Revista de la Facultad de Ciencias
Agrarias. Universidad Nacional de Cuyo. En prensa. ISSN (en línea) 1853-8665.
Original article
Physicochemical
Properties, Bioactive Compounds, and Antioxidant Activity of Andean Fruits:
Optimization of Extraction by Response Surface Methodology
Propiedades
fisicoquímicas, compuestos bioactivos y actividad antioxidante de frutas
andinas: optimización de la extracción mediante metodología de superficie de
respuesta
Vicente Amirpasha
Tirado-Kulieva1*,
Jimy Oblitas2,
Noemí León-Roque3,
Wilson Castro4
1Asociación para la Ciencia e Innovación Agraria para la Red
Norte - AgroRed Norte. Piura 20009. Perú.
2Universidad Privada del Norte. Facultad de Ingeniería. Cajamarca
06002. Cajamarca. Perú.
3Universidad Nacional Pedro Ruiz Gallo. Facultad de Ingeniería
Química e Industrias Alimentarias. Lambayeque 14013. Lambayeque. Perú.
4Universidad Nacional de Frontera. Facultad de Ingeniería de
Industrias Alimentarias y Biotecnología. Sullana 20103. Piura. Perú.
*vamir0803@gmail.com
Abstract
Native fruits from
the Peruvian Andes, such as ushpa (Vaccinium floribundum Kunth)
and sachon (Hesperomeles obtusifolia (Pers.) Lindl.), grow in
high-altitude environments that favor the accumulation of bioactive compounds.
However, limited characterization has restricted their sustainable utilization.
This study analyzed the physicochemical properties, total phenolic content
(TPC), total flavonoid content (TFC), total anthocyanin content (TAC), vitamin
C (VC), and antioxidant activity (AA) of both fruits. It also evaluated the
effects of solvent type (ethanol and methanol) and concentration (70% and 80%),
and extraction time (60, 90, and 120 minutes) on extraction efficiency using
response surface methodology. Both fruits exhibited higher levels of bioactive
compounds and antioxidant activity compared to other berry species. Optimal
conditions for ushpa (64.86% methanol, 139.68 minutes) and for sachon
(64.86% ethanol, 90 minutes) yielded TPC = 3,587 and 948 mg GAE/100 g, TFC
= 1,821 and 701 mg CE/100 g, TAC = 252 and 8 mg C3G/100 g, and AA = 563 and 501
μmol TE/g, respectively. These findings support further research and potential
valorization of these native fruits.
Keywords: andean berries,
paramo, polyphenols, vitamin C, antioxidant activity, RSM
Resumen
Los frutos nativos
de los Andes peruanos, como ushpa (Vaccinium floribundum Kunth) y
sachon (Hesperomeles obtusifolia (Pers.) Lindl.), crecen en
ambientes de altura que favorecen la acumulación de compuestos bioactivos. Sin
embargo, la caracterización limitada ha restringido su uso sostenible. Este
estudio evaluó las propiedades fisicoquímicas, contenido total de fenoles (TPC),
contenido total de flavonoides (TFC), contenido total de antocianinas (TAC),
vitamina C (VC) y actividad antioxidante (AA) de ambos frutos. Asimismo, se
analizó el efecto del tipo (etanol y metanol) y concentración (70% y 80%) de
solvente y tiempo de extracción (60, 90 y 120 minutos) mediante metodología de
superficie de respuesta. Ambas frutas presentaron niveles superiores de
compuestos bioactivos y actividad antioxidante en comparación con otras
especies de bayas. Las condiciones óptimas para ushpa (64,86% de
metanol, 139,68 minutos) y sachon (64,86% de etanol, 90 minutos)
generaron valores de TPC = 3 587 mg y 948 GAE/100 g, TFC = 1 821 y 701 mg
CE/100 g, TAC = 252 y 8 mg C3G/100 g, y AA = 563 y 501 μmol TE/g,
respectivamente. Estos resultados respaldan futuras investigaciones y posible
valorización de estos frutos nativos.
Palabras clave: bayas andinas,
páramo, polifenoles, vitamina C, actividad antioxidante, RSM
Originales: Recepción: 25/07/2025
- Aceptación: 18/10/2025
Introduction
Peru, a globally
recognized megadiverse country, is home to more than 780 edible plant species,
including 525 native fruits (MIDAGRI, 2022). Within its
diverse ecosystems, the Andean paramo, a high-mountain ecosystem located
between 3,500 and 4,200 meters above sea level, stands out for its rich plant
biodiversity and extreme climatic conditions. These conditions have favored the
development of native fruit species with unique sensory and functional
properties that remain largely unexplored (Torres-Guevara et
al., 2020).
Compared with
countries such as Ecuador (Guevara-Terán et al.,
2022), Brazil (Rigolon et al., 2020), Chile
(Oyarzún et al., 2020), Cameroon (Bayang et al., 2021), and China (Liu
et al., 2022), where native fruits have been extensively characterized,
studies in the Peruvian Andes remain scarce. For example, Torres-Guevara
et al. (2023) identified 39 native fruit species in northern Peru,
most of which are still unknown in commercial markets. Berries were predominant
among them, widely recognized for their richness in vitamin C and phenolic
compounds that confer strong antioxidant activity (Bezerra et
al., 2024).
Two native berries
from the Andean paramo, ushpa (Vaccinium floribundum Kunth) and sachon
(Hesperomeles obtusifolia (Pers.) Lindl.), have long been consumed
by local communities for both nutritional and medicinal purposes. Despite their
ethnobotanical relevance, scientific studies on these species are scarce and
mostly limited to preliminary assessments of their phytochemical composition (Torres-Guevara et al., 2020).
In this context,
the present study provides a comprehensive analysis of the physicochemical
traits (soluble solids and pH), total phenolic content (TPC), total flavonoid
content (TFC), total anthocyanin content (TAC), vitamin C (VC), and antioxidant
activity (AA) of V. floribundum and H. obtusifolia from the
Peruvian Andean paramo. Recognizing that efficient extraction processes
depend on multiple interrelated variables, this study applies multivariate
optimization to identify the best extraction conditions.
