Rev. FCA UNCuyo | 2026 | 58(1) | ISSN 1853-8665
Production and animal health
https://doi.org/10.48162/rev.39.213
Synergistic Action of the Essential Oils of Elionorus muticus and Lippia junelliana Against the Causative Agent of Chalkbrood in Bees (Ascosphaera apis)
Acción sinérgica de los aceites esenciales de Elionorus muticus y Lippia junelliana sobre el agente causal de la cría yesificada en abejas (Ascosphaera apis)
Maria Laura Susevich 2,
Francisco Jose Reynaldi 5,
Graciela Noemi Albo 1
1 Universidad Nacional de La Plata (UNLP). Facultad de Ciencias Agrarias y Forestales. 119 y 60 y s/n. C. P. 1900. La Plata. Buenos Aires. Argentina.
2 Universidad Nacional de La Plata (UNLP). Facultad de Ciencias Naturales y Museo. Centro de Estudios Parasitológicos y de Vectores. Bvard. 120 entre Av. 60 y 64 S/N. C. P. 1900. La Plata. Buenos Aires. Argentina.
3 Universidad Nacional de La Plata (UNLP). Facultad de Ciencias Naturales y Museo. Instituto de Botánica Carlos Spegazzini. 122 y s/n. C. P. 1900 La Plata. Buenos Aires. Argentina.
4 Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET). Godoy Cruz 2290. Ciudad Autónoma de Buenos Aires 1425. Argentina.
5 Universidad Nacional de La Plata (UNLP). Facultad de Ciencias Veterinarias. Cátedra de Micología Médica. Centro de Microbiología Básica y Aplicada (CEMIBA). Calle 60 y 118 s/n. C. P. 1900. La Plata. Buenos Aires. Argentina.
* maricel.vega@agro.unlp.edu.ar
Abstract
Ascosphaera apis is the fungus that causes chalkbrood in Apis mellifera. Traditionally, its control has relied on synthetic fungicides, which can be harmful to bees and ecosystems. An alternative treatment has explored safer alternatives, such as the antimicrobial properties of plant-based essential oils. This study evaluated the combined effect of the essential oils of Elionorus muticus and Lippia junelliana on 10 isolates of A. apis using a “chessboard” experimental design. Minimum inhibitory concentrations, fungal growth rates and inhibition percentages were determined. Analyses were performed with ANOVA and post-hoc tests. The results showed that the combination of 200 μL/L of E. muticus and 25 μL/L of L. junelliana (C5) completely inhibited fungal growth after 144 h, with MIC50 significantly more effective than controls. The combined use of E. muticus and L. junelliana shows synergistic activity (FICI ≤0.5) in controlling A. apis and achieves full inhibition of the fungus under controlled conditions. These findings suggest that essential oils may be a viable alternative for the sustainable management of bee diseases.
Keywords: biocontrol, pathogenic fungi, natural products, apiculture
Resumen
Ascosphaera apis es el hongo que causa la cría yesificada en Apis mellifera. Tradicionalmente, su control ha dependido de fungicidas sintéticos, que pueden ser dañinos para las abejas y el ecosistema. Por ello, se han investigado alternativas más seguras, como los aceites esenciales de plantas, que poseen propiedades antimicrobianas. Este estudio evaluó el efecto combinado de los AEs de Elionorus muticus y Lippia junelliana sobre diez aislamientos de A. apis, utilizando un diseño experimental tipo “tablero de ajedrez”. Se determinaron las concentraciones inhibitorias mínimas y se calcularon las tasas de crecimiento fúngico y porcentajes de inhibición mediante ANOVA y pruebas post hoc. Los resultados indicaron que la combinación de 200 μL/L de E. muticus y 25 μL/L de L. junelliana (C5) inhibió completamente el crecimiento del hongo tras 144 horas, con una CIM50 significativamente más efectiva que los controles. La combinación de aceites esenciales E. muticus y L. junelliana presenta una solución prometedora para el control de A. apis, con un efecto sinérgico ≥0,5; que permite una inhibición total del hongo en condiciones controladas. Estos hallazgos sugieren que los AEs podrían ser una alternativa viable para el manejo sostenible de enfermedades en abejas.
Palabras clave: biocontrol, hongo patógeno, productos naturales, apicultura
Originales: Recepción: 20/03/2025 - Aceptación: 26/09/2025
Introduction
Mycotic infections are major contributors to the decline of honey bee (Apis mellifera L.) populations, causing significant economic losses in apiculture (6). Among them, the entomopathogenic fungus Ascosphaera apis (Maasen ex Claussen) Spiltoir & Olive (1955) causes ascospherosis, or chalkbrood, characterized by dehydrated larvae that change from white to black (depending on the number of sexual reproduction structures present). The infection begins when larvae inadvertently ingest fungal spores with their food (5). Once in larval hindgut, the spores germinate, and subsequent mycelial development causes larval death (22). The fungus affects the bee brood during the pupal stage, reducing the colony’s health and productivity (1). Transmission is facilitated by the accumulation of A. apis spores in the hive, on beekeeping tools, and in food sources such as nectar and pollen (32).
