissues (41). On the other hand, seed inoculation with
A. brasilense stimulates root growth, increases root exploration
capacity and promotes biological nitrogen fixation, which may be related to
higher grain production and ear length in inoculated plants. Specifically, in
maize, other studies have shown that inoculations with nitrogen-fixing bacteria
result in a 9% increase in maize grain production (16).
Our results show that SA and A. brasilense had similar
stimulating effects on growth and yield of maize plants, from early stages of
plant ontogeny. This physiological effect may be related to the ability to
interact with other hormones (37) or promote hormone synthesis (16). However, we suggest caution before issuing a hypothesis on this matter
as, apart from the above-mentioned properties, A. brasilense contributes
to plant nutrition through biological nitrogen fixation, a fact that may mask
the effects of both bioactive products on plant growth and development.
Conclusions
We consider that the results obtained show robust evidence supporting
theoretical and methodological bases, as well as sufficient evidence on the
physiological effect of S.A and the bacterium A. brasilense on growth
and grain production of maize under field conditions. Such results are relevant
to the design of biological fertilization strategies with the respective
reduction of pollutants. Considering economic and environmental sustainability,
both products can be considered within the agronomic practices of maize
cultivation.
The results indicated that SA and A. brasilense exhibited
similar stimulating effects on growth and yield of maize plants, from early
stages. SA induced greater plant biomass accumulation, with a tendency to
maximize root dry mass, while A. brasilense stimulated greater leaf
growth and plant height, consequently inducing increased ear growth and grain
production.
1. Abreu, M. E.; Munné-Bosch, S. 2009.
Salicylic acid deficiency in NahG transgenic lines and sid2 mutants increases
seed yield in the annual plant Arabidopsis thaliana, Journal of
Experimental Botany. 60: 1261-1271.
2.
Bashan, Y.; de-Bashan, L. E. 2010. How the plant
growth-promoting bacterium Azospirillum promotes plant growth - a
critical assessment. Adv. Agron. 108: 77-136. doi:
10.1016/S0065-2113(10)08002-8
3. Bertasello, L. E. T.; Filla, V. A.;
Prates Coelho, A.; Vitti Môro, G. 2021. Agronomic performance
of maize (Zea mays) genotypes under Azospirillum brasilense application
and mineral fertilization. Revista de la Facultad de Ciencias Agrarias. Universidad Nacional
de Cuyo. Mendoza. Argentina. 53(1): 68-78.
4. Camargo, I.; Rodríguez, N. 2006. New perspectives
to study the biomass allocation and its relationship with the functioning of
plants in neotropical ecosystems. Acta biológica colombiana.
11: 75-87.
5.
Carcaño-Montiel, M. G.; Ferrera-Cerrato, R.; Pérez-Moreno, J.; Molina-Galán,
D.; Bashan, Y. 2016. Actividad nitrogenasa, producción de fitohormonas,
sideróforos y antibiosis en cepas de Azospirillum y Klebsiella aisladas
de maíz y teocintle. Terra Latinoamericana. 24: 493-502.
6.
Casierra, F.; Aguilar, O. Estrés por aluminio en las plantas: reacciones en el
suelo, síntomas en vegetales y posibilidades de corrección. 2007. Revista
Colombiana de Ciencias Hortícolas. 1: 246-256.
7.
Cassán, F.; Penna, C.; Creus, C.; Radovancich, D.; Monteleleone, E.; Salamone,
I. G.; Lett, L. 2010. Protocolo para el control de calidad de inoculantes que
contienen Azospirillum sp. Buenos Aires, Argentina: Asociación Argentina
de Microbiología. [consultado el 1 de febrero
de 2023].
8.
Cassán, F. D.; Díaz-Zorita, M. 2016. Azospirillum
sp. in current agriculture: from the
laboratory to the field. Soil Biol. Biochem. 103:117-130. doi:
10.1016/j.soilbio.2016.08.020
9.