We hypothesize that both species exhibit high levels of
bioactive compounds and antioxidant activity, and that extraction parameters
significantly influence their recovery. These findings provide insights into
the bioactive potential of these native species and support future research and
valorization.
Materials
and Methods
Chemicals
and Reagents
All chemicals used
in this study were of analytical grade or higher. Gallic acid, the
Folin-Ciocalteu reagent, and quercetin standard were purchased from
Millipore-Sigma (Steinheim, Germany). HPLC-grade methanol, along with
analytical-grade or higher ethanol and methanol were sourced from Supelco
(Bellefonte, PA, USA). The ascorbic acid standard was obtained from Merck
(Darmstadt, Germany). 6-Hydroxy-2,5,7,8
-tetramethylchroman-2-carboxylic acid (Trolox) was acquired from Sigma-Aldrich
(Steinheim, Germany).
Sample
Collection
In April 2023, 1 kg of mature fruits of both sachon (H.
obtusifolia) and ushpa (V. floribundum) were collected from
the paramo ecosystem in the locality of Totora, Pacaipampa district,
Ayabaca province, Piura region, Peru, at an elevation of 3,000-3,700 meters
above sea level. The characteristics of this ecosystem, along with the
distribution of these species and other relevant information, are described in Torres-Guevara et al. (2023). Species
identification was based on previous studies by botanist Fidel A.
Torres-Guevara. These species (figure 1) were selected
because, although they are not commonly commercialized, they are widely used by
local communities for nutritional and medicinal purposes.
Figure
1. Fruit-bearing shrubs of (a) V. floribundum Kunth
and (b) H. obtusifolia (Pers.) Lindl.
Figura 1. Frutales
de (a) V. floribundum Kunth y (b) H. obtusifolia (Pers.) Lindl.
Physicochemical
Analyses
Fruits were washed,
dried, and homogenized. Juice was filtered and analyzed for pH (pHep-HI98107,
HANNA Instruments, Italy) and total soluble solids (°Brix) using a HI96801
digital refractometer (HANNA Instruments, Italy). Analyses were conducted in
quintuplicate at room temperature. Color parameters (L*, a*, b*) were measured
in the CIELab* color space using a FRU WR-10QC colorimeter (Shenzhen Wave
Optoelectronics Technology Ltd., China), with measurements taken at five random
points on each sample’s surface.
Extraction
and Chemical Analysis
Samples were freeze-dried
(BK-FD10PT, Biobase, China), ground (IKA M20 universal mill, Germany), and
sieved using a Ro-Tap RX29-16 shaker (WS Tyler, USA). Particle fractions
between 150 and 300 μm were collected, packaged, and stored at -20°C in a
refrigerated incubator (FOC 215I, Velp Scientifica, Italy).
Extract
Preparation
Extracts were
prepared by maceration with agitation, adapting the method from a previous
study (León-Roque et al., 2023). The evaluated
factors were: solvent type (ethanol or methanol), solvent concentration (70% or
80%), and extraction time (60, 90, or 120 minutes). Briefly, 0.5 g of sample
was mixed with 10 mL of solvent and extracted at room temperature using a
vortex mixer (Labnet International, USA). Extracts were centrifuged (5000×g, 15
minutes, 4°C) in a Bio RS-24 multi-rotator (Boeco, Germany), and the
supernatants were stored at -20°C in 2 mL amber tubes. Before chromatographic
analysis, extracts were filtered through 0.45 μm PTFE syringe filters.
Phenolic
Compounds
TPC was determined
using the Folin–Ciocalteu method adapted from Magalhães et
al. (2010). Absorbance was measured in triplicate at 765 nm using a
Genesys 30 UV-Vis spectrophotometer (Thermo Scientific, USA), and results were
expressed as mg gallic acid equivalents (mg GAE) per 100 g of dry basis (d.b.).
TFC was measured by
the aluminum chloride colorimetric assay described by Abderrahim
et al. (2015). Absorbance was recorded in triplicate at 415 nm, and
results were expressed as mg catechin equivalents (mg CE) per 100 g d.b.
TAC was quantified
using the pH differential method described by Giusti &
Wrolstad (2001). Absorbance was recorded at the visible maximum wavelength
(λ_vis-max) and at 700 nm. Analyses were performed in triplicate, and results
were expressed as mg cyanidin-3-glucoside equivalents (C3G) per 100 g d.b.
Vitamin C
VC concentration
was determined in triplicate by HPLC-DAD (Shimadzu LCMS-2020) using ascorbic
acid as the standard. A stock solution of 500 ppm was prepared, and calibration
was constructed from 0-250 ppm. Chromatographic detection was performed at 254
nm using a Shim-pack GIST C18 (5 μm, 150 × 4.6 mm) column at a flow rate of 0.5
mL/min and an injection volume of 10 μL. The column temperature was maintained
at 30°C, and the mobile phase consisted of a methanol-water (80/20 v/v, solvent
B) for 10 minutes. Extracts were filtered through 0.45 μm syringe filters into
2 mL vials, which were then placed in the autosampler. Results were expressed
as mg VC per liter d.b., and data were processed using LabSolutions software.
In
Vitro Antioxidant Activity
AA was determined
in triplicate using the ABTS radical cation assay described by Re et al. (1999), and results were expressed as
μmol Trolox equivalents (TE) per gram d.b.
Experimental
Design
The effects of
solvent type, solvent concentration, and extraction time on TPC, TFC, TAC, and
AA were evaluated using response surface methodology (RSM), with 12 randomized
runs and 36 total experiments. Data were fitted to a second-order polynomial
equation, and analysis of variance (ANOVA) was applied to assess the
significance of individual factors and their interactions. Regression
coefficients were used to establish the direction of the effects, where
positive values reflected increases in the response, while negative values
indicated decreases. A Pareto chart of standardized effects illustrated the
magnitude and importance of each factor.