Currently, ascospherosis is considered a cosmopolitan disease (25). Chemicals are widely used to treat and control chalkbrood, but their use is allowed only in certain countries and under critical conditions. Another alternative is the use of natural products, such as essential oils (EOs) and plant extracts (25). According to some researchers, synthetic fungicides have been linked to increased bee mortality, colony contamination, and reduced beneficial fungi that ferment pollen and support bee nutrition (34). Therefore, natural and ecologically safe products provide a promising option for managing chalkbrood.
Essential Oils (EOs) have long been known to exert antimicrobial effects against pathogens in plants, animals, insects, and honey bees (21, 27). EOs are aromatic substances produced by the secondary metabolism of aromatic plants and can be extracted from leaves, flowers, fruits, seeds, sprouts, rhizomes, roots and bark (23). Lippia junelliana (Mold.) Tronc. (Verbenaceae) is an aromatic shrub endemic to the Prepuna and Chaqueña biogeographic regions of Argentina (18). Elionorus muticus (Spreng.) is a native grass widely distributed in Argentina and other South American countries (24). Vega et al. (2022) determined that 500 μL/L of L. junelliana was safe and palatable to honey bees. Albo et al. (2014) established the minimum inhibitory concentration (MIC50) of L. junelliana EO capable of controlling the chalkbrood pathogen. Up to now, few studies have examined the use of E. muticus to control ascospherosis (2).
Mráz et al. (2023) suggested that one limitation of EO treatment is the substantial amount of vegetal material required to obtain enough for in vivo application. Combining different EOs could address this issue, allowing a reduction in the applied dose in honey bee colonies. However, most studies have focused on the effects of individual EOs. The present study aimed to evaluate the in vitro effects of a combination of E. muticus and L. junelliana on the pathogenic fungus A. apis and to determine the fractional inhibitory concentration index (FICI) of the combinations for potential field application.
Materials and Methods
Isolation and Culture of A. apis
Ten A. apis isolates were selected for this study due to their reported entomopathogenic capacity in A. mellifera brood (table 1) (29). These isolates were stored at -20°C in vials containing 20% glycerol in sterile distilled water and deposited in the Animal Production Course I collection, Facultad de Ciencias Agrarias y Forestales, Universidad Nacional de La Plata (FCAyF, UNLP). For fungal inoculum, isolates of A. apis were cultured on Petri dishes with MY20 medium (5 g peptone, 3 g yeast extract, 3 g malt extract, 200 g glucose and 20 g agar per liter of distilled water) at 35°C for 7 days, following the methodology of Albo et al. (2016).
Table 1. Fungal isolates of A. apis and their geographical origin.
Tabla 1. Aislamientos fúngicos de A. apis y su origen geográfico.
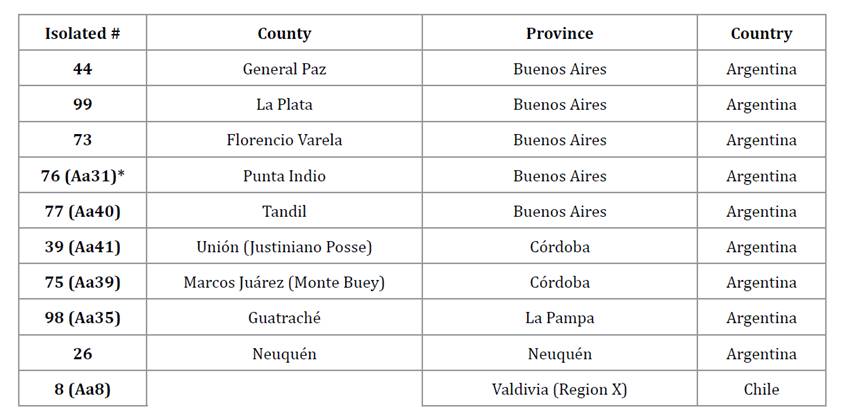
*Isolation number assigned by Reynaldi et al., 2003.
*Número de aislamiento asignado en Reynaldi et al., 2003.
Essential Oils
The EOs of E. muticus and L. junelliana were provided by the Aromatics Group, Institute of Instituto Nacional de Tecnología Agropecuaria (INTA), Castelar, Buenos Aires, Argentina. The EOs were extracted from plant tissue via Clevenger hydrodistillation (IRAM Standard No. 18.729) and stored in brown glass vials at -20°C for later use. Vega et al. (2022) described the chemical characterization of the EOs. The main components of L. junelliana are cis- and trans-ocimenone, myrcenone, cis-davone, and camphor, while those of E. muticus are geranial, neral, geranyl acetate, linalool, and methyl heptenone.