CEIEG. 2011. Ubicación Geográfica; Climas; Vegetación y Uso del Suelo;
Edafología; Geología; Fisiografía; Hidrografía; Áreas Naturales Protegidas.
Población. Villaflores, Chiapas.
10.
Chibeba, A. M.; Guimarães, M. D. F.; Brito, O. R.; Nogueira, M. A.; Araujo, R.
S.; Hungria, M. 2015. Co-inoculation of soybean with Bradyrhizobium
and Azospirillum promotes early nodulation. Am. J. Plant Sci. 6:1641-1649.
https://doi.org/10.4236/ajps.2015.610164
11.
Cruz-Macías, O. W.; Rodríguez-Larramendi, R. L.; Salas-Marina, M. A.;
Hernández-García, V.; Campos-Saldaña, R. A.; Chávez-Hernández, M. H.;
Gordillo-Curiel, A. 2020. Efecto de la materia orgánica y la capacidad de
intercambio catiónico en la acidez de suelos cultivados con maíz en dos
regiones de Chiapas, México. Terra Latinoamericana. 38: 475-480. DOI:
https://doi.org/10.28940/terra.v38i3.506
12.
El-Khawas, H.; Adachi, K. 1999. Identification
and quantification of auxins in culture media of Azospirillum and Klebsiella
and their effect on rice roots.
Biol. Fertil. Soils. 28: 377-381.
13. Fukami, J.; Nogueira, M. A.; Araujo,
R. S.; Hungria, M. 2016. Accessing inoculation methods 327 of
maize and wheat with Azospirillum brasilense. AMB Express. 6(1): 3.
14.
Fukami, J.; Abrantes, J. L. F.; del Cerro, P.; Nogueira, M. A.; Ollero, F. J.;
Megías, M.; Hungria, M. 2017. Revealing
strategies of quorum sensing in Azospirillum brasilense strains 325
Ab-V5 and Ab-V6. Archives of Microbiology: 1-10. 326
15.
Galindo, F. S.; Teixeira-Filho, M. C. M.; Buzetti, S.; Santini, J. M. K.;
Alves, J. M. K.; Ludkiewicz, M. G. Z. 2017. Wheat yield in the Cerrado as affected by nitrogen fertilization and
inoculation with Azospirillum brasilense. Pesqi. Agropecu. Bras. 52:
794-805. doi:10.1590/s0100-204x2017000900012
16.
Galindo, F. S.; Teixeira Filho, M. C. M.; Buzetti, S.; Pagliari, P. H.;
Santini, J. M. K. 2019. Maize yield response to nitrogen rates and
sources associated with Azospirillum brasilense. Agron J. 111: 1985-1997.
https://doi.org/10.2134/agronj2018.07.0481
17.
Gavilanes, F. Z.; Souza Andrade, D.; Zucareli, C.; Horácio, E. H.; Sarkis
Yunes, J.; Barbosa, A. P.; Ribeiro Alves, L. A.; García Cuzatty, L.; Raju
Maddela, N.; Guimarães, M. de F. 2020. Co-inoculation
of Anabaena cylindrica with Azospirillum brasilense increases
grain yield of maize hybrids. Rhizosphere. 15: 100224. doi:
10.1016/j.rhisph.2020.100224
18.
Gordillo Curiel, A.; Rodríguez Larramendi, L. A.; Salas Marina, M. Á.; Rosales
Esquinca, M. de los Á.. 2020. Efecto del ácido
salicílico sobre la germinación y crecimiento inicial del café (Coffea
arabica var. Costa Rica 95). Revista de la Facultad de Agronomía de la
Universidad del Zulia. 38(1): 43-59. Retrieved from https://produccioncientificaluz.org/index.php/agronomia/article/view/34729
19.
Grellet-Naval, N.; Vera, L.; Leggio, N. F.; Fernández, G. P.; Sánchez, D. A.;
Fernández U. J.; Romero, E. R.; Tórtora, M. L. 2017. Evaluación de la cepa Azospirillum
brasilense Az39 como biofertilizante para el cultivo de sorgo azucarado.