Since maximizing
one response variable may compromise another, global optimization was conducted
using a multiple-response strategy based on the desirability function. This
approach converts each response into a standardized scale (0-1), where 1
represents the optimal condition.
Vitamin C content
was excluded from the optimization process, as it was not detected in most
methanol-based extractions of both ushpa and sachon.
Data
Analysis
Physicochemical differences between ushpa and sachon were
assessed using Student’s t-test (α = 0.05). The effects of extraction
treatments were analyzed via ANOVA (α = 0.05) followed by Tukey’s test for
multiple comparisons. Statistical analyses were conducted with Minitab 18.0
(Minitab Inc., USA), while optimization analyses were performed using
Statistica 12 (Stat Soft Inc., USA) (α = 0.05).
Results
and Discussion
Physicochemical
Characteristics
The physicochemical
analysis of ushpa and sachon fruits revealed significant
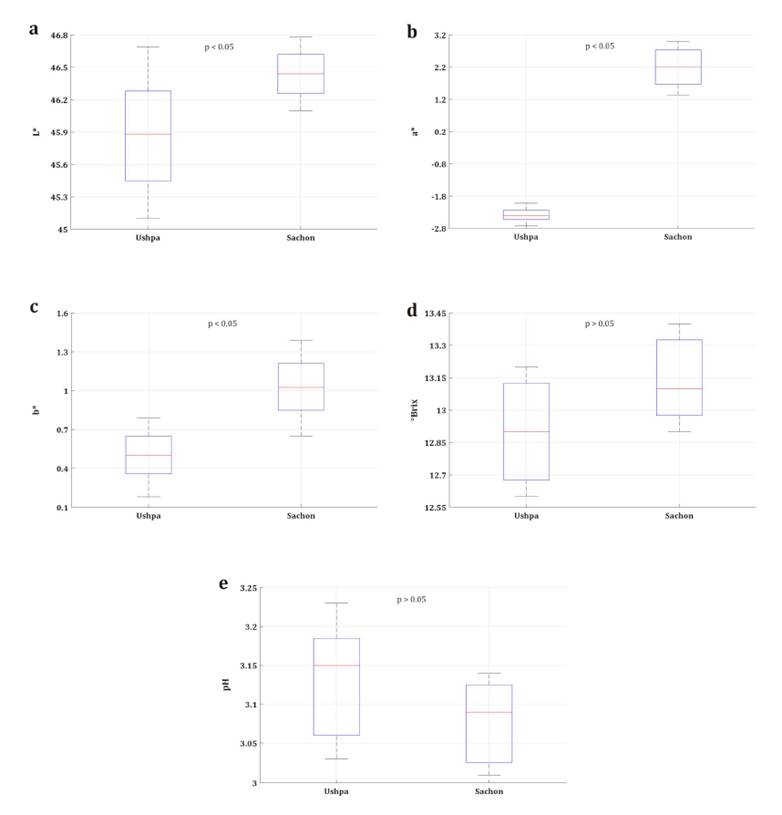
differences (p < 0.05) in color parameters (figure 2(a-c)),
with higher values in sachon (L*: 46.44 ± 0.21, a*: 2.18 ± 0.55, b*:
1.03 ± 0.21) compared to ushpa (L*: 45.88 ± 0.47, a*: -2.38 ± 0.20, b*:
0.50 ± 0.17).
Figure
2. Values of (a) L*, (b) a*, (c) b*, (d) °Brix, and (e)
pH in ushpa and sachon.
Figura
2. Valores de (a) L*, (b) a*, (c) b*,
(d) °Brix, y (e) pH en ushpa y sachon.
Lower L* values
have been reported in other berries, such as 18.6 in Rubus ulmifolius,
20.0 in Aristotelia chilensis (Mattson et al.,
2022), 31.1 in Rubus sp., 27.2 in Vaccinium sp. (Rigolon et al., 2020), and 25.7 in Vaccinium
myrtillus L. (Vega et al., 2023). The L*
value tends to decrease during fruit ripening, leading to a darker appearance,
although high L* values have also been recorded in ripe fruits such as Fragaria
vesca L. (56.7) and Prunus avium L. (62.9) (Vega
et al., 2023).
Regarding total
soluble solids (°Brix) content (figure 2(d)), no significant
differences were observed between ushpa (12.90 ± 3.13) and sachon (13.14
± 3.08) (p > 0.05). Both fruits showed values comparable to other berries,
such as Vitis vinifera L. (16.9) and Rubus idaeus (7.0-11.0) (Frías-Moreno et al., 2021; Gomes et al., 2021).
These values indicate a sweetness level similar to that of widely commercial
fruits, suggesting good potential for direct consumption or industrial
processing.
Several authors
have noted that °Brix content can vary depending on cultivar, climate, ripening
stage, and agronomic practices (King et al., 2021).
Saad et al. (2022) reported values ranging from
3.8 to 8.2 in Fragaria × ananassa, depending on the maturity stage. Seki et al. (2024) found values of 8.1 and 10.0 in
red and white Fragaria sp., respectively. Cuesta-Riaño
et al. (2022) reported values of 5.7 and 7.8 in Rubus glaucus Benth
and Rubus adenotrichos, respectively. Aliman et
al. (2020) documented high variability in °Brix among Vaccinium
myrtillus L. cultivars depending on the harvest year.