Experimental Procedure
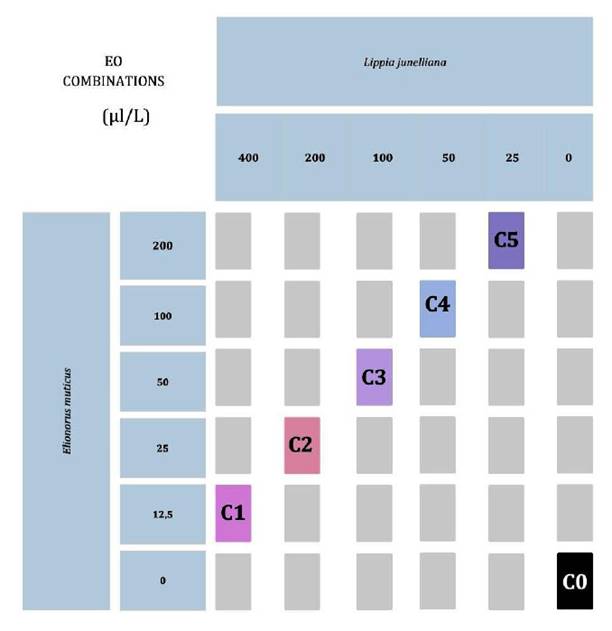
To assess the effect of the combination of EOs on the entomopathogenic fungus, the “chessboard” method was used (13). Initial concentrations of 200 μL/L for E. muticus and 400 μL/L for L. junelliana were selected based on preliminary trials (unpublished data). The experimental design, shown in figure 1, included five decreasing concentrations in a geometric series. Dilutions of the EOs were prepared using 2.5% v/v ethanol at 70°C as a diluent, which was then added to the melted MY20 liquid medium (45°C) and poured into Petri dishes to create each combination. Approximately 20 ml of medium was poured into glass Petri dishes (9 cm x 1.5 cm). Mycelium discs (7 mm diameter) from each fungal isolate were placed on the medium surface with the different EO/diluent combinations in duplicate. As a control, we inoculated each strain in MY20 medium. All plates were incubated at 35°C, 12% CO₂, 65% relative humidity, and in darkness for 144 h. The fungal growth diameter (mm) was measured with a caliper every 24 h.
Figure 1. Chessboard. The matrix was structured with rows representing E. muticus concentrations and columns representing L. junelliana concentrations. Each box at the row-column intersection represents a combination of E. muticus and L. junelliana.
Figura 1. Tablero de ajedrez. La matriz se estructuró utilizando filas (Concentraciones de E. muticus) y columnas (Concentraciones de L. junelliana), en la cual cada casilla (intersección de fila y columna) representa una combinación de E. muticus y L. junelliana.
Growth Parameters
Based on the data obtained, the absolute growth was calculated for each culture time, and growth curves were constructed. The exponential growth rate per hour (GR; mm/h) was determined using the following equation 1:
TC = Σ (DCC - DCT)/DCC × 100% (1)
DCC corresponds to the control growth diameter and DCT to the treatment growth diameter.
TC = C24h + C48h + C72h + C144h/4 (2)
C24h = total mycelium length - 7 (3)
C48h = (total mycelium length - 7) - C24h (4)
C72h = (total mycelium length - 7) - C48h (5)
C144 h = (total mycelium length - 7) - C72h (6)
ANOVA followed by Tukey’s post-hoc test (p = 0.05) was used to determine differences among the GRs for each treatment.
Descriptive statistics were calculated for the final fungal growth diameter at the end of the experiment (144 h). ANOVA followed by Holm-Sidak’s test was used to determine differences between the control and EOs combinations. Then, Tukey’s post-hoc test (p ≤= 0.05) was used to determine differences among EO combinations, excluding the control.
Percent Growth Inhibition
The percentage of growth inhibition (GI) was calculated according to Chen et al. (2019) using the following formula (Eq. 7):
IC (%) = (DCC - DCT)/DCC × 100% (7)
Where DCC is the control growth diameter and DCT is the treatment growth diameter.
A bifactorial ANOVA followed by Tukey’s post hoc test (p ≤ 0.05) was conducted to assess differences among EO combinations and culture times on growth inhibition percentage. Before statistical analysis, percentage data were transformed using arcsine √p.
Minimum Inhibitory Concentration (MIC50)
To assess the efficacy of the combination of EOs against the entomopathogenic fungus, the number of inhibited strains relative to the control was calculated (10). The MIC50 was determined at the time points of the linear growth phase (72 and 144 h). It was defined as the lowest EO concentration causing 50% or more inhibition compared to the control (4).