Revista Industrial y Agrícola de Tucumán. 94: 31-39.
20.
Hayat, Q.; Hayat, S.; Irfan, M.; Ahmad, A. 2010. Effect
of exogenous salicylic acid under changing environment: a review. Environ. Exp.
Bot. 68: 14-25. doi: 10.1016/j.envexpbot.2009.08.005
21. Hayat, S.; Ahmad, A. 2007. Salicylic Acid a Plant Hormone. Springer Publishers,
Dordrecht, The Netherlands.
22. Hernández, A.; Rives, N.; Caballero, A.; Hernández, A. N.; Heydrich,
M. 2004. Caracterización de rizobacterias asociados al cultivo del maíz en la
producción de metabolitos del tipo AIA, sideróforos y ácido salicílico, Rev.
Col. Biotec. 6: 6-13.
23.
Hugalde, I.; Riaz, S.; Agüero, C. B.; Romero, N.; Barrios-Masias, F.; Nguyen,
A. V.; Vila, H.; McElrone, A.; Gómez Talquenca, S.; Arancibia, C.; Walker, M.
A. 2017. Physiological and genetic control of
vigour in a ‘Ramsey’ × ‘Riparia Gloire de Montpellier’ population. Acta
Horticulture. 1188: 205-212.
24.
Hungria, M.; Campo, R. J.; Souza, E. M.; Pedrosa, O. F. 2010. Inoculation with selected strains of Azospirillum brasilense and A.
lipoferum improves yields of maize and wheat in Brazil. Plant Soil. 331: 413-425.
https://doi.org/10.1007/s11104-009-0262-0
25.
Montejo-Martínez, D.; Casanova-Lugo, F.; García-Gómez, M.; Oros-Ortega, I.;
Díaz-Echeverría, V.; Morales-Maldonado, E. R. 2018. Foliar and radical response of maize to biological-chemical
fertilization in a Luvisol soil. Agronomía Mesoamericana.
29: 325-341. https:// dx.doi. org/10.15517/ma.v29i2.29511
26.
Nandy, S.; Das, T.; Dey, A. 2021. Role
of jasmonic acid and salicylic acid signaling in secondary metabolite
production. In: Aftab, T.; Yusuf, M. (eds). Jasmonates and salicylates signaling
in plants. signaling and communication in
plants. Springer.
Cham. https://doi. org/10.1007/978-3-030-75805-9_5
27.
Norma Oficial Mexicana NOM-021-RECNAT-2000. 2002. Norma Oficial Mexicana que
establece las especificaciones de fertilidad, salinidad y clasificación de
suelos. Estudios, muestreos y análisis. Diario Oficial de la Federación.
México, D. F.
28.
Pankievicz, V. C. S.; do Amaral, F. P.; Santos, K. F. D. N.; Agtuca, B.; Xu,
Y.; Schueller, M. J.; Arisi, A. C. M.; Steffens, M. B.; de Souza, E. M.;
Pedrosa, F. O.; Stacey, G.; Ferrieri, R. A. 2015. Robust biological nitrogen fixation in a model grass-bacterial
association. Plant J. 81: 907-919. doi:10.1111/tpj.12777
29.
Peña-Datoli, M.; Hidalgo-Moreno, C.; González-Hernández, V.; Alcántar-González,
E.; Etchevers- Barra, J. 2016. Recubrimiento de semillas de maíz (Zea mays
l.) con quitosano y alginato de sodio y su efecto en el desarrollo radical.
Agrociencia. 50: 1091-1106.
30. Pereg, L.; de-Bashan, L. E.; Bashan,
Y. 2016. Assessment of affinity and specificity of Azospirillum
for plants. Plant Soil. 399: 389-414.
https://doi.org/10.1007/s11104-015-2778-9
31. Poorter, H.; Nagel, O. 2000. The role
of biomass allocation in the growth response of plants to different levels of
light, CO2,
nutrients and water: a quantitative review. Australian Journal of Plant Physiolgy.
