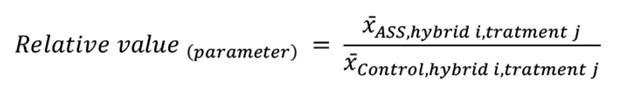Rev. FCA UNCuyo | 2026 | 58(1) | ISSN 1853-8665
Ecophysiology and crop management
https://doi.org/10.48162/rev.39.212
Ammonium Sulfate Improves Nutrient Status and Photosynthetic Activity in Sorghum (Sorghum bicolor L.) Grown in Alkaline-Saline-Sodic Soils
El sulfato de amonio mejora la absorción de nutrientes y la eficiencia fotosintética del Sorghum bicolor L. cultivado en suelos alcalinos-salinos-sódicos
María Belén Ciacci 1,
Rodrigo Ezequiel Domínguez 1,
Silvia Del Carmen Imhoff 3,
Dolores Ángela Bustos 1,
1 Instituto de Fisiología y Recursos Genéticos Vegetales (IFRGV). Centro de Investigaciones Agropecuarias (CIAP). Instituto Nacional de Tecnología Agropecuaria (INTA). Unidad de Estudios Agropecuarios (UDEA-CONICET). Av. 11 de Septiembre 4755. X5020ICA. Córdoba. Argentina.
2 Grupo de Manejo de cultivo suelo y agua. Estación Experimental Agropecuaria Oliveros. Instituto Nacional de Tecnología Agropecuaria (INTA). Ruta Nacional 11 km 353. (C2206) Oliveros. Santa Fe. Argentina.
3 ICiAgro Litoral (CONICET-UNL). Kreder 2805. Esperanza. Santa Fe.
4 Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET). Argentina.
* luna.fernando@inta.gob.ar
Abstract
This study investigated whether fertilization with urea or ammonium sulfate could mitigate the growth limitations of sorghum imposed by alkaline-saline-sodic (ASS) soils. We hypothesized that the application of these nitrogen sources would improve the plant’s mineral status and photosynthetic activity. Experiments were conducted using three sorghum hybrids under both field and greenhouse conditions, comparing ASS soil to a non-ASS control. The treatments included fertilization with ammonium sulfate, urea, and a non-fertilized control. At the end of both experiments, soil and plant samples were analyzed to assess soil pH, nutrient content, and chlorophyll fluorescence. Our results demonstrate that ammonium sulfate, but not urea, significantly improved plant mineral status by inducing acidification in the ASS soil. This improved status led to increased photosynthetic rates and biomass production compared to unfertilized controls. These findings position ammonium sulfate as an effective tool for improving sorghum growth and productivity in challenging marginal soils.
Keywords: Sorghum bicolor, alkaline-saline-sodic soils (ASS), ammonium sulfate (AS) fertilization, urea (U), plant mineral nutrition, photosynthesis
Resumen
El objetivo de este estudio fue determinar si la fertilización con urea o sulfato de amonio logra mitigar las restricciones en el crecimiento del sorgo impuestas por suelos salino-sódicos alcalinos (SSA), mejorando el estado mineral de la planta y, en consecuencia, su actividad fotosintética. Para ello, se llevaron a cabo ensayos de campo y de invernadero, usando tres híbridos de sorgo en dos tipos de suelo: uno control y otro SSA. Los tratamientos incluyeron fertilización con sulfato de amonio y urea, además de un tratamiento sin fertilizar, empleado como control. Se recolectaron muestras de suelo y planta al final de cada experimento para evaluar el pH del suelo, el contenido de nutrientes y la fluorescencia de la clorofila. Nuestros resultados demuestran que, a diferencia de la urea, la aplicación de sulfato de amonio mejoró significativamente el estado mineral de la planta en respuesta a la acidificación del suelo SSA. Además, la aplicación de sulfato de amonio aumentó las tasas fotosintéticas y la producción de biomasa en comparación con las no fertilizadas. Estos hallazgos posicionan al sulfato de amonio como una herramienta agronómica eficaz para mejorar el crecimiento del cultivo de sorgo en suelos marginales.
Palabras clave: Sorghum bicolor, salino-sódicos alcalinos (SSA), fertilización con sulfato de amonio (SA), urea (U), nutrición mineral de las plantas, fotosíntesis
Originales: Recepción: 17/07/2025- Aceptación: 22/12/2025
Introduction
Alkaline saline-sodic soils (ASS) negatively impact crop growth and productivity due to the combined effect of salinity, sodicity, and alkalinity. These soils are characterized by an electrical conductivity (EC) exceeding 4 dS m-1 (units used throughout the manuscript, ~40 mM NaCl), a sodium adsorption ratio (SAR) greater than 13 meq L-1, an exchangeable sodium percentage (ESP) over 15%, and a pH ranging from 8.5 to 10. High sodium ion (Na+) concentrations degrade soil structure by promoting clay dispersion, thereby reducing structural stability and water infiltration rates in these soils (Rengasamy, 2010). Furthermore, the high pH associated with alkalinity severely limits plant growth and nutrient uptake of essential elements, including nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), iron (Fe), and zinc (Zn) (Marschner & Rengel, 2012; Sparks, 2003). However, this method is often costly, requires the application of substantial amounts of gypsum, and its effectiveness depends on high infiltration capacity and the presence of adequate drainage systems (González et al., 2019). In addition to chemical amendments, biological processes occurring in the rhizosphere also contribute to nutrient cycling and overall soil functioning. Microorganisms associated with crop rhizospheres may play a relevant role in nutrient transformations and plant–soil interactions under agricultural conditions (Tupa-Andrade et al., 2025). Conversely, nitrogen fertilizers are widely used to meet crop N requirements. Among N sources, ammonium sulfate (AS) offers potential agronomic advantages over urea (U) in alkali saline-sodic soil environments (Chien et al., 2011). The main difference lies in the ability of AS to promote soil acidification. Specifically, through the nitrification process, AS theoretically generates twice the soil acidity per unit of applied N compared with urea (Adams, 1984; Pierre, 1928).
Sorghum (Sorghum bicolor (L.) Moench) has emerged as a highly relevant forage crop, particularly for marginal lands in regions like Córdoba, due to its inherent stress-tolerance mechanisms. Its adaptive traits include the capacity for osmotic adjustment, selective ion uptake, and a deep root system. These characteristics enable it to maintain productivity under water and saline stress, positioning it as a strategic resource for livestock systems in ASS-affected areas, where climate variability and soil degradation limit agricultural options (Giorda & Ortiz, 2012; RAGT Semillas, 2025). In these environments, salinity and alkalinity often impair photosynthetic efficiency by restricting CO2 diffusion through the stomata and mesophyll, or by directly altering the photosynthetic metabolism (Chaves et al., 2009). These changes can induce secondary damage, such as oxidative stress, which negatively affects the structure and function of photosystem II (PSII) components (Flexas et al., 2004; Lawlor & Cornic, 2002). One of the most widely used techniques for assessing the structural and functional status of PSII is chlorophyll fluorescence kinetics, specifically OJIP analysis. This mathematical model provides critical information on the redox state of the PSII electron transport chain components, and the magnitude of OJIP parameters response is linked to the salinity and alkalinity stress tolerance of the evaluated plant hybrids (Kalaji et al., 2018; Stirbet & Govindjee, 2011). Based on the evidence that AS is a superior acidifying agent and that nutrient deficiency and photosynthetic damage are significant ASS constraints, we propose the following hypotheses: (1) AS is a more effective N fertilization strategy than urea for improving the nutritional status of sorghum plants in ASS soils, due to its superior soil acidification capacity and resultant enhancement of nutrient availability; and (2) Fertilizing sorghum grown in ASS soil with either AS or U will reduce damage to PSII electron transport chain components, as evaluated by OJIP analysis, with AS providing better protection due to improved plant nutritional status. Hence, this study aimed to: (i) evaluate whether ammonium sulfate or urea application can mitigate the nutrient deficiencies caused by ASS soils in sorghum hybrids under both field and greenhouse conditions; and (ii) determine the effects of AS and urea application in ASS soils on PSII activity of sorghum hybrids with different salinity-alkalinity tolerance, using OJIP analysis.