Fractional Inhibitory Concentration Index (FICI)
The FICI value was calculated from the results obtained for MIC50 using the following equations (36):
FICi = FICa + FICb (8)
FICa = A/MICa (9)
FICb = B/MICb (10)
A, MIC of EO A in combination; MICa, MIC of EO A alone; B, MIC of EO B in combination; MICb, MIC of EO B alone.
FICI values were interpreted according to Amassmoud et al. (2023) as follows: synergism (FICI ≤ 0.5), additive (0.5 < FICI ≤ 1.0), neutral (1 < FICI ≤ 4.0), or antagonism (FICI > 4.0).
Results
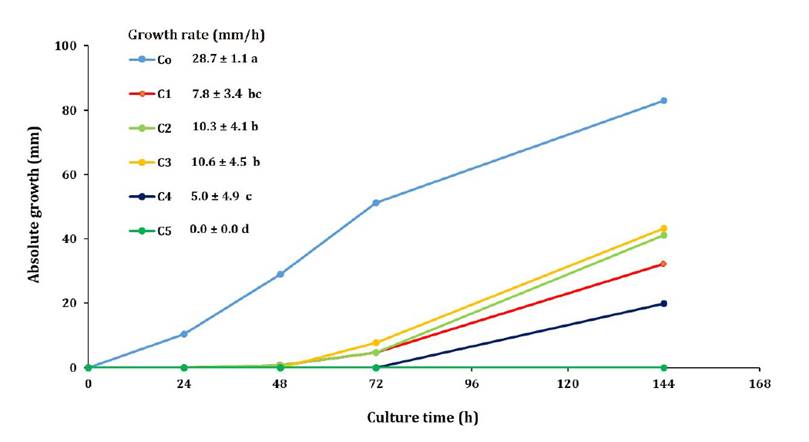
Figure 2, shows the growth curves and rates of A. apis for the EO combinations and control. All EO combinations inhibited fungal growth during the logarithmic phase. A. apis required more than 72 h to overcome the fungistatic effect produced by C4 (100 μL/L E. muticus and 50 μL/L L. junelliana). Fungal growth was not observed when exposed to C5 (200 μL/L E. muticus and 25 μL/L L. junelliana) after 144 h of culture. All EO combinations showed growth rates below 50% compared to the control (F: 102.29; p < 0.001).
Different letters in the growth rate values indicate significant differences among treatments (Tukey’s test, p < 0.05).
Letras distintas en los valores de tasa de crecimiento indican diferencias significativas entre los tratamientos (Test de Tukey, p < 0,05).
Figure 2. Growth curves and rates of A. apis.
Figura 2. Curvas y tasas de crecimiento de A. apis.
Table 2, shows descriptive and parametric analyses of A. apis growth at the final time point (144 h) after exposure to the EO combinations. ANOVA revealed significant differences among treatments (F: 136.08; p < 0.0001), with each EO combination differing from the control with a highly significant p value (p ≤ 0.001, Holm-Sidak’s test). Growth was strongly affected by combination C5, followed by C4 (Tukey’s test, p < 0.05).
Table 2. Descriptive statistics and post-hoc parametric analysis of EO combinations on A. apis growth diameter (mm) at 144 h of culture.
Tabla 2. Estadística descriptiva y análisis paramétrico post-hoc de la combinación de los AEs sobre el diámetro de crecimiento (mm) de A. apis a las 144 h de cultivo.
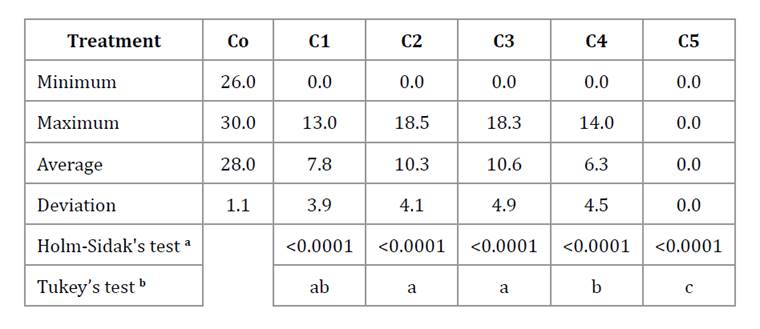
a p value obtained. b Different letters indicate significant differences among treatments (p < 0.05).
a, valor de p obtenido. b, Letras distintas indican diferencias significativas entre los tratamientos (p < 0,05).