27(12): 595-607. doi: 10.1071/PP99173_CO
32.
Rangel-Lucio, J. A.; Rodríguez-Mendoza, M. N.; Ferrera-Cerrato, R.;
Castellanos-Ramos, J. Z.; Ramírez-Gama, R. M.; Alvarado-Bárcenas, E. 2011.
Afinidad y efecto de Azospirillum spp. en maíz.
Agronomía Mesoamericana. 22(2): 269-279.
33. Raskin, I. 1992. Role
of salicylic acid in plants. Ann. Rev. Plant Physiol. Molec. Biol. 43:
439-463.
34. Rivas-San Vicente, M.; Plasencia, J. 2011.
Salicylic acid beyond: its role in plant growth and development. J. Exp. Bot. 62: 1-18.
35.
Rodríguez-Larramendi, L. A.; Ramírez, M. G.; Gómez-Rincón, M. A.;
Guevara-Hernández, F.; Salas- Marina, M. Á.; Gordillo-Curiel, A. 2017. Efectos
del ácido salicílico en la germinación y crecimiento inicial de plántulas de
frijol (Phaseolus vulgaris L.). Rev. Fac. Agron. (LUZ). 34: 253-269.
36.
Servicio de Información Agroalimentaria y Pesquera (SIAP). 2021. Producción
agrícola. Gobierno de México. https://www.gob.mx/siap/acciones-y-programas/produccion-agricola-33119 (fecha de consulta:
02/08/2022).
37.
Shakirova, F. M.; Sakhabutdinova, A. R.; Bezrukova, V.; Fatkhutdinova, R. A.;
Fatkhutdinova, D. R. 2003. Changes
in the hormonal status of wheat seedlings induced by salicylic acid and
salinity, Plant Science. 164: 317-322.
38.
Sierra-Macías, M.; Becerra-Leor, E. N.; Palafox-Caballero, A.; Barrón-Freyre,
S.; Cano-Reyes, O.; Zambada-Martínez, A.; Sandoval-Rincón, A.; Romero-Mora, J.
2004. Caracterización de híbridos de maíz (Zea mays L.) con alta calidad
de proteína por su rendimiento y tolerancia a pudrición de mazorca en el
sureste de México. Revista Mexicana de Fitopatología. 22: 268-276.
39.
StatSoft, Inc. 2007. STATISTICA (data analysis software system), version 8.0. http://www.statsoft.com.
40.
Stevens, J.; Senaratna, T.; Sivasithamparam, K. 2006. Salicylic
acid induces salinity tolerance in tomato (Lycopersicon esculentum cv.
Roma): Associated changes in gas exchange, water relations and membrane stabilisation.
Plant Growth Regul. 49: 77-83. https://doi.
org/10.1007/s10725-006-0019-1
41. Tucuch-Hass, C.; Alcátara, G. G.;
Trejo, T. L. I.; Volke, H. H.; Salinas, M. Y. y Larqué, S. A. 2017. Efecto del ácido
salicílico en el crecimiento, estatus nutrimental y rendimiento en maíz (Zea
mays). Agrociencia. 51: 771-781.
42.
Villar, R.; Ruiz-Robleto, J.; Quero, J.; Poorter, H.; Valladares, F.; Marañón,
T. 2004. Tasas de crecimiento en especies leñosas: aspectos funcionales e
implicaciones ecológicas. En: Valladares, F. (Ed.), Ecología del bosque
mediterráneo en un mundo cambiante. Madrid, España: Ministerio de Medio
Ambiente. EGRAF, S. A. 193-230.
43. Zeffa, D. M.; Perini, L. J.; Silva, M. B.; de Sousa, N. V.;
Scapim, C. A.; Oliveira, A. L. M. d.; Texeira do Amaral, J. A.; Simoes, A. G.
L. 2019. Azospirillum brasilense promotes increases in growth and nitrogen use
efficiency of maize genotypes. PLoS ONE. 14(4): e0215332. https://doi.
org/10.1371/journal.pone.0215332.