Materials and Methods
Study Site and Soil Characterization
The experiments were conducted in Villa Fontana, Córdoba, Argentina (30°52’26.9” S, 63°05’29.4” W, and 107 m above sea level). Two distinct soil types were utilized, sourced from fields situated 5 km apart: a control soil (slightly acidic) and a saline-sodic-alkaline (ASS) soil. Both soils shared a loamy-clayey texture. For the field experiment, the control soil exhibited a pH of 6.32, an electrical conductivity (EC) of 0.52 dS m-1, an organic matter content of 1.5%, a total carbon content of 1.01%, a total N content of 0.10%, available P content of 16.8 mg kg-1, and exchangeable Ca content of 10.24, Mg 2.37, K 1.54, Na 0.22 cmol(+) kg-1. The ASS soil, conversely, had a pH of 8.27, electrical conductivity of 8.84 dS m-1, an ESP of 28.8%, organic matter of 1.1%, total carbon of 0.64%, total N of 0.06%, available P of 6.8 mg kg-1, Ca 11.92, Mg 3.48, K 1.41, Na 6.76 cmol(+) kg-1. For the greenhouse experiment, the control and ASS soils had a pH values of 6.3 and 9.7, an electrical conductivities of 0.5 and 6.2 dS m-1, a cation exchange capacity of 1.4% and 31.8%, organic matter of 1.89% and 1.37%, total carbon of 1.10% and 0.79%, total N 0.109% and 0.084%, available P 11.3 and 16.9 mg kg-1, Ca 6.5 and 7.5, Mg 3.5 and 1.5, K 1.8 and 2.7, Na 0.2 and 5.4 cmol(+) kg-1, respectively. Detailed soil properties for both experiments are provided in Supplementary Tables S1 and S2.
Experimental Design and Treatments
The field experiment was conducted during the 2018/2019 growing season using a split-plot design with five replicates (blocks). The main plots consisted of the two soil types -a control and ASS- while subplots were created from the factorial combinations of three sorghum hybrids and three fertilizer treatments, totaling 90 subplots (2 soil types × 3 hybrids × 3 fertilizer treatments × 5 blocks). The three evaluated sorghum hybrids were INTA Peman Silero (SIP), Argensor 155 DP (155), and Argensil 163 D (163). The three fertilizer treatments included Ammonium sulfate (AS), Urea (U), and a non-fertilized control (Non-fert). Fertilizers were applied at a rate of 120 kg N ha-1, divided equally into two applications: 50% at planting and 50% at the V5 growth stage. Each plot measured 1.26 × 6.0 m, with three rows separated by 0.42 m, and had a final density of 15 plants m-2. Standard agronomic practices, including weed control and irrigation, were maintained. The greenhouse experiment was conducted during 2020/2021 using a completely randomized factorial design with five replicates per treatment (n = 5), resulting in 90 pots (2 soil types × 3 hybrids × 3 fertilizer treatments × 5 replicates). Plants were cultivated in 3-L pots under controlled conditions with an average temperature of 28°C and photosynthetically active radiation (PAR) of 1500 μmol m-2 s-1. Plants were watered three times a week to maintain field capacity at 0.30 cm³ water content.
Sampling Protocol
Plants were harvested 60 days after emergence (DAE) in the field experiment and 35 DAE in the greenhouse trial. The shorter period in the greenhouse was due to the spatial constraints of pot cultivation, which limit root development and overall plant growth compared with field conditions. The same sampling protocol was used in both experiments, where five plants were collected per treatment combination (n = 5). In the field experiment, samples were collected from the central row of each subplot across all five replicate blocks (total N = 90). The greenhouse experiment maintained an equivalent total sample size (N = 90). For the aerial plant sampling, shoots were harvested by cutting at 0.5 cm above the soil surface. Leaf samples used for nutrient analyses were collected from the same plants used for biomass determination. Soil samples for pH analysis were collected from each subplot at harvest. Subsamples of 1.0 g of soil were collected at a depth of 20 cm in the field and at 15 cm in 18 cm-high pots. Soil pH was determined at the base of each plant, along the sowing line, at a depth of 0-2 cm. Measurements were performed in a 1:2.5 soil: water suspension after 30 minutes of equilibration using a calibrated pH meter.
Measurements
Soil pH was measured at harvest in both experiments using the procedure described in the sampling protocol. For plant dry weight determination, harvested shoots were dried in a ventilated oven at 60°C until constant weight was reached, and aerial dry matter was determined for each sampled plant. Nutrient analysis was performed on specific leaf tissues to assess nutrient status due to different nutrient mobility accurately: younger leaves (apical) typically show higher concentrations of mobile nutrients (Ca, Mg, K), while older leaves (basal) better reflect the plant’s N and P status. These nutrients were also evaluated in the greenhouse plants using the same protocol as in the field experiment. Total N was determined by the Kjeldahl method, P by the colorimetric method using nitrovanadate-molybdate reagent, K was quantified by flame spectrophotometry, and Ca and Mg by atomic absorption spectrometry. This differential sampling reflected nutrient mobility: For the Photosystem II (PSII) component analysis, the chlorophyll-a fluorescence transient (ChlF) rate was measured on the newest fully expanded leaf at harvest (60 DAE in the field and 35 DAE in the greenhouse), using a Pocket-PEA fluorometer (Plant Efficiency Analyzer, Hansatech Instruments Ltd., King’s Lynn, Norfolk, UK). Before the ChlF measurements, the leaves were dark-adapted for at least 30 minutes using specific leaf clips to ensure complete oxidation of the reaction centers (RCs). An actinic 1-second light pulse of 3500 μmol photons m-2 s-1 was applied to reach maximum fluorescence emission (Fm or P step) at approximately 300 ms. The minimum fluorescence (Fo O step) was recorded at ~50 ms, and the intermediate steps, called J and I, were recorded at 2 ms and 30 ms, respectively. The OJIP parameters were calculated using Pocket-PEA manufacturing software (Strasser et al., 2004; Supplementary Table 3).
Statistical Analysis
Statistical comparisons were performed to evaluate differences among hybrids (genotypic variation), fertilizer treatments (AS and U), soil conditions (ASS vs. control), and their interactions. Soil pH, plant biomass, foliar nutrient concentrations, and PSII chlorophyll fluorescence data were analyzed in R (v3.6.2) using RStudio (v1.3.959) and the tidyverse package (v1.3.0). Data normality was assessed through the Shapiro-Wilk test and visual inspection of Q-Q plots. As the assumptions of normality were not met in most cases, even after applying logarithmic, square-root, and Box-Cox transformations, non-parametric tests were employed. The Kruskal-Wallis test was used to evaluate overall group differences, and the pairwise Wilcoxon rank-sum test was subsequently applied for post-hoc comparisons when significant effects were detected (p < 0.05).