With respect to pH
(figure 2(e)), both ushpa and sachon exhibited
values close to 3.1, with no significant differences between them (p >
0.05). This similarity is attributed to shared environmental factors in the
paramo ecosystem that influence acidity. For example, environmental conditions
have been shown to affect the quality, including the acidity, of both
cultivated and wild apples collected from different locations in China (Li et al., 2021). Other berries exhibit a range of
pH values: 3.6 in grapes, 2.9-3.1 in Rubus idaeus (Frías-Moreno
et al., 2021), 3.09-3.5 in different Aronia mitschurinii harvests
(King et al., 2021), 3.1 in Rubus glaucus Benth
and 3.2 in Rubus adenotrichos (Cuesta-Riaño et
al., 2022), and 3.2-3.6 in Vaccinium myrtillus L. (Aliman et al., 2020).
Chemical
Analysis and Antioxidant Activity
Table
1, presents the results for ushpa and sachon extracts, which
confirmed our hypothesis of their high levels of TPC, TFC, TAC, VC, and AA,
highlighting their strong bioactive potential. Comparative values for other
berries are provided in Table S1.
Table
1. TPC, TFC, TAC, VC, and AA of ushpa and sachon
extracts.
Tabla
1. TPC, TFC, TAC, VC, and AA de
extractos de ushpa y sachon.
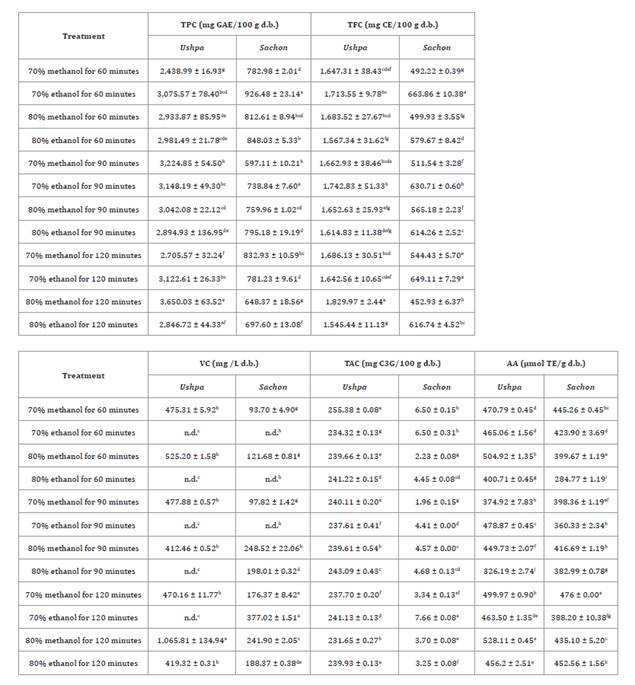
Results
are presented as mean ± standard deviation. Different letters within each
column indicate statistically significant differences between treatments (p
< 0.05). Significant differences (p < 0.05) in TPC, TFC, TAC, VC, and AA
were observed between ushpa and sachon. n.d. = not detected.
Los
resultados se presentan como media ± desviación estándar. Letras diferentes
dentro de cada columna indican diferencias estadísticamente significativas
entre tratamientos (p < 0,05). Se observaron diferencias significativas (p
< 0,05) en TPC, TFC, TAC, VC y AA entre ushpa y sachon. n.d. = no detectado.
TPC,
TFC and TAC
Ushpa displayed higher
TPC (3,004.62 mg GAE/100 g d.b. vs. 772.15 mg GAE/100 g d.b.) and TFC (1,661.10
mg CE/100 g d.b. vs. 564.08 mg CE/100 g d.b.), exceeding levels found in other
berries, including Vaccinium corymbosum L. (Aliman et
al., 2020) and Vaccinium myrtillus L. (Vega et
al., 2023). A study on Vaccinium floribundum in Ecuador reported
much lower TPC (Monge-Sevilla et al., 2024),
highlighting the high regional variability in phenolic content. While sachon
exhibited considerable TPC and TFC levels, they were lower than those
reported for Aronia mitschurinii (King et al.,
2021), Aristotelia chilensis (Mattson et al.,
2022), and Sorbus aucuparia L. (Orsavová et
al., 2023).
Ushpa (240.50 mg C3G/100 g d.b.) also had significantly higher TAC
than sachon (4.46 mg C3G/100 g d.b.), consistent with the dark blue
pigmentation of ushpa. TAC values in ushpa surpassed those
reported for Rubus ulmifolius (Mattson et al.,
2022), Rubus idaeus (Frías-Moreno et al.,
2021), Sorbus aucuparia L. (Orsavová et al.,
2023), and Vitis vinifera L. (Gomes et al.,
2021).
Similarly, plant
genetics also plays a key role in metabolite biosynthesis and antioxidant
activity. For instance, Lebedev et al. (2022)
reported a strong positive correlation (r = 0.8) between the genetic diversity
of Rubus idaeus L. and its TPC, TFC, and TAC. However, another study
found no correlation between TPC, TFC, TAC, and AA in Vaccinium macrocarpon Aiton
and its genetic profile (Debnath & An, 2019). This
discrepancy may be due to the use of non-specific genetic markers (Lebedev et al., 2022), or to the complex
interaction between environmental conditions and genetics, as observed in VC
content of Fragaria × ananassa Duchesne (Ali &
Serçe, 2022).
Vitamin C
Ushpa had significantly
higher VC content than sachon (568.92 vs. 200.48 mg/L d.b.). This
difference may be due to bioactive compounds in sachon being more
strongly bound within its solid matrix, requiring more intensive extraction
methods (Cuesta-Riaño et al., 2022; Mattson et
al., 2022).
VC levels in ushpa
were higher than those in Vaccinium floribundum and Rubus
glabratus Kunth (Monge-Sevilla et al., 2024),
but lower than those in Sorbus aucuparia L. and Rubus idaeus (Frías-Moreno et al., 2021; Orsavová et al., 2023).
VC content is influenced by genotype and environmental factors such as altitude
and sunlight (Bayang et al., 2021). In Vaccinium
floribundum, VC content increased with elevation (Guevara-Terán
et al., 2022).
Antioxidant
Activity (AA)
AA measured with
ABTS was significantly higher in ushpa than in sachon (449.47 vs.
