Physiological and Biochemical Responses of Neltuma ruscifolia under Na2SO4 Stress
DOI:
https://doi.org/10.48162/rev.39.188Keywords:
phytohormones, ion homeostasis, antioxidant enzymes, photosynthesis, salinity stressAbstract

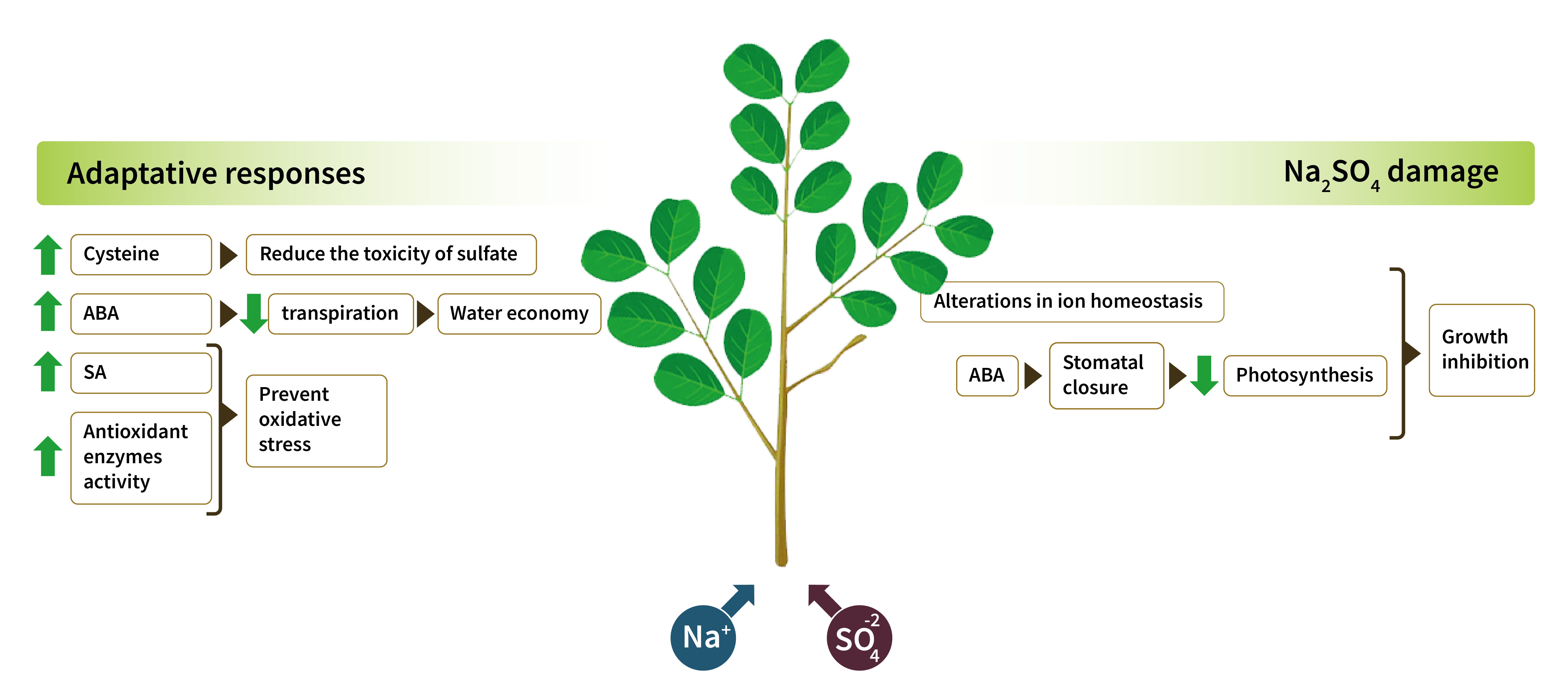
Salt stress limits plant production in arid and semi-arid zones. Although Na2SO4 is frequent in saline soils, most studies on plant physiological responses to salt stress were conducted using NaCl. This study aimed to determine the effect of Na2SO4 salt stress on physiological and biochemical responses in Neltuma ruscifolia. Increasing concentrations of Na2SO4 were added to 6-month-old plants grown hydroponically in 25% Hoagland nutrient solution. After 60 days of saline treatments, biomass, cysteine concentration, gas exchange, mineral composition, abscisic acid and salicylic acid concentrations, and antioxidant enzyme activity were determined. It is concluded that 200 mmol L-1 Na2SO4 is the threshold for N. ruscifolia seedling growth. Growth inhibition can be attributed to altered ionic homeostasis and photosynthesis inhibition after stomatal closure. Nevertheless, the species shows adaptive responses to this salt. Stomatal closure and increased foliar concentrations of abscisic acid contribute to water economy, while cysteine synthesis reduces sulfate toxicity. In parallel, salt stress induces salicylic acid accumulation in leaves, increasing the activity of antioxidant enzymes that prevent oxidative stress.