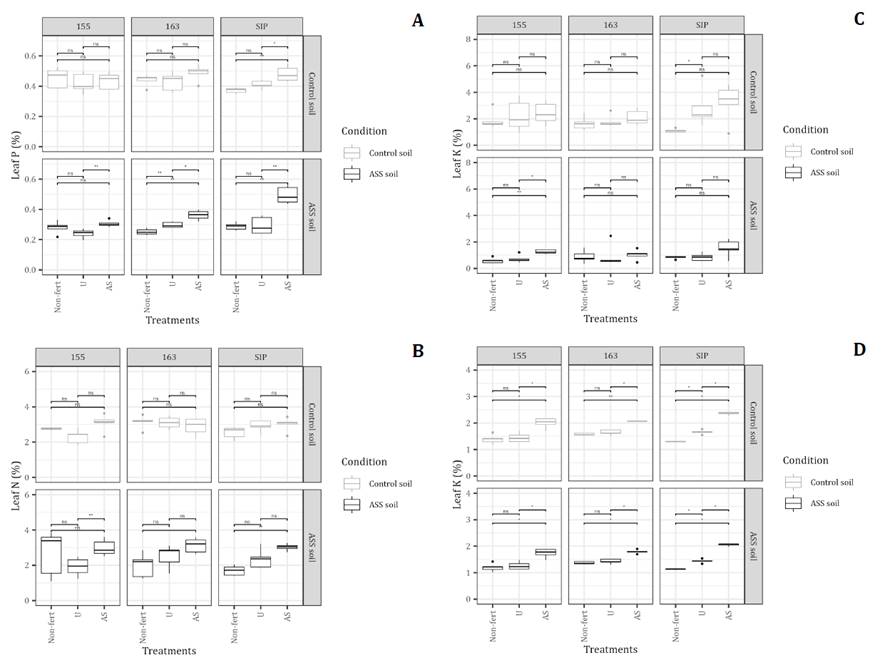
In this study, the chlorophyll fluorescence parameters derived from the OJIP test were not normalized using a standard procedure. Instead, data normalization was achieved by expressing the values relative to the corresponding control soil condition, a common approach to account for potential differences across hybrids and fertilizer treatments. Specifically, for each hybrid and fertilizer treatment combination, the mean value obtained under ASS soil conditions (x̄ASS, hybrid i, tratment j) was divided by the corresponding mean value measured in the control soil for the same hybrid × fertilizer combination (x̄Control, hybrid i, tratment j). This calculation yielded a dimensionless ratio that expresses the relative performance of plants grown in ASS soil compared with their counterparts under non-stress conditions (Equation 1).
Where x̄ represents the average of the valuated parameter for each sorghum hybrid (i) and N treatment (j).
Results
Soil pH and Plant Dry Weight
Soil pH
The two experiments demonstrated a consistent pattern regarding the effect of N source on soil pH across the two soil types. In the field experiment, the pH of the control soil (initially slightly acidic) remained stable, showing no significant differences across all sorghum hybrids and fertilizer treatments, with values ranging narrowly from 6.26 to 6.36 (figure 1A). In sharp contrast, ASS soil exhibited marked pH responses to fertilization. The AS application significantly reduced soil pH across all hybrids, from 8.16 (Non-fert) to 7.53, representing an approximately 0.6-unit decrease. Urea application did not affect ASS soil pH relative to the unfertilized control. However, the magnitude of AS-induced acidification varied among hybrids: in hybrid 155, all N treatments differed significantly from one another; while in hybrids 163 and SIP, AS achieved substantially lower pH values than both U and Non-fert treatments, which themselves did not statistically differ.
Data represent mean ± standard error (n = 5). Asterisks indicate significant differences between treatments within each hybrid-soil combination: ns = p > 0.05, * = p ≤ 0.05, ** = p ≤ 0.01, *** = p ≤ 0.001. Black dots represent outliers.
Los datos representan medias ± error estándar (n = 5). Los asteriscos indican diferencias significativas entre tratamientos dentro de cada combinación híbrido-suelo: ns = p > 0,05, * = p ≤ 0,05, ** = p ≤ 0,01, *** = p ≤ 0,001. Los puntos negros representan valores atípicos.
Figure 1. Soil pH in control and alkaline-saline-sodic (ASS) soils under a) field and b) greenhouse conditions for three sorghum hybrids (155, 163, SIP) fertilized with ammonium sulfate (AS), urea (U), or unfertilized (Non-fert), with n = 5.
Figura 1. pH del suelo en suelos control y alcalino-salino-sódicos (ASS) bajo condiciones de a) campo y b) invernadero para tres híbridos de sorgo (155, 163, SIP) fertilizados con sulfato de amonio (AS), urea (U) o sin fertilizar (Non-fert).
Similar patterns were observed in the greenhouse experiment (figure 1B). The pH in the control soil remained stable across all hybrids and treatments. In ASS soil, AS application led to a more pronounced reduction, dropping substantially from 9.54 (Non-fert) to 8.52, an approximate 1-unit decrease. Consistent with the field trial, the U application showed no significant acidifying effect compared to the unfertilized control, with both treatments maintaining high pH levels around 9.5 in the ASS soil.
Plant Dry Weight
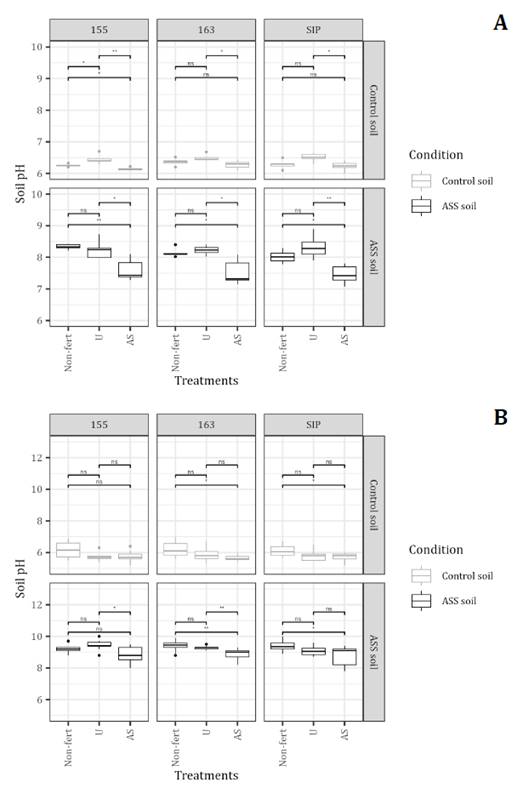
ASS soils negatively affected sorghum growth in both field and greenhouse experiments. In the field experiment, the impact of N fertilization on biomass production in the ASS soil varied depending on the N source, hybrid, and soil type. In hybrid 155, which was identified as the most sensitive, AS treatment yielded significantly lower biomass than the U treatment under ASS conditions (62% less than the control soil), and both treatments produced biomass significantly lower than the Non-fert treatment (figure 2A). In contrast, in the more tolerant hybrids (163 and SIP), AS treatment significantly improved sorghum growth in ASS soils relative to both U and Non-fert treatments, achieving dry weight levels comparable to those observed in control soils. In these tolerant hybrids, AS treatment significantly increased biomass compared to both U and Non-fert treatments in ASS soil. Conversely, the U application did not produce significant changes in dry weight relative to non-fertilized controls in either ASS soil or control soil.
Asterisks indicate significant differences between treatments within each hybridsoil combination: ns = p > 0.05, * = p ≤ 0.05, ** = p ≤ 0.01, *** = p ≤ 0.001. Black dots represent outliers. Data represent mean ± standard error (n = 5).
Los asteriscos indican diferencias significativas entre tratamientos dentro de cada combinación híbrido-suelo: ns = p > 0,05, * = p ≤ 0,05, ** = p ≤ 0,01, *** = p ≤ 0,001. Los puntos negros representan valores atípicos. Los datos representan medias ± error estándar (n = 5).
Figure 2. Fertilization effect (AS, U, Non-fert) on plant dry weight of three sorghum hybrids (155, 163, SIP) in control and ASS soils under a) field and b) greenhouse conditions.