401.03 μmol TE/g d.b.), consistent with findings in other fruit species,
such as Opuntia ficus-indica, Myrica rubra, Vaccinium
floribundum, and Rubus glabratus (El Mannoubi,
2021; Liu et al., 2022; Monge-Sevilla et al., 2024).
However, although ushpa
exhibited greater AA, the difference with sachon was smaller than
expected, possibly due to the presence of other antioxidant compounds not
measured in this study, such as tannins (Varo et al.,
2021). For example, in Vaccinium corymbosum L., the cultivar Jewel
contained more resveratrol but had lower AA than Windsor, while Millenia had
the highest VC yet lower AA than both (Varo et al.,
2021).
AA levels, like TPC
and TFC, are influenced by altitude, solar exposure, and fruit variety, with
higher AA reported in Vaccinium floribundum harvested at greater
elevations (Guevara-Terán et al., 2022).
It is also
important to note that AA results can vary depending on the assay (table
1). For instance, Vega et al. (2023)
reported that using the DPPH method, Vaccinium myrtillus L. showed the
highest AA, followed by Fragaria vesca L. and Prunus avium L.
However, when the FRAP assay was used, the ranking partially reversed, with
strawberries showing the highest AA, followed by blueberries and cherries.
Effect
of Extraction Conditions
Certain extraction
parameters had a significant impact on the outcomes, consistent with our
hypothesis. However, as detailed below, the magnitude and direction of these
effects varied depending on the fruit species and the metabolite or response
assessed.
Influence
of Solvent Type and Concentration
The solvent type
significantly impacted the extraction of bioactive compounds (Figure
S1 and Figure S2), with methanol showing greater efficacy in extracting TFC
and maximizing AA in ushpa, while in sachon, methanol had a
negative effect on TPC and TFC but a positive effect on TAC and AA (p <
0.05). This variability is likely due to the relative polarity of solvents,
with methanol being slightly more polar than ethanol, which favors the
extraction of polar compounds such as anthocyanins but may hinder the
extraction of non-polar compounds (More & Arya, 2021).
Previous studies also highlight these trends. In Opuntia
ficus-indica, 80% ethanol outperformed methanol in the extraction of
polyphenols (El Mannoubi, 2021). However, in Băbească
neagră peels, ethanol also enhanced TAC extraction (Serea
et al., 2023), whereas for TFC, ethanol was more efficient in the
peel of Opuntia ficus-indica, while methanol was superior in the pulp (El Mannoubi, 2021). These results underscore that solvent
effectiveness also depends on the fruit part and the specific class of metabolites.
Solvent
concentration also showed significant effects (p < 0.05). In ushpa,
it positively affected TFC, while in sachon, it positively influenced
TFC, TAC, and AA. Moreover, in ushpa, the interaction between solvent
type (ethanol) and concentration was significant for both TPC and TFC.
More diluted
mixtures allow for greater solvent penetration into the plant matrix, thereby
promoting phenolic release. However, high ethanol concentrations reduce
polarity, hindering polar compound extraction. This relationship is not linear;
for example, Serea et al. (2023) reported that
50% ethanol was optimal for TAC in Băbească neagră peels, whereas 85%
was optimal for TPC.
Interestingly, pure
methanol also enhanced TPC, TFC, and AA, as observed in seeds of Carica
papaya L. (Robles-Apodaca et al., 2024), Passiflora
edulis, and Nephelium lappaceum L. (Sai-Ut et
al., 2023). The addition of water increases polyphenol ionization,
promoting their solubilization. For instance, in Vaccinium floribundum,
20% ethanol improved TAC extraction compared to 60% (Pérez et
al., 2021). In Sambucus nigra L., 45% ethanol was suitable for
TPC extraction (Pascariu et al., 2024).
Influence
of Extraction Time
Extraction time had
variable effects depending on the species and the bioactive compound evaluated.
In ushpa, longer extraction times positively affected TAC and AA, while
in sachon, extraction time had both linear and quadratic effects on AA.
However, prolonged extraction tended to negatively impact TPC, particularly in ushpa,
and both species exhibited a decline in TPC over extended extraction periods.
This suggests that while longer extraction may improve yields, it can also lead
to compound degradation due to exposure to oxygen, light, or heat.
The nature of this
response also depends on the interaction between time and solvent. For example,
in ushpa, extraction with methanol at short times was particularly
favorable for TAC, whereas prolonged methanol use reduced both TPC and TFC.
These results align with findings in Carica papaya L. seeds, where
methanol exhibited a non-linear behavior depending on both time and
concentration. At longer extraction times, the highest TFC yields were obtained
when using either pure water (0% methanol) or absolute methanol (100%) as the
extraction solvent (Robles-Apodaca et al., 2024).
The literature
reports similarly variable results. In Passiflora edulis and Nephelium
lappaceum L., time had no effect on TPC at a fixed ethanol concentration (Sai-Ut et al., 2023). However, in Băbească
neagră peels, a short extraction (25 minutes) with 85% ethanol reduced TAC
and AA, while a longer duration (≥44 minutes) and 64% ethanol improved AA (Serea et al., 2023). Similarly, in Malpighia
emarginata DC. residues, TAC extraction was more
efficient with low ethanol concentrations but progressively declined with
extended extraction times (Cerino et al., 2023).
Global
Desirability Optimization
Given the varied
effects of extraction conditions on bioactive compounds, global desirability
optimization was applied (Figure S3). For ushpa,
the optimal conditions determined were 64.86% methanol and an extraction time
of 139.68 minutes, resulting in estimated values of TPC = 3,586.8 mg GAE/100 g
d.b., TFC = 1,820.8 mg CE/100 g d.b., TAC = 251.87 mg C3G/100 g d.b., and AA =
562.76 μmol TE/g d.b. In contrast, sachon exhibited optimal conditions
with 64.86% ethanol and 90 minutes of extraction, achieving TPC of 948.37 mg
GAE/100 g d.b., TFC of 701.06 mg CE/100 g d.b., TAC of 7.84 mg C3G/100 g d.b.,
and AA of 500.71 μmol TE/g d.b.