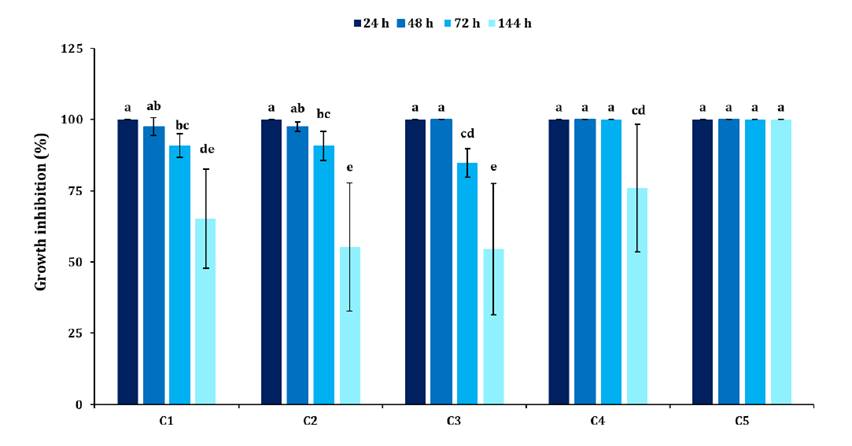
The relative growth inhibition of A. apis showed significant differences (F: 9.01; p < 0.001), depending on the EO combination and culture time (figure 3). All EO combinations and culture times inhibited 50% of fungal growth; however, C5 was the only one that achieved 100% inhibition at all time points.
Different letters indicate significant differences among treatments (p < 0.05).
Letras distintas indican diferencias significativas entre los tratamientos (p < 0,05).
Figure 3. Growth inhibition (%) of A. apis the EO combinations.
Figura 3. Inhibición porcentual del crecimiento de A. apis en las diferentes combinaciones de los Aceites esenciales.
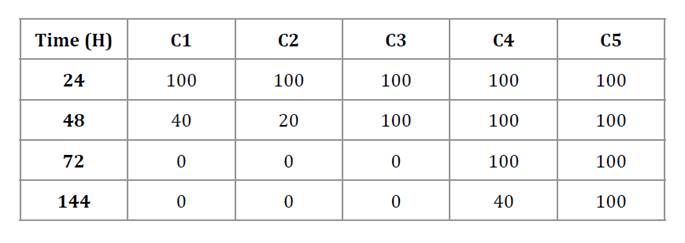
The relative growth inhibition of A. apis strains at each culture time is shown in table 3. The percentage of affected strains (< 50%) decreased as culture time increased for combinations C1, C2, C3, and C4, while C5 maintained inhibition of 50% or more at all time points. Based on the MIC₅₀ values, the FICI was calculated. The FICI for C5 was 0.5, indicating that the combination of both EOs exerted a synergistic effect that led to the total inhibition of A. apis strains.
FICA = 200/400 = 0.5
FICB = 25/3200 = 0.007
FICi = 0.5 + 0.0 = 0.5
Table 3. Relative percentage inhibition of A. apis strains at different culture times.
Tabla 3. Porcentaje relativo de inhibición del número de cepas de A. apis en los distintos tiempos de cultivo.
Discussion
Essential oils have well-known antimicrobial properties and are used to treat infections caused by several pathogens (5). Previous studies (briefly described below) have examined Lippia junelliana and Elionurus muticus separately against pests and pathogens, but never in combination. This study investigated the inhibitory effects of combining L. junelliana and E. muticus against A. apis.
The EO of L. junelliana is mainly composed of myrcenone and cis-davanone (33). Comelli et al. (2023) reported strong insecticidal activity against Oryzaephilus mercator (L.) (Nitidulidae), with LC50 values of 7.02–7.17 μL/L and LC100 values of 10.00-20.00 μL/L. El Youssfi et al. (2024) demonstrated that this EO exhibits virucidal activity against the hemorrhagic yellow fever virus (Junín virus), with a MIC50 (VC50) of 20 ppm. Regarding its antifungal activity, in vitro tests showed that L. junelliana effectively controlled molds and yeast, including Candida krusei, C. albicans, and Aspergillus spp. of the section fumigati (13). Regarding pests and pathogens in beekeeping, Dellacasa et al. (2003) found that L. junelliana repelled the mite Varroa destructor at 1.9 μL per bee. The inhibitory effect of this EO against A. apis was studied alone but never in combination (15).
The E. muticus essential oil used in this study was mainly composed of approximately 50% α-citral (geranial) and 25% β-citral (neral) (33). In vivo tests demonstrated its fumigant activity against the agricultural pest Tetranychus urticae C.L. Koch (Acari) at low concentrations (0.03-1 μL/L) (17). Regarding its bactericidal activity, E. muticus was effective against Escherichia coli, coagulase-negative Staphylococcus, S. aureus, S. pseudintermedius, S. uberis, P. aeruginosa, and Bacillus cereus, with MIC values ranging from 1 to 150 mg/mL (8). Fungicidal activity was observed against Candida albicans (9), C. krusei, and C. neoformans Vuillemin (20), with MIC values of 0.5-5 μg/mL. Fungistatic activity was reported against C. parapsilosis and C. utilis (28).