Figure 2. Efecto de la fertilización (AS, U, sin fertilizar) sobre el peso seco de tres híbridos de sorgo (155, 163, SIP) en suelos testigo y ASS, bajo condiciones de a) campo y b) invernadero.
In the greenhouse experiment, fertilization treatments in the control soil produced no significant differences in dry weight across hybrids or N sources (figure 2B). However, in ASS soil, AS significantly increased dry weight compared to both U and Non-fert treatments, with improvements ranging from 2- to 5-fold depending on the hybrid. Consistent with field observations, U fertilization showed no significant effect on plant dry weight compared to Non-fert controls in ASS soil.
Leaf Nutrient Concentration
Field Experiment Nutrient Status
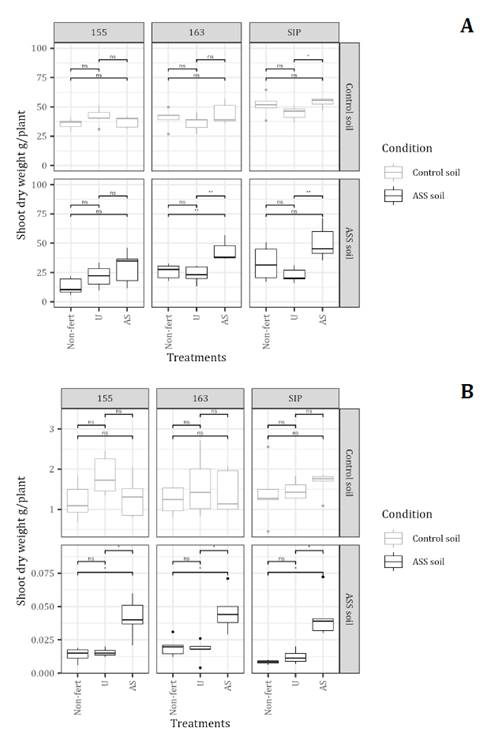
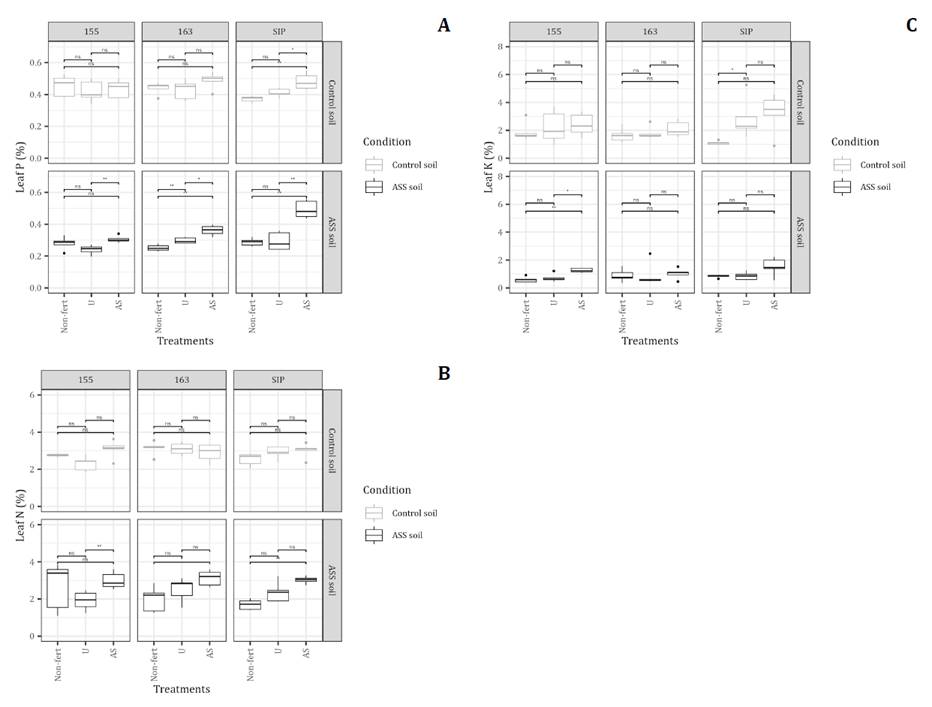
Sorghum plants grown in the control soil exhibited a 55% higher concentration of foliar P than those grown in unfertilized ASS soil. The most significant beneficial effect of AS fertilization in ASS soils was observed in the hybrid SIP, which showed P concentrations comparable to those of plants grown in the control soil and fertilized with AS in ASS soils (0.48% and 0.49%, respectively). In contrast, the lowest foliar P concentrations were recorded in plants grown in unfertilized control soils (0.37%) and in U-fertilized ASS soils (0.30%). For N concentration, significant differences were detected between soil conditions (C, p ≤ 0.05) and fertilizer treatments (T, p ≤ 0.05), with a significant C×T interaction (p ≤ 0.05). This indicates that the effect of fertilization depends on the soil condition.
In the control soil, neither AS nor U fertilization significantly affected leaf N concentration in any of the hybrids. In contrast, in ASS soil, AS fertilization resulted in foliar N concentrations that differed substantially from U and Non-fert. In hybrids 163 and SIP, the values were 56.5% and 78% higher, respectively; while in 155, fertilization with AS did not differ significantly from the Non-Fert treatment. Across soil conditions, U-fertilization had no significant impact on N concentration in any of the hybrids. Regarding Ca concentration, significant differences were observed among hybrids, soil conditions, fertilizer treatments, and the hybrid-by-condition (H x C) interaction (p ≤ 0.05). Generally, Ca concentration was significantly higher in the control soil across all treatments, except for the AS treatment in ASS soil, which had a Ca concentration similar to that of the control soil. In ASS soil, the highest values were observed for SIP and 163 hybrids (0.88% and 0.81%, respectively; (figure 3). Finally, significant differences in K concentration were found among hybrids (p ≤ 0.05), conditions (p ≤ 0.05), treatments (p ≤ 0.05), and the hybrid-by-treatment (H×T) interaction (p ≤ 0.05). The lowest K concentration was found in hybrids 155 and SIP, grown in ASS soil under the Non-fert and U treatments (~1.15%). In comparison, the highest K values were consistently recorded in plants fertilized with AS, regardless of soil type and hybrid (>1.8%).
Concentrations were determined in sorghum leaves:a) P, b)N, c)Ca, and d) K . The P, N, and K were analyzed in basal leaves, and Ca in apical leaves, at 60 DAE. Asterisks indicate significant differences between treatments within each hybrid-soil combination: ns = p> 0.05, * = p≤ 0.05, ** = p≤ 0.01, *** = p≤ 0.001.The black dots represent outliers.
Se determinaron las concentraciones en las hojas de sorgo: a) P, b) N, c) Ca y d) K. El P, el N y el K se analizaron en las hojas basales, y el Ca en las hojas apicales, a los 60 días después de la siembra (DAE). Los asteriscos indican diferencias significativas entre los tratamientos dentro de cada combinación de híbrido y suelo: ns = p > 0,05, * = p ≤ 0,05, ** = p ≤ 0,01, *** = p ≤ 0,001. Los puntos negros representan valores atípicos.
Figure 3. Mineral composition (%) of sorghum leaves (Hybrids 155, SIP, and 163) under field experimental conditions, with treatments (Non-fertilized, Urea, and Ammonium Sulfate.
Figura 3. Composición mineral (%) de las hojas de sorgo (híbridos 155, SIP y 163) en condiciones experimentales de campo, con tratamientos (sin fertilizar, urea y sulfato de amonio).