Previous studies have reported equally variable optimal
conditions. For instance, in Băbească neagră grape peels, using 85%
ethanol for 52.14 minutes yielded TPC and TAC values ranging from 24.67 to
43.97 mg/g and 1.71 to 2.74 mg C3G/g, respectively (Serea et
al., 2023). In passion fruit and rambutan seeds, maximum TPC and AA
were achieved with 67% and 54% ethanol, respectively, after 186 and 221 minutes
(Sai-Ut et al., 2023). In Carica papaya L.
seeds, extraction with 100% methanol for 6 hours resulted in 6.17 mg GAE/g (TPC)
and 52.75 mg QE/g (TFC) (Robles-Apodaca et al., 2024).
In Malpighia emarginata DC. residues, TAC of
2.54 mg/g was reached with 12% ethanol and 25 minutes of extraction (Cerino et al., 2023).
Conclusions
and Future Directions
This study provides
the first systematic characterization of the physicochemical, chemical, and
antioxidant properties of two native Andean fruits from northern Peru: ushpa
and sachon. Both species showed physicochemical parameters (color,
°Brix, and pH) comparable to commercial berries, and remarkably high TPC, TFC,
TAC, and AA values, often surpassing those of widely consumed fruits. These
traits likely reflect their adaptation to harsh, high-altitude environments
with intense solar radiation, which promotes the accumulation of secondary
bioactive metabolites.
The study of
extraction conditions revealed significant, species-specific effects on each
attribute. This complexity led to the use of a multivariate optimization
approach based on global desirability, which identified specific extraction
conditions for maximizing bioactive compound recovery and antioxidant activity.
These findings not
only provide academic insights but also highlight the potential of ushpa and
sachon as sources of functional ingredients for the food, nutraceutical,
and cosmetic industries. However, the limited scientific knowledge about native
species in Peru, particularly regarding their chemical composition and
functionality, remains a barrier to their integration into the agri-food chain.
Future research should include proximate composition and detailed phytochemical
profiling through chromatographic techniques to identify and quantify
individual metabolites. Additionally, while the in vitro antioxidant tests used
here offer an initial assessment, future studies should incorporate cellular,
animal, or clinical models to better evaluate their real-world functionality.
Abderrahim, F.,
Huanatico, E., Segura, R., Arribas, S., Gonzalez, M. C., & Condezo-Hoyos,
L. (2015). Physical features, phenolic compounds, betalains and total
antioxidant capacity of coloured quinoa seeds (Chenopodium quinoa Willd.)
from Peruvian Altiplano. Food Chemistry, 183, 83-90.
https://doi.org/10.1016/j.foodchem.2015.03.029
Ali, M. N., &
Serçe, S. (2022). Vitamin C and fruit quality consensus in breeding elite
European strawberry under multiple interactions of environment. Molecular
Biology Reports, 49(12), 11573-11586.
https://doi.org/10.1007/s11033-022-07849-5
Aliman, J.,
Michalak, I., Bušatlić, E., Aliman, L., Kulina, M., Radović, M., &
Hasanbegović, J. (2020). Study of the physicochemical properties of highbush blueberry
and wild bilberry fruit in central Bosnia. Turkish Journal of Agriculture
and Forestry, 44(2), 156-168. https://doi. org/10.3906/tar-1902-36
Bayang, J. P.,
Laya, A., Kolla, M. C., & Koubala, B. B. (2021). Variation of physical
properties, nutritional value and bioactive nutrients in dry and fresh wild
edible fruits of twenty-three species from Far North region of Cameroon. Journal
of Agriculture and Food Research, 4, 100146.
https://doi.org/10.1016/j.jafr.2021.100146
Bezerra, M.,
Ribeiro, M., Cosme, F., & Nunes, F. M. (2024). Overview of the distinctive
characteristics of strawberry, raspberry, and blueberry in berries, berry
wines, and berry spirits. Comprehensive Reviews in Food Science and Food
Safety, 23, e13354. https://doi. org/10.1111/1541-4337.13354
Cerino, M. C.,
Pinela, J., Caleja, C., Saux, C., Pereira, E., & Barros, L. (2023). Dynamic
Maceration of Acerola (Malpighia emarginata DC.) Fruit Waste: An
Optimization Study to Recover Anthocyanins. Agronomy, 13, 2202.
https://doi.org/10.3390/agronomy13092202
Cuesta-Riaño, C.
S., Castro-Guascaa, M. P., & Tarazona-Díaz, M. P. (2022). Anthocyanin
Extract from Blackberry Used as an Indicator of Hydrogen Potential. International
Journal of Fruit Science, 22(1), 224–234.
https://doi.org/10.1080/15538362.2022.2037036
Debnath, S. C.,
& An, D. (2019). Antioxidant properties and structured biodiversity in a
diverse set of wild cranberry clones. Heliyon, 5(4), e01493.
https://doi.org/10.1016/j.heliyon.2019. e01493
El Mannoubi, I. (2021). Effect of extraction solvent on phenolic
composition, antioxidant and antibacterial activities of skin and pulp of
Tunisian red and yellow–orange Opuntia Ficus Indica fruits. Journal of Food
Measurement and Characterization, 15(1), 643–651. https://
doi.org/10.1007/s11694-020-00673-0
Frías-Moreno, M.
N., Parra-Quezada, R. Á., Ruíz-Carrizales, J., González-Aguilar, G. A.,
Sepulveda, D., Molina-Corral, F. J., Jacobo-Cuellar, J. L., & Olivas, G. I.