Davis and Ward (2003) determined that the minimum fungicidal concentration against A. apis was 250 ppm for C. citratus, Eucalyptus citriodora, and Leptospermum petersonii oils, all rich in citral. Similarly, the essential oil of Litsea cubeba (Lour.) Pers (Lauraceae) showed fungicidal activity against A. Apis at 50 μg/mL using the agar diffusion method (4, 26). Dellacasa et al. (2003) reported that 800 ppm of Tagetes minuta, which shares components with L. junelliana, was required to control Ascosphaera apis in vitro. In our study, the combination of E. muticus (200 μL/L) and L. junelliana (25 μL/L), referred to as C5, produced a FICI of 0.5, showing the most effective control of A. apis through synergistic action. This effect likely resulted from the complementary interaction of the oils’ active components, yielding greater antifungal efficacy than either oil used alone. This finding aligns with recent research showing that synergistic essential oil combinations can fully inhibit various fungal pathogens (35). According to previous studies, citral, a major component of the E. muticus EO used here, effectively inhibited both dematiaceous fungi and the hyaline fungus causing chalkbrood (7, 29). Our combination of L. junelliana and E. muticus reduced the required L. junelliana amount by at least 75% compared to the 800 ppm reported by Dellacasa et al. (2003) for Tagetes minuta, which contains similar major components.
It is important to highlight the time-dependent effects of the essential oil combinations on A. apis. Combination C4 (100 μL/L of E. muticus and 50 μL/L of L. junelliana) showed a fungistatic effect, requiring more than 72 h for the fungus to recover. However, it did not fully inhibit fungal growth. Analysis of relative inhibition percentages and their temporal variation revealed significant differences in the efficacy of the essential oil combinations. Combinations C1, C2, C3, and C4 showed reduced effects over time, whereas C5 consistently maintained inhibition above 50% throughout the study. Sustained complete inhibition is desirable for controlling fungal pathogens, providing more effective and long-lasting protection (20). The synergistic effect and temporal stability observed make C5 a promising candidate for controlling Ascosphaera apis.
Conclusions
Our results showed that combining L. junelliana (25 μL/L) and E. muticus (200 μL/L) synergistically inhibited A. apis growth. This combination outperformed the inhibitory efficacy of each oil alone and reduced the required amount of L. junelliana by at least 75% compared to previously reported values, lowering the costs of potential health management. Although our results provide valuable information on in vitro control dosages, further studies on the toxicity of the tested compounds in larval, pupal, and adult honey bees are needed to successfully apply this knowledge in field conditions.
1. Albo, G. N.; Reynaldi, F. J.; Elder, H.; Juárez, M. A.; Córdoba, S. B. 2014. Control in vitro de Ascosphaera apis con Schinus molle y Lippia junelliana. Efectos tóxicos potenciales en abeja melífera. XI Congreso Latinoamericano de Apicultura FILAPI.
2. Albo, G. N.; Altamirano, R.; Henning, C.; Della Vedova, R.; Córdoba, S. B. 2015. Inhibición in vitro de Ascosphaera apis con aceites esenciales de gramíneas nativas e introducidas. III Congreso Argentino de Microbiología Agrícola y Ambiental. Ciudad Autónoma de Buenos Aires. Argentina.
3. Albo, G., Reynaldi, F., Córdoba, S., & Malbrán, C. 2016. Evaluación de la actividad inhibitoria, in vivo e in vitro, del aceite esencial de Cymbopogon citratus y cinco diluyentes sobre cultivos de Ascosphaera apis. Analecta veterinaria. 36: 5–11.
4. Amassmoud, O.; Abbad, I.; Iriti, M.; Hassani, L.; Mezrioui, N.; Abbad, A. 2023. Antibacterial activity of essential oils combinations based on Thymus broussonnetii, and their synergism with some antibiotics. Current Microbiology. 80(12): 398.
5. Ansari, M. J.; Al-Ghamdi, A.; Usmani, S.; Khan, K. A.; Alqarni, A. S.; Kaur, M.; Al-Waili, N. 2017. In vitro evaluation of the effects of some plant essential oils on Ascosphaera apis, the causative agent of Chalkbrood disease. Saudi Journal of Biological Sciences. 24(5): 1001-1006. https://doi.org/10.1016/j.sjbs.2016.04.016
6. Aziz, M. A.; Alam, S. 2024. Diseases of Honeybee (Apis mellifera). In Melittology-New Advances. IntechOpen.
7. Capetti, F.; Tacchini, M.; Marengo, A.; Cagliero, C.; Bicchi, C.; Rubiolo, P.; Sgorbini, B. 2021. Citralcontaining essential oils as potential tyrosinase inhibitors: A bio-guided fractionation approach. Plants. 10(5): 969.