Greenhouse Experiment Nutrient Status
In the greenhouse trial, significant differences in sorghum leaf Ca concentration were observed between conditions (C, p ≤ 0.05), treatments (T) (p ≤ 0.05), and C×T interaction (p ≤ 0.05) (figure 4, page 11-12). Specifically, plants in unfertilized ASS had a 62.3% lower Ca concentration than those in the control soil. Furthermore, AS treatment resulted in a Ca concentration that differed significantly from both the U and Non-fert treatments in ASS soil. For Mg concentration, significant differences were found between conditions (p ≤ 0.05) and treatments (p ≤ 0.05). Plants grown in unfertilized ASS soil had a significantly lower Mg concentration than those in the control soil. The AS treatment resulted in Mg concentration that differed considerably from the U and non-fertilized treatments, with the most notable difference observed in the SIP hybrid, which showed a 147% increase compared to the unfertilized ASS treatment. Finally, for K concentration, significant differences were observed only between conditions (p ≤ 0.05). The effect of ASS on K concentration differed between greenhouse and field conditions. While the AS treatment resulted in higher K concentrations than the U and non-fertilized treatments, these differences were less pronounced than those observed in the field. No significant differences in K concentration were found among the three hybrids (figure 4).
Treatments were Nonfertilized (C), Urea (U), and Ammonium Sulfate (AS). Asterisks indicate significant differences between treatments within each hybrid-soil combination: ns = p > 0.05, * = p ≤ 0.05, ** = p ≤ 0.01, *** = p ≤ 0.001. Black dots represent outliers.
Los tratamientos fueron sin fertilizar (C), urea (U) y sulfato de amonio (AS). Los asteriscos indican diferencias significativas entre tratamientos dentro de cada combinación híbrido-suelo: ns = p > 0,05, * = p ≤ 0,05, ** = p ≤ 0,01, *** = p ≤ 0,001. Los puntos negros representan valores atípicos.
Figure 4. Mineral composition (%) of sorghum leaves from hybrids 155, SIP, and 163 grown under greenhouse conditions in control and ASS soils: a) Ca, b) Mg, and c) K.
Figura 4. Composición mineral (%) de las hojas de sorgo de los híbridos 155, SIP y 163 cultivados en condiciones de invernadero en suelos de control y ASS: a) Ca, b) Mg y c) K.
Photosystem II component (PSII)
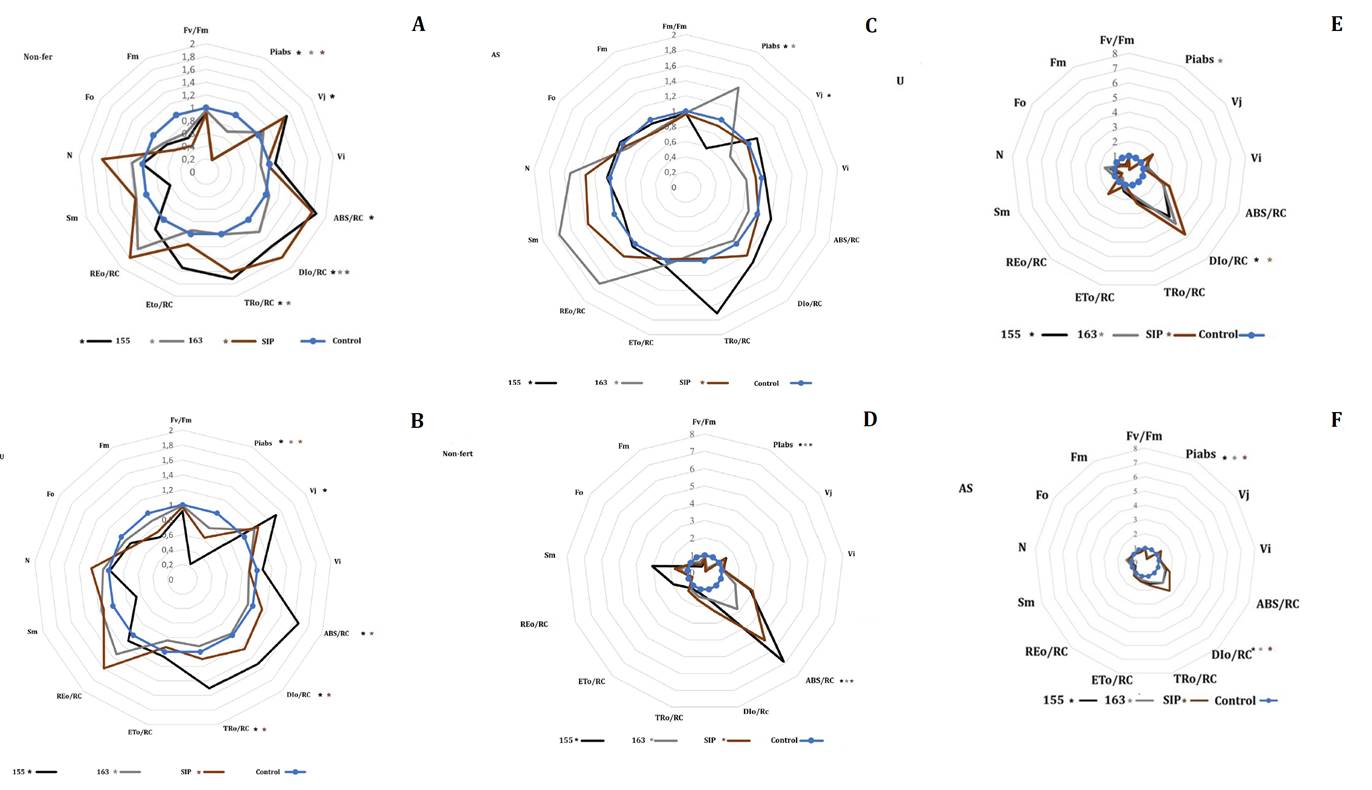
Chlorophyll fluorescence parameters were significantly affected by ASS soil at 60 DAE in the field, with responses varying with hybrid tolerance and treatment relative to plants grown in control soil (figure 5).
Data show fertilization effects (A-C: field; D-F: greenhouse). Values are normalized to the control soil mean for each genotype (blue line). Measurements were performed on fully expanded leaves at 60 DAE in the field and 35 DAE in the greenhouse. Group differences were evaluated using the Kruskal-Wallis test, followed by pairwise Wilcoxon tests when significant effects (*) were detected (p < 0.05).
Los datos muestran los efectos de la fertilización (A-C: campo; D-F: invernadero). Los valores están normalizados respecto a la media del suelo control de cada genotipo (línea azul). Las mediciones se realizaron en hojas completamente expandidas a los 60 DAE en campo y a los 35 DAE en invernadero. Las diferencias entre grupos se evaluaron mediante la prueba de Kruskal-Wallis, seguida de pruebas pareadas de Wilcoxon cuando se detectaron efectos significativos (*) (p < 0,05).
Figure 5. Radar plots of chlorophyll fluorescence (OJIP test) in sorghum hybrids (155: black, 163: light gray, SIP: brown) grown in ASS soil.
Figura 5. Gráficos de radar de fluorescencia de clorofila (test OJIP) de híbridos.