Highlights:
- The threshold for growth of Neltuma ruscifolia is 200 mmol L-1 Na2SO4.
- Salt stress altered ionic homeostasis.
- Sodium sulfate inhibited photosynthesis due to stomatal closure.
- Salt stress increased concentrations of abscisic acid and salicylic acid.
- Salt stress triggers the species' antioxidant response.
Downloads
References
Aghajanzadeh, T. A.; Reich, M.; Hawkesford, M. J.; Burow, M. 2019. Sulfur metabolism in Allium cepa is hardly affected by chloride and sulfate salinity. Archives of Agronomy and Soil Science. 65: 45-95. https://doi.org/10.1080/03650340.2018.1540037
Ahanger, M. A.; Aziz, U.; Alsahli, A. A.; Alyemeni, M. N.; Ahmad, P. 2019. Influence of exogenous salicylic acid and nitric oxide on growth, photosynthesis, and ascorbate-glutathione cycle in salt-stressed Vigna angularis. Biomolecules. 26: 42. https://doi.org/10.3390/ biom10010042
Ahmadizadeh, M.; Vispo, N. A.; Calapit-Palao, C. D. O.; Pangaan, I. D.; Viña, C. D.; Singh, R. K. 2016. Reproductive stage salinity tolerance in rice: a complex trait to phenotype. Indian Journal of Plant Physiology. 21: 528-536. https://doi.org/10.1007/s40502-016-0268-6
Barhoumi, Z. 2018. Physiological response of the facultative halophyte, Aeluropus littoralis, to different salt types and levels. Plant Biosystems. 153: 298-305. https://doi.org/10.1080 /11263504.2018.1478901
Boletta, P.; Ravelo, A.; Planchuelo, A.; Grilli, M. 2006. Assessing deforestation in the Argentine Chaco. Forest Ecology and Management. 228: 108-114. https://doi.org/10.1016/j. foreco.2006.02.045
Bradford, M. M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry. 72: 248-54. https://doi.org/10.1006/abio.1976.9999
Bravo, S. J.; Pece, M.; del Corro, F.; Ojeda Brozovich, F.; Lepiscopo, M. 2016. Anatomical changes in roots and hypocotyls of Prosopis ruscifolia (Fabaceae) seedlings exposed to saline stress. Revista de Biología Tropical. 64: 1007-1017.
Chrysargyris, A.; Xylia, P.; Botsaris, G.; Tzortzakis, N. 2017. Antioxidant and antibacterial activities, mineral and essential oil composition of spearmint (Mentha spicata L.) affected by the potassium levels. Industrial Crops and Products. 103: 202-212. https://doi.org/10.1016/j. indcrop.2017.04.010
Dalzotto, D.; Sharry, S.; Piñuel, L.; Boeri, P. (en prensa). Challenges in germination of Neltuma caldenia in semi-arid regions: optimization of germination protocols, influence of saline stress and seed quality. Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina.
Devinar, G.; Llanes, A.; Masciarelli, O.; Luna, V. 2013. Different relative humidity conditions combined with chloride and sulfate salinity treatments modify abscisic acid and salicylic acid levels in the halophyte Prosopis strombulifera. Plant Growth Regulation. 70: 247-256. https:// doi.org//10.1007/s10725-013-9796-5
Durgbanshi, A.; Arbona, V.; Pozo, O.; Miersch, O.; Sancho, J. V.; Gómez-Cadenas, A. 2005. Simultaneous determination of multiple phytohormones in plant extracts by liquid chromatography-electrospray tandem mass spectrometry. Journal of Agricultural and Food Chemistry. 53: 8437-8442. https://doi.org/10.1021/jf050884b
Galizzi, F.; Angueira, C.; Prieto, D. 1999. Suelos de la planta piloto de drenaje del INTA, Santiago del Estero. Revista Quebracho. 7: 52-60.