8. Celaya, L. S.; Le Vraux, M. A.; Heit, C. I.; Viturro, C. I.; Martina, P. F. 2023. Phytochemical and Biological Profile of Essential Oils of Elionurus muticus (Spreng.) Growing in Northeastern Argentina. Chemistry & Biodiversity. 20(6), e202201105. https://doi.org/10.1002/cbdv.202201105
9. Chagonda, L. S.; Fungirayi, B. 2016. Antifungal activity of the essential oil of Elionurus muticus (Spreng) Kunth from Zimbabwe against Candida albicans, C. krusei and Cryptococcus neoformans. J Multidiscip Eng Sci Technol. 3: 5331-5335.
10. Chen, C. J.; Li, Q. Q.; Ma, Y. N.; Wang, W.; Cheng, Y. X.; Xu, F. R.; Dong, X. 2019. Antifungal effect of essential oils from five kinds of Rutaceae plants-avoiding pesticide residue and resistance. Chemistry & Biodiversity. 16(4): e1800688.
11. Clinical and Laboratory Standards Institute (CLSI). 2008. Reference method for broth dilution antifungal susceptibility testing of filamentous fungi. Approved Standard CLSI. Doc. M 38- A2. 2° Ed. Wayne.
12. Comelli, N. C.; Diez, P. A.; Rodríguez, M. R.; Denett, G. O.; López, T. E.; Bracamonte, D. M.; Ortiz, E. V.; Sampietro, D. A.; Duchowicz, P. R. 2023. Excito-repellent and Pesticide-Likeness Properties of Essential Oils on Carpophilus dimidiatus (Fabricius) (Nitidulidae) and Oryzaephilus mercator (L.) (Silvanidae). Journal of Chemical Information and Modeling. https://doi.org/10.1021/acs.jcim.3c01198
13. Córdoba, S.; Vivot, W.; Szusz, W.; Albo, G. 2019. Antifungal Activity of Essential Oils Against Candida Species Isolated from Clinical Samples. Mycopathologia. 184(5): 615-623. https://doi.org/10.1007/s11046-019-00364-5
14. Davis, C.; Ward, W. 2003. Control of chalkbrood disease with natural products: a report for the RIRDC. Publication N° 03/107. Kingston. ACT. AU. p. 1-23.
15. Dellacasa, A. D.; Bailac, P. N.; Ponzi, M. I.; Ruffinengo, S. R.; Eguaras, M. J. 2003. In vitro activity of essential oils from San Luis-Argentina against Ascosphaera apis. Journal of Essential Oil Research. 15(4): 282-285.
16. El Youssfi, C.; Dadou, S.; Loukili, E. H.; El Hammoudani, Y.; Soujaa, H.; Rejdali, M.; Zerrouk Mohammed, H.; Mourabit, N.; Aarab, S. 2024. Biological activities of essential oils: A mini-review. BIO Web of Conferences, 109, 01031. EDP Sciences.
17. Hauschild, R.; Ott, A.; da Silva, M. A. S. 2020. Ação fumigante de óleos essenciais de Baccharis dracunculifolia e Elionurus muticus contra o Ácaro rajado (Tetranychus urticae, Koch 1836) em morangueiro. Cadernos de Agroecologia. 15(2).
18. Juárez, M. A.; Elechosa, M. A.; Molina, A. M.; Viturro, C. I.; Molina, A. C.; Heit, C. I. 2007. Estudio del aceite esencial de Lippia junelliana (Mold) Tronc. Obtenido de colectas en Tucumán, Argentina. Boletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas. 6(5): 254-255.
19. Krutmuang, P.; Rajula, J.; Pittarate, S.; Chatima, C.; Thungrabeab, M.; Mekchay, S.; Senthil-Nathan, S. 2022. The inhibitory action of plant extracts on the mycelial growth of Ascosphaera apis, the causative agent of chalkbrood disease in Honey bee. Toxicology Reports. 9: 713-719.
20. Lameck, S.; Chagonda, L.; Fungirayi, B. 2016. Antifungal Activity of the Essential Oil of Elionurus muticus (Spreng) Kunth from Zimbabwe against Candida albicans, C. krusei and Cryptococcus neoformans. Journal of Multidisciplinary Engineering Science and Technology. 3: 2458-9403.
21. Lazăr, R. N.; Pătruică, S. 2020. Use of Essential Oils in Bees. Scientific Papers Animal Science and Biotechnologies. 53(1): 74-74.
22. Lopes, L. Q. S.; Quatrin, P. M.; De Souza, M. E.; De Almeida Vaucher, R.; Vianna Santos, R. C. 2015. Fungal infections in honey bees. Fungal Genomics. Biol. 5: 1-4.