Hybrid 155 was the most sensitive. PIabs decreased by 80% under both Non-fert (p = 0.0008) and U (p = 0.001) treatments, and by 43% with AS (p = 0.0004) (figures 5A-C). The Fo and Fm were not statistically significant; as a result, Fv/Fm remained unchanged. In hybrid 155, ABS/RC, TRo/RC, and DIo/RC increased by 83.7% (p = 0.0012), 71.7% (p = 0.001), and 54.9% (p = 0.0012), respectively, under the Non-fert treatment. In U-treated plants, increases of 65% (p = 0.0012), 50% (p = 0.001), and 51% (p = 0.0011) were observed. In contrast, the changes observed under AS treatment were smaller and not statistically significant (20%, 71%, and 32%, respectively). In addition, Vj increased by 52.9% (p = 0.0043) under Non-fert and by 51.4% (p = 0.0012) with U, while only a 12% increase was detected with AS (p = 0.0018).In hybrid SIP, PIabs decreased by 79.8% (p = 0.0008) under Non-fert, and by 31.8% (p = 0.0011) with U. ABS/RC, DIo/RC, and TRo/RC increased by 77.1% (p = 0.0005), 78.6% (p = 0.0004), and 61.3% (p = 0.0006), respectively, in the Non-fert treatment compared to the control. These increases were slightly reduced with U, while AS showed no significant effect. In hybrid 163, the impact of ASS soil was milder. PIabs decreased by 29% (p = 0.0001) under Non-fert and by 27% (p = 0.0001) with U, whereas AS significantly improved PIabs by 47%, indicating a mitigating effect. Under greenhouse conditions, fluorescence alterations were more pronounced (figures 5D-F), especially in hybrid 155. DIo/RC increased by 586% in Non-fert 155, 421% in SIP, and 181% in 163 (p = 0.01 in all cases). The U treatment induced 317% increases in 155 (p = 0.0004) and 476.2% in SIP (p = 0.0009). The AS application reduces DIo/RC to 92.7% in hybrids 155 and 163, and to 167% in SIP. In Non-fert plants, PIabs dropped sharply by 86% (p = 0.0004) in hybrid 155, 73.8% (p = 0.0001) in 163, and 92.9% (p = 0.0005) in SIP. The U treatment did not prevent PIabs decreases. However, AS significantly reduced PIabs losses to 62.4% in 155, 65.6% in 163, and 75% in SIP.
Discussion
Soil Acidification and its Effect on Nutrient Availability
The AS application reduced ASS soil pH in both the field and greenhouse experiments (figures 1A and 1B), effectively enhancing macronutrient availability as confirmed by leaf analysis (figure 3 and figure 4). Unlike the U, AS improved nutrient uptake, although physiological responses varied with hybrid tolerance. These results align with Fageria et al. (2010), who observed similar beneficial effects with split AS applications. In the greenhouse experiment, the application of AS significantly reduced soil pH in the ASS soil. However, the reduction was almost half that observed in the field experiment, probably because in pots the roots grow less and, consequently, soil acidification is less. Regardless of the experimental conditions, U fertilization did not significantly change soil pH in either control or ASS soils. Upon dissolution in water, urea undergoes urease-catalyzed hydrolysis to form ammonium and bicarbonate ions, initially increasing soil pH near the application zone. Subsequently, as nitrification proceeds, the pH in the retention zone gradually decreases. In this study, soil pH was measured 30 days after fertilization in field and greenhouse experiments. Given this relatively short time interval between fertilizer application and pH measurement, the nitrification process may not have fully progressed, limiting the extent of observed pH changes (Kissel et al., 2008). These results largely supported our first hypothesis, that the application of AS significantly reduced soil pH in both field and greenhouse experiments, particularly in ASS soil. This reduction in pH was associated with increased nutrient availability, as indicated by higher nutrient concentrations in the sorghum leaves. However, the magnitude of the pH reduction was greater in the field compared to the greenhouse, likely due to differences in root system development, soil volume, and soil buffering capacity. Although U did not significantly affect soil pH, it is important to note that nitrification can reduce soil pH over time. The relatively short duration of the greenhouse experiment may have limited the extent of pH changes observed with U fertilization.
Nitrogen Dynamics and Uptake in ASS Soils
High soil pH generally restricts the availability of essential macro- and microelements for crop growth, particularly N, P, K, Mg, Ca, Fe, and Zn, which is consistent with our findings. In this study, we confirmed that the application of AS was superior to U in enhancing sorghum nutrient absorption in most of the tested hybrids (figures 3 and figure 4). The greater acidifying effect of AS is the primary explanation for this (Jacoby et al., 2017). The initial N content in the ASS soil was lower (0.06%) than in the control soil, probably due to the low organic matter content (1.1% vs 1.7%) (Supplementary Table 1).
Sorghum plants grown in the AAS soil showed significantly lower N concentration in their leaves, especially in the SIP hybrid (figure 3). This was expected due to the inhibition of N mineralization and soil enzymatic activities, which are essential for organic matter decomposition, by both high salinity and alkalinity. It is also likely that the lower N content in the leaves of U-fertilized sorghum plants compared to those fertilized with AS could be associated with the inactivation of urease enzymes, either by excess carbonate/bicarbonate, and/or high electrical conductivity (EC) (Sharma et al., 2023). Optimal nitrification occurs at pH 8.5. Therefore, a reduction in soil pH, as occurred in the AS treatment, may have helped maintain the NO3-/NH4+ balance, which is advantageous for sorghum growth. In addition, the increased availability of NH4+ (Bernardo et al., 1984) may have increased plant tolerance to high soil EC by improving their Na/K homeostasis in ASS soil (de Souza Miranda et al., 2016).
Phosphorus Availability and Uptake Mechanisms
In general, the three sorghum hybrids grown in the ASS soil field showed lower P concentrations in their leaves (figure 3). The solubility of P decreases as soil pH increases above ~6.5, either due to adsorption on CaCO3 surfaces or precipitation as a function of soil P content. Our study revealed that fertilization with AS more efficiently solubilized and mobilized P than U, as sorghum plants grown in ASS soil showed P values like those of plants grown in control soil (Clark & Baligar, 2000) (figure 3). This finding is consistent with those sudies showing that lowering soil pH by 1.6 units through ammonium fertilization improves P mobilization in ryegrass seedlings (Gahoonia et al., 1992). Similarly, Blair et al. (1970) demonstrated that pH changes at the soil-root interface caused by AS fertilization in alkaline soil altered the H2PO4- /HPO4 2- ratio on the root surface in maize, increasing the abundance of the H2PO4 form and consequently improving P uptake by the roots.
Secondary Macronutrients: Calcium, Magnesium, and Potassium Dynamics
Ca availability is reduced under salinity and even more so in alkaline-saline-sodic (ASS) soils, where excess Na can hinder Ca uptake. Our results confirm the negative impact of ASS soil on foliar Ca in sorghum and the partial effectiveness of AS and U in improving its solubilization (Hadi & Karimi, 2012; Saqib et al., 2020; figure 3 and figure 4). Mg concentration was also significantly lower in plants grown in ASS soil (Ranaweera & De Silva, 2021). AS improved Mg uptake under greenhouse conditions (figure 4), but overall, greenhouse plants absorbed less Mg than field-grown ones. This may be related to the developmental stage, the higher pH in greenhouse ASS soil (9.7), or antagonism with Ca and K (Bontpart et al., 2024; Cavalcante et al., 2018; Jacoby et al., 2017). Sorghum plants grown in ASS soil, both in the field and greenhouses, showed lower K absorption compared to those grown in control soil (figure 3 and figure 4). Saline-alkaline soils, such as ASS, have high concentrations of Ca2+ and Mg2+, which limit K+ uptake by competing for absorption sites on root surfaces. Consequently, sorghum plants in the ASS soil may have experienced K deficiency even though chemical analyses of this soil showed sufficient K for sorghum growth (Supplementary Table 2). Additionally, a possible antagonistic effect between K and Na may have occurred due to the high K concentration in the studied soil. Finally, lower water uptake and, consequently, lower K+ uptake may have resulted in a lower K+ concentration in sorghum leaves in ASS soil (Wakeel, 2013). On the other hand, both U and AS fertilization favored the solubilization, mobilization, and absorption of K+ by sorghum plants in the ASS soil (figure 3 and figure 4). This response could be governed by pH, as decreasing it allows H+ to exert stronger competition with exchangeable K+ at soil exchange sites, thereby shifting it towards the soil solution (Naeem et al., 2023).