(2021). Quality, bioactive compounds and antioxidant capacity of raspberries
cultivated in northern Mexico. International Journal of Food Properties,
24(1), 603–614. https://doi.org/10.1080/10942912.2021.19 08352
Giusti, M. M.,
& Wrolstad, R. E. (2001). Characterization and Measurement of Anthocyanins
by UV-Visible Spectroscopy. Current Protocols in Food Analytical Chemistry,
0, F1.2.1-F1.2.13.
https://doi.org/https://doi.org/10.1002/0471142913.faf0102s00
Gomes, V., Rendall,
R., Reis, M. S., Mendes-Ferreira, A., & Melo-Pinto, P. (2021).
Determination of sugar, ph, and anthocyanin contents in port wine grape berries
through hyperspectral imaging: An extensive comparison of linear and non-linear
predictive methods. Applied Sciences, 11, 10319.
https://doi.org/10.3390/app112110319
Guevara-Terán, M.,
Padilla-Arias, K., Beltrán-Novoa, A., González-Paramás, A. M., Giampieri, F.,
Battino, M., Vásquez-Castillo, W., Fernandez-Soto, P., Tejera, E., &
Alvarez-Suarez, J. M. (2022). Influence of Altitudes and Development Stages on
the Chemical Composition, Antioxidant, and Antimicrobial Capacity of the Wild
Andean Blueberry (Vaccinium floribundum Kunth). Molecules, 27,
7525. https://doi.org/10.3390/molecules27217525
King, E. S., Cho,
J., Li, H., Jiang, X., Madler, A. K., Weishair, M. K., Glenn, S., Brand, M. H.,
Xu, C., & Bolling, B. W. (2021). Time of harvest affects United
States-grown Aronia mitschurinii berry polyphenols, °Brix, and acidity. Journal
of Agriculture and Food Research, 6, 100248.
https://doi.org/10.1016/j.jafr.2021.100248
Lebedev, V. G.,
Lebedeva, T. N., Vidyagina, E. O., Sorokopudov, V. N., Popova, A. A., &
Shestibratov, K. A. (2022). Relationship between Phenolic Compounds and
Antioxidant Activity in Berries and Leaves of Raspberry Genotypes and Their
Genotyping by SSR Markers. Antioxidants, 11, 1961.
https://doi.org/10.3390/antiox11101961
León-Roque, N.,
Romero Guzmán, B. M., Oblitas-Cruz, J. F., & Hidalgo-Chávez, D. W. (2023).
Optimization of total polyphenol extraction and flavonoid screening by mass
spectrometry in mango (Mangifera indica L.) waste from Peru. Food
Science and Technology, 43(e105322). https://
doi.org/10.1590/fst.105322
Li, Y., Sun, H.,
Li, J., Qin, S., Yang, W., Ma, X., Qiao, X., & Yang, B. (2021). Effects of
genetic background and altitude on sugars, malic acid and ascorbic acid in
fruits of wild and cultivated apples (Malus sp.). Foods, 10,
2950. https://doi.org/10.3390/foods10122950
Liu, Z., Zhang, J.,
Lu, S., Tang, W., Zhou, Y., & Quek, S. Y. (2022). Effects of different
drying methods on phenolic components and in vitro hypoglycemic
activities of pulp extracts from two Chinese bayberry (Myrica rubra Sieb.
et Zucc.) cultivars. Food Science and Human Wellness, 11(2),
366–373. https://doi.org/10.1016/j.fshw.2021.11.014
Magalhães, L. M.,
Santos, F., Segundo, M. A., Reis, S., & Lima, J. L. F. C. (2010). Rapid
microplate high-throughput methodology for assessment of Folin-Ciocalteu
reducing capacity. Talanta, 83(2), 441-447.
https://doi.org/10.1016/j.talanta.2010.09.042
Mattson, M. G., Sozzi,
A., Corfield, R., Gagneten, M., Franceschinis, L., Schebor, C., &
Salvatori, D. (2022). Colorant and antioxidant properties of freeze-dried
extracts from wild berries: use of ultrasound-assisted extraction method and
drivers of liking of colored yogurts. Journal of Food Science and Technology,
59(3), 944–955. https://doi.org/10.1007/s13197-021- 05096-3
MIDAGRI. (2022). Situación
actual de las frutas y verduras en el Perú: Producción, exportación e
importación. https://repositorio.midagri.gob.pe/handle/20.500.13036/1227
Monge-Sevilla, R.
D., Fernández, L., Espinoza-Montero, P. J., Méndez-Durazno, C., Cisneros-Pérez,
P. A., Romero-Estévez, D., Bolaños-Méndez, D., Alvarez-Paguay, J., & Jadán,
M. (2024). Chemical composition and antioxidant properties of native Ecuadorian
fruits: Rubus glabratus Kunth, Vaccinium floribundum Kunth, and Opuntia
soederstromiana. Heliyon, 10, e30593.
https://doi.org/10.1016/j.heliyon.2024.e30593
More, P. R., &
Arya, S. S. (2021). Intensification of bio-actives extraction from pomegranate
peel using pulsed ultrasound: Effect of factors, correlation, optimization and
antioxidant bioactivities. Ultrasonics Sonochemistry, 72, 105423.
https://doi.org/10.1016/j.ultsonch.2020.105423
Orsavová, J.,
Juríková, T., Bednaříková, R., & Mlček, J. (2023). Total Phenolic and Total
Flavonoid Content, Individual Phenolic Compounds and Antioxidant Activity in
Sweet Rowanberry Cultivars. Antioxidants, 12, 913.
https://doi.org/10.3390/antiox12040913
Oyarzún, P.,
Cornejo, P., Gómez-Alonso, S., & Ruiz, A. (2020). Influence of Profiles and
Concentrations of Phenolic Compounds in the Coloration and Antioxidant
Properties of Gaultheria poeppigii Fruits from Southern Chile. Plant
Foods for Human Nutrition, 75(4), 532-539. https://doi.
org/10.1007/s11130-020-00843-x
Pascariu, O. E.,
Dias, L. G., & Israel-Roming, F. (2024). Optimization of Extraction Method
of Bioactive Compounds from Elderberries (Sambucus nigra L.) and Testing
Extract Stability. Horticulturae, 10(7).
https://doi.org/10.3390/horticulturae10070743
Pérez, B., Endara, A., Garrido, J., & Ramírez-Cárdenas, L.