23. Mohamed, A. A.; Alotaibi, B. M. 2023. Essential oils of some medicinal plants and their biological activities: a mini review. Journal of Umm Al-Qura University for Applied Sciences. 9(1): 40-49.
24. Moreno, E. M.; Almirón, N. E. A.; Peichoto, M. C.; Solís Neffa, V. G. 2018. Variabilidad genética en poblaciones de Elionurus muticus (Poaceae) de Corrientes, Argentina, a partir de marcadores moleculares de ADN nuclear y cloroplástico. Boletín de La Sociedad Argentina de Botánica. 53(2): 1-10. https://www.scielo.org.ar/scielo.php?script=sci_abstract&pid=S1851-3722018000200012&lng=es&nrm=iso&tlng=en
25. Mráz, P.; Žabka, M.; Hoštičková, I.; Kopecký, M.; Bohatá, A.; Tomčala, A.; Hýbl, M. 2023. Effect of selected botanical compounds on Ascosphaera apis and Apis mellifera. Industrial Crops and Products. 197: 116649. https://doi.org/10.1016/j.indcrop.2023.116649
26. Nardoni, S.; D’Ascenzi, C.; Rocchigiani, G.; Papini, R.; Pistelli, L.; Formato, G.; Najar, B.; Mancianti, F. 2017. Stonebrood and chalkbrood in Apis mellifera causing fungi: In vitro sensitivity to some essential oils. Natural Product Research. 32: 1-6. https://doi.org/10.1080/14786419.2017.1306703
27. Pérez Panti, O.; García de la Cruz, R.; González Hernández, H.; Sánchez Soto, S.; Moscoso Ramírez, P. A.; Izquierdo Reyes, F. 2024. Bio-efficacy of entomopathogenic fungi and vegetable oils against the pink pineapple mealybug: Dysmicoccus brevipes (Cockerell). Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina. 56(1): 83-93. DOI: https://doi.org/10.48162/rev.39.125
28. Puppin, D. de G. P. B. 2018. Ação antifúngica e toxicidade do óleo essencial de Elionurus muticus (capim-limão brasileiro). http://repositorio.unicamp.br/jspui/handle/REPOSIP/333555
29. Pusceddu, M.; Floris, I.; Mangia, N. P.; Angioni, A.; Satta, A. 2021. In vitro activity of several essential oils extracted from aromatic plants against Ascosphaera apis. Veterinary sciences. 8(5): 80.
30. Reynaldi, F. J.; López, A. C.; Albo, G. N.; Alippi, A. M. 2003. Differentiation of Ascosphaera apis isolates by rep-PCR fingerprinting and determination of chalkbrood incidence in Argentinean honey samples. Journal of Apicultural Research. 42(4): 68-76.
31. Spiltoir, C. F.; Olive, L. S. 1955. A reclassification of the genus Pericystis Betts. Mycologia. 47(2): 238-244.
32. Tapia-González, J. M.; Alcazar-Oceguera, G.; Macías-Macías, J. O.; Contreras-Escareño, F.; Tapia- Rivera, J. C.; Petukhova, T.; Guzmán-Novoa, E. 2020. Ascosferosis en abejas melíferas y su relación con factores ambientales en Jalisco, México. Revista mexicana de ciencias pecuarias. 11(2): 468-478.
33. Vega, M.; Reynaldi, F. J.; Altamirano, R.; Marcos, D.; Juárez, M. A. 2022. Preliminary report on the toxicity of Lippia junelliana (Lamiales: Verbenaceae) and Elionorus muticus (Poales: Poaceae) on Apis mellifera (Hymenoptera: Apidae). Revista de la Sociedad Entomológica Argentina. 81(1).
34. Yoder, J. A.; Jajack, A. J.; Rosselot, A. E.; Smith, T. J.; Yerke, M. C.; Sammataro, D. 2013. Fungicide contamination reduces beneficial fungi in bee bread based on an area-wide field study in honey bee, Apis mellifera, colonies. Journal of Toxicology and Environmental Health. Part A. 76(10): 587-600. https://doi.org/10.1080/15287394.2013.798846
35. Yuan, X.; Cao, D.; Xiang, Y.; Jiang, X.; Liu, J.; Bi, K.; Dong, X.; Wu, T.; Zhang, Y. 2024. Antifungal activity of essential oils and their potential synergistic effect with amphotericin B. Scientific Reports. 14(1): 31125. https://doi.org/10.1038/s41598-024-82380-0
36. Zeng, Z. Y.; Li, Q. Q.; Huo, Y. Y.; Chen, C. J.; Duan, S. S.; Xu, F. R.; Cheng Y. X.; Dong, X. 2021. Inhibitory effects of essential oils from Asteraceae plant against pathogenic fungi of Panax notoginseng. Journal of Applied Microbiology. 130(2): 592-603.