Photosynthetic Responses and Photosystem II Functionality
Our data revealed that one of the main limitations for sorghum plants grown in ASS soils was low nitrogen availability, although both soils, ASS and control, initially had equivalent N percentages (figure 3 and figure 4). The condition of the ASS soil significantly affected chlorophyll concentration in sorghum leaves in both the field and the greenhouse. This suggests that low N uptake from plants grown in ASS soils reduced chlorophyll biosynthesis in sorghum leaves. Consequently, the ASS condition affected the operation of the components of the PSII electron transport chain. Due to the low N level in the leaves, reduced synthesis of key photosynthetic enzymes, particularly Rubisco, may have led to an imbalance between PSII light absorption and electron transport in the Calvin cycle.
The extent to which these physiological variables were affected varied with the hybrid’s tolerance to ASS stress. The differences between hybrids can be attributed to their ability to maintain their photosynthetic structure under such conditions, thereby enabling them to regulate the electron transport system. (Liu et al., 2024; Sui et al., 2015; Suzuki et al., 2023).
The superior effectiveness of AS over U in overcoming the restrictions imposed by the ASS soil was also evidenced by the higher chlorophyll concentration in the leaves, which was associated with increased N uptake. This response appears to depend on sorghum’s genetic background with respect to N use efficiency, given the discrepancy in N concentration among these hybrids. For example, ASS soil condition affected most of the PSII components of the most sensitive sorghum hybrid (155), such as the photosynthetic index PI (Alkhader et al., 2019) (also known as the vitality index), which expresses the three main functional steps of PSII photosynthetic activity. However, the values of PIabs in the AS treatment were like those of the control in the SIP hybrid or even higher than those of the control in the hybrid 163. (Strasser et al., 2004). The ASS-sensitive hybrid (155) and, to a lesser degree, the moderately tolerant SIP, exhibited significant alterations in the PSII antenna (ABS/RC), indicating loss of LHC integrity and changes in the chlorophyll a/b ratio, which were associated with increased Reactive Oxygen Species (ROS) generation, lipid peroxidation, and chlorophyll degradation (Bordenave et al., 2019). AS fertilization alleviated these effects, preserving PSII, PSI, and cytochrome b6/f activity and maintaining electron transport within the thylakoids, as evidenced by increased Vj fluorescence and more efficient QA reduction (Kalaji et al., 2018; Sachdev et al., 2021; Zhang et al., 2018). Compared to U fertilization, AS proved more effective in the tolerant hybrids (SIP and 163) by acidifying the rhizosphere, enhancing N, P, and Fe availability, and sustaining higher photosynthetic efficiency. These findings support our second hypothesis that AS fertilization reduces damage to PSII electron transport chain components in sorghum grown in ASS soil, with AS providing superior protection due to improved plant nutritional status. Overall, these results underscore the substantial potential of AS fertilization to optimize sorghum performance in saline-alkaline environments.
Conclusion
This study demonstrates that alkaline-saline-sodic (ASS) soils in the semi-arid region of Córdoba, Argentina, severely constrain sorghum growth by reducing biomass accumulation and impairing photosynthetic efficiency. Among the evaluated treatments, ammonium sulfate (AS) fertilization proved the most effective strategy at mitigating these effects by acidifying soil and improving nutrient availability. The AS application significantly lowered soil pH under both field and greenhouse conditions and increased foliar P content. Hybrids SIP and 163 exhibited greater physiological and agronomic resilience under ASS stress, showing improved Photosystem II performance and higher foliar nutrient content with AS fertilization, whereas hybrid 155 was comparatively more sensitive. These findings highlight the importance of combining stress-tolerant genotypes with appropriate N sources to sustain productivity in degraded soils. Replacing conventional urea with split applications of AS at approximately 120 kg N ha-1 is recommended for ASS soils, as this practice restores sorghum yield potential and enhances forage nutritional quality by increasing N and P content. Overall, this research provides science-based, locally adapted management strategies to optimize sorghum performance and promote resilient, sustainable production systems in semi-arid regions affected by alkaline-saline-sodic conditions.
Adams, F. (1984). Crop response to lime in the southern United States. In F. Adams (Ed.). Soil acidity and liming, 12, 211-265. American Society of Agronomy. https://doi.org/10.2134/agronmonogr12.2ed.c5
Alkhader, A. M., Qaryouti, M. M., & Okasheh, T. Y. M. (2019). Effect of nitrogen on yield, quality, and irrigation water use efficiency of drip fertigated grafted watermelon (Citrullus lanatus) grown on a calcareous soil. Journal of Plant Nutrition, 42(1), 1-12. https://doi.org/10.1080/01904167.2019.1568464
Bernardo, L. M., Clark, R. B., & Maranville, J. W. (1984). Nitrate/ammonium ratio effects on mineral element uptake by sorghum. Journal of Plant Nutrition, 7(10), 1401-1414. https://doi.org/10.1080/01904168409363290
Blair, G. J., Miller, M. H., & Mitchell, W. A. (1970). Nitrate and ammonium as sources of nitrogen for corn and their influence on the uptake of other ions. Agronomy Journal, 62(4), 530-532. https://doi.org/10.2134/agronj1970.00021962006200040031x
Bontpart, T., Weiss, A., Vile, D., Gérard, F., Lacombe, B., Reichheld, J. P., & Mari, S. (2024). Growing on calcareous soils and facing climate change. Trends in Plant Science. https://doi.org/10.1016/j.tplants.2024.03.013
Bordenave, C. D., Rocco, R., Maiale, S. J., Campestre, M. P., Ruiz, O. A., Rodríguez, A. A., & Menéndez, A. B. (2019). Chlorophyll a fluorescence analysis reveals divergent photosystem II responses to saline, alkaline, and saline-alkaline stresses in the two Lotus japonicus model ecotypes MG20 and Gifu-129. Acta Physiologiae Plantarum, 41(9), 167. https://doi.org/10.1007/s11738-019-2956-0
Cavalcante, T. J., Castoldi, G., Rodrigues, C. R., Nogueira, M. M., & Albert, A. M. (2018). Marcha de absorção de macro- e micronutrientes na biomassa de sorgo. Pesquisa Agropecuária Tropical, 48(4), 364-373. https://doi.org/10.1590/1983-40632018v4851874
Chaves, M. M., Flexas, J., & Pinheiro, C. (2009). Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Annals of Botany, 103(4), 551-560. https://doi.org/10.1093/aob/mcn125
Chien, S. H., Gearhart, M. M., & Villagarcía, S. (2011). Comparison of ammonium sulfate with other nitrogen and sulfur fertilizers in increasing crop production and minimizing environmental impact: A review. Soil Science, 176(7), 327-335. https://doi.org/10.1097/SS.0b013e31821f0816
Clark, R. B., & Baligar, V. C. (2000). Acidic and alkaline soil constraints on plant mineral nutrition. Plant- Environment Interactions, 133–177.
De Souza Miranda, R., Gomes-Filho, E., Prisco, J. T., & Alvarez-Pizarro, J. C. (2016). Ammonium improves tolerance to salinity stress in Sorghum bicolor plants. Plant Growth Regulation, 78(1), 121-131. https://doi.org/10.1007/s10725-015-0079-1
Fageria, N. K., dos Santos, A. B., & Moraes, M. F. (2010). Influence of urea and ammonium sulfate on soil acidity indices and lowland rice production. Communications in Soil Science and Plant Analysis, 41(13), 1565-1575. https://doi.org/10.1080/00103624.2010.485237
Flexas, J., Bota, J., Loreto, F., Cornic, G., & Sharkey, T. D. (2004). Diffusive and metabolic limitations to photosynthesis under drought and salinity in C3 plants. Plant Biology, 6(3), 269-279. https://doi.org/10.1055/s-2004-820867
Gahoonia, T. S., Claassen, N., & Jungk, A. (1992). Mobilization of phosphate in different soils by ryegrass supplied with ammonium or nitrate. Plant and Soil, 140(2), 241-248. https://doi.org/10.1007/BF00010600
Giorda, L., & Ortiz, S. (2012). El cultivo de sorgo en la región semiárida central de Córdoba: aportes al sistema de producción ganadera. INTA Manfredi.