Giannopolitis, N.; Ries, S. K. 1977. Superoxide dismutase. I. Occurrence in higher plants. Plant Physiology. 59: 309-314. https://doi.org/10.1104/pp.59.2.309
Giménez, A. M.; Moglia, J. G. 2003. Árboles del Chaco Argentino. Guía para el reconocimiento dendrológico. Editorial El Liberal. Argentina. 307p.
Irakoze, W.; Vanpee, B.; Rufyikiri, G.; Dailly, H.; Nijimbere, S.; Lutts, S. 2019. Comparative effects of chloride and sulfate salinities on two contrasting rice cultivars (Oryza sativa L.) at the seedling stage. Journal of Plant Nutrition. 42: 1-15. https://doi.org/10.1080/01904167.2 019.1584222
Li, J.; Pu, L.; Han, M.; Zhu, M.; Zhang, R.; Xiang, Y. 2014. Soil salinization research in China: Advances and prospects. Journal of Geographical Science. 24: 943-960.
Meloni, D. A. 2017. Fisiología Vegetal: respuestas de especies leñosas al estrés salino. Editorial Universidad Nacional de Santiago del Estero, Argentina. 165 p.
Meloni, D. A.; Gulotta, M. R.; Martínez, C. A. 2008a. Prosopis ruscifolia Griseb. (vinal) tolera concentraciones salinas equivalentes al agua de mar y excluye iones tóxicos de la parte aérea. Revista Quebracho. 16: 32-40.
Meloni, D. A.; Gulotta, M. R.; Oliva Cano, M. A. 2008b. El estrés salino incrementa la actividad de enzimas antioxidantes y la concentración de polifenoles en vinal (Prosopis ruscifolia G.). Revista Quebracho. 15: 27-31.
Meloni, D. A.; Gulotta, M. R.; Silva, D. M.; Arraiza, M. P. 2019. Effects of salt stress on germination, seedling growth, osmotic adjustment, and chlorophyll fluorescence in Prosopis alba G. Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina. 51(1): 69-78.
Miura, K.; Okamoto, H.; Okuma, E.; Shiba, H.; Kamada, H.; Hasegawa, P. M.; Murata, Y. 2013. SIZ1 deficiency causes reduced stomatal aperture and enhanced drought tolerance via controlling salicylic acid-induced accumulation of reactive oxygen species in Arabidopsis. Plant Journal. 73: 91-104. https://doi.org/10.1111/tpj.12014
Munns, R.; Gilliham, M. 2015. Salinity tolerance of crops-what is the cost? New Phytology. 208: 668-673. https://doi.org/10.1111/nph.13519
Nakano, Y.; Asada, K. 1981. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiology. 22: 867-880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Palchetti, M. V.; Llanes, A.; Reginato, M.; Barboza, G.; Luna, V.; Cantero, J. J. 2020. Germination responses of Lycium humile, an extreme halophytic Solanaceae: understanding its distribution in saline mudflats of the southern Puna. Acta Botanica Brasilica. 34: 540-548. https://doi.org/10.1590/0102-33062020abb0034
Peterson, A.; Murphy, K. 2015. Tolerance of lowland quinoa cultivars to sodium chloride and sodium sulfate salinity. Crop Science. 55: 331-338. https://doi.org/10.2135/cropsci2014.04.0271
Piraino, S.; Roig, F. A. 2024. Landform heterogeneity drives multi-stemmed Neltuma flexuosa growth dynamics. Implication for the Central Monte Desert forest management. Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina. 56(1): 26-34. DOI: https://doi.org/10.48162/rev.39.120
Reginato, M.; Sosa, L.; Llanes, A.; Hampp, E.; Vettorazzi, N.; Reinoso, H.; Luna, V. 2014. Growth responses and ion accumulation in the halophytic legume Prosopis strombulifera are determined by Na2SO4 and NaCl. Plant Biology. 