(2021). Extraction of anthocyanins from mortiño (Vaccinium floribundum)
and determination of their antioxidant capacity. Revista Facultad Nacional
de Agronomia Medellin, 74(1), 9453–9460. https://doi.org/10.15446/
rfnam.v74n1.89089
Re, R., Nicoletta,
P., Anna, P., Ananth, P., Min, Y., & Catherine, R.-E. (1999). Antioxidant
activity applying an improved ABTS radical cation decolorization assay. Free
Radical Biology and Medicine, 26(9-10), 1231-1237.
Rigolon, T. C. B.,
de Barros, F. A. R., Vieira, É. N. R., & Stringheta, P. C. (2020).
Prediction of total phenolics, anthocyanins and antioxidant capacity of
blackberry (Rubus sp.), blueberry (Vaccinium sp.) and jaboticaba (Plinia
cauliflora (Mart.) Kausel) skin using colorimetric parameters. Food
Science and Technology, 40, 620–625.
https://doi.org/10.1590/fst.34219
Robles-Apodaca, S.
M., González-Vega, R. I., Ruíz-Cruz, S., Estrada-Alvarado, M. I., Cira-Chávez,
L. A., Márquez-Ríos, E., Del-Toro-Sánchez, C. L., Ornelas-Paz, J. de J.,
Suárez-Jiménez, G. M., & Ocaño-Higuera, V. M. (2024). Optimization of
Extraction Process for Improving Polyphenols and Antioxidant Activity from
Papaya Seeds (Carica papaya L.) Using Response Surface Methodology. Processes,
12(2729). https://doi.org/10.3390/pr12122729
Saad, A. G., Azam,
M. M., & Amer, B. M. A. (2022). Quality Analysis Prediction and
Discriminating Strawberry Maturity with a Hand-held Vis–NIR Spectrometer. Food
Analytical Methods, 15(3), 689–699.
https://doi.org/10.1007/s12161-021-02166-2
Sai-Ut, S.,
Kingwascharapong, P., Mazumder, M. A. R., & Rawdkuen, S. (2023).
Optimization of extraction of phenolic compounds and antioxidants from passion
fruit and rambutan seeds using response surface methodology. Journal of
Agriculture and Food Research, 14(100888).
https://doi.org/10.1016/j.jafr.2023.100888
Seki, H., Murakami,
H., Ma, T., Tsuchikawa, S., & Inagaki, T. (2024). Evaluating Soluble Solids
in White Strawberries: A Comparative Analysis of Vis-NIR and NIR Spectroscopy. Foods,
13(2274). https://doi.org/10.3390/foods13142274
Serea, D.,
Constantin, O. E., Horincar, G., Stănciuc, N., Aprodu, I., Bahrim, G. E., &
Râpeanu, G. (2023). Optimization of Extraction Parameters of Anthocyanin
Compounds and Antioxidant Properties from Red Grape (Băbească neagră)
Peels. Inventions, 8(59). https://doi.
org/10.3390/inventions8020059
Torres-Guevara, F.
A., Yupanqui, M. L. G., & Suárez-Rebaza, L. A. (2020). Sustancias
bioactivas y actividad antioxidante de frutales nativos de páramos y bosques de
neblina del norte peruano. Revista Peruana de Medicina Integrativa, 5(4),
129–134. https://doi.org/10.26722/ rpmi.2020.54.185
Torres-Guevara, F.
A., Ganoza-Yupanqui, M. L., Mantilla-Rodriguez, E., Suárez-Rebaza, L. A., &
Bussmann, R. W. (2023). Ethnobotany of fruit species native to paramos and
cloud forests of Northern Peru. Ethnobotany Research and Applications, 25(10).
https://doi.org/10.32859/ era.25.10.1-15
Varo, M. Á.,
Martín-Gómez, J., Mérida, J., & Serratosa, M. P. (2021). Bioactive
compounds and antioxidant activity of highbush blueberry (Vaccinium corymbosum)
grown in southern Spain. European Food Research and Technology, 247(5),
1199–1208. https://doi.org/10.1007/ s00217-021-03701-5
Vega, E. N.,
García-Herrera, P., Ciudad-Mulero, M., Dias, M. I., Matallana-González, M. C.,
Cámara, M., Tardío, J., Molina, M., Pinela, J., C. S. P. Pires, T., Barros, L.,
Fernández-Ruiz, V., & Morales, P. (2023). Wild sweet cherry, strawberry and
bilberry as underestimated sources of natural colorants and bioactive compounds
with functional properties. Food Chemistry, 414(135669).
https://doi.org/10.1016/j.foodchem.2023.135669
Acknowledgments
The authors thank
Fidel A. Torres Guevara and David Gálvez Paucar for providing the ushpa and
sachon illustrations, and Luis Juarez Calderón and Juan Garrido
Arismendis for their support in the physicochemical analyses.
Funding
This work was
funded by the National Council of Science, Technology, and Technological
Innovation (CONCYTEC) and the National Program for Scientific Research and
Advanced Studies (PROCIENCIA) under the “E041-2024-03 Basic Research Projects”
competition, through contract PE501089307- 2024. The project titled “Ciencia
detrás del conocimiento nativo: Perfil bioactivo y actividad antioxidante con
enfoque quimiométrico, y análisis filogenético de frutales nativos de páramo y
bosques nublados de Piura en el contexto de cambio climático y seguridad
alimentaria” was supported through this funding.
Ethics declarations
The authors declare no conflict of interest. All experiments
were conducted in strict compliance with regulations for genetic resources
established by the local authority, the National Forest and Wildlife Service
(SERFOR).