González, F. J., Martínez, A. L., & Pérez, R. M. (2019). Efecto del yeso agrícola en la mejora de los suelos salinos y en el rendimiento de los cultivos. Revista de Ciencia y Tecnología Agrícola, 12(2), 85-94. https://doi.org/10.1234/rcta.2019.12.2.85
Hadi, M. R., & Karimi, N. (2012). El papel del calcio en la tolerancia a la sal de las plantas. Revista de Nutrición Vegetal, 35(6), 905-927. https://doi.org/10.1080/01904167.2012.717158
Jacoby, R., Peukert, M., Succurro, A., Koprivova, A., & Kopriva, S. (2017). The role of soil microorganisms in plant mineral nutrition-current knowledge and future directions. Frontiers in Plant Science, 8. https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2017.01617
Kalaji, H. M., Bąba, W., Gediga, K., Goltsev, V., Samborska, I. A., Cetner, M. D., Dimitrova, S., Piszcz, U., Bielecki, K., Karmowska, K., Dankov, K., & Kompała-Bąba, A. (2018). Chlorophyll fluorescence as a tool for nutrient status identification in rapeseed plants. Photosynthesis Research, 136(3), 329-343. https://doi.org/10.1007/s11120-017-0467-7
Kissel, D. E., Cabrera, M. L., & Paramasivam, S. (2008). Ammonium, ammonia, and urea reactions in soils. In J. S. Schepers & W. R. Raun (Eds.). Nitrogen in agricultural systems (p. 101-155). American Society of Agronomy. https://doi.org/10.2134/agronmonogr49.c4
Lawlor, D. W., & Cornic, G. (2002). Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant, Cell & Environment, 25(2), 275-294. https://doi.org/10.1046/j.0016-8025.2001.00814.x
Liu, C., Gu, W., Li, B., Feng, Y., Liu, C., Shi, X., & Zhou, Y. (2024). Screening key sorghum germplasms for low-nitrogen tolerance at the seedling stage and identifying from the carbon and nitrogen metabolism. Frontiers in Plant Science, 15. https://doi.org/10.3389/fpls.2024.1340509
Marschner, P., & Rengel, Z. (2012). Nutrient availability in soils. In P. Marschner (Ed.). Marschner’s mineral nutrition of higher plants (3rd ed., p. 315-330). Academic Press. https://doi.org/10.1016/B978-0-12-384905-2.00012-1
Naeem, A., Deppermann, P., & Mühling, K. H. (2023). Ammonium fertilization enhances nutrient uptake, specifically manganese and zinc, and growth of maize in unlimed and limed acidic sandy soil. Nitrogen, 4(2), 239-252. https://doi.org/10.3390/nitrogen4020017
Pierre, W. H. (1928). Nitrogenous fertilizers and soil acidity: I. Effect of various nitrogenous fertilizers on soil reaction. Agronomy Journal, 20(3), 254-269. https://doi.org/10.2134/agronj1928.00021962002000030006x
RAGT Semillas Argentina, (2025, febrero 24). Sorgo, una herramienta versátil para la conversión de reservas forrajeras. RAGT Semillas Argentina. https://ragt-semillas.com.ar/sorgounaherramienta-versátil-para-la-conversión-de-reservas-forrajeras
Ranaweera, Y., & De Silva, R. C. L. (2021). A laboratory study on the effects of fertilizer addition on mobilization, percolation, and leaching of calcium ions in soils of the Anuradhapura district. International Conference on Applied and Pure Sciences.
Rengasamy, P. (2010). Soil processes affecting crop production in salt-affected soils. Functional Plant Biology, 37(7), 613-620. https://doi.org/10.1071/FP09249
Sachdev, S., Ansari, S. A., Ansari, M. I., Fujita, M., & Hasanuzzaman, M. (2021). Abiotic stress and reactive oxygen species: Generation, signaling, and defense mechanisms. Antioxidants,10(2). https://doi.org/10.3390/antiox10020277
Saqib, M., Abbas, G., & Akhtar, J. (2020). Root-mediated acidification and resistance to low calcium improve wheat (Triticum aestivum) performance in saline-sodic conditions. Plant Physiology and Biochemistry, 156, 201-208. https://doi.org/10.1016/j.plaphy.2020.09.001
Sharma, S., Gupta, N., Chakkal, A. S., Sharma, N., Alamri, S., Siddiqui, M. H., & Haider, F. U. (2023). Changes in enzyme activities in salt-affected soils during the incubation study of diverse particle sizes of rice straw. Agriculture, 13(9), Article 1694. https://doi.org/10.3390/agriculture13091694
Sparks, D. L. (2003). The chemistry of saline and sodic soils. In D. L. Sparks (Ed.), Environmental soil chemistry (2nd ed., p. 285-300). Academic Press. https://doi.org/10.1016/B978-012656446-4/50010-4
Stirbet, A., & Govindjee, G. (2011). On the relation between the Kautsky effect (chlorophyll a fluorescence induction) and Photosystem II: Basics and applications of the OJIP fluorescence transient. Journal of Photochemistry and Photobiology B: Biology, 104(1-2), 236-257. https://doi.org/10.1016/j.jphotobiol.2010.12.010
Strasser, R. J., Tsimilli-Michael, M., & Srivastava, A. (2004). Analysis of the chlorophyll a fluorescence transient. In G. C. Papageorgiou & Govindjee (Eds.). Chlorophyll a fluorescence: A signatura of photosynthesis (p. 321-362). Springer Netherlands. https://doi.org/10.1007/978-1-4020-3218-9_12
Sui, N., Yang, Z., Liu, M., & Wang, B. (2015). Identification and transcriptomic profiling of genes involved in increasing sugar content during salt stress in sweet sorghum leaves. BMC Genomics, 16(1), 534. https://doi.org/10.1186/s12864-015-1760-5
Suzuki, Y., Ohsaki, K., Takahashi, Y., Wada, S., Miyake, C., & Makino, A. (2023). The behavior of photosystems II and I is modulated by N partitioning to Rubisco in mature leaves acclimated to low N levels and in senescent leaves in rice. Plant and Cell Physiology, 64(1), 55-63. https://doi.org/10.1093/pcp/pcac139
Tupa-Andrade, L.; Caro-Castro, J.; León-Quispe, J. (2025). Isolation of Polyhydroxyalkanoate (PHA)- Producing Azotobacter spp. from Crop Rhizospheres Located in Lima, Peru. Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina. 57(2): 45-56. DOI: https://doi.org/10.48162/rev.39.170.
Wakeel, A. (2013). Potassium–sodium interactions in soil and plant under saline-sodic conditions. Journal of Plant Nutrition and Soil Science, 176(3), 344-354. https://doi.org/10.1002/jpln.201200417
Zhang, H., Xu, N., Wu, X., Wang, J., Ma, S., Li, X., & Sun, G. (2018). Effects of four types of sodium salt stress on plant growth and photosynthetic apparatus in sorghum leaves. Journal of Plant Interactions, 13(1), 506-513. https://doi.org/10.1080/17429145.2018.1526978.
https://drive.google.com/file/d/11htDuZXPbt9TSFc0g_jkY7wtjwsBS0cq/view?usp=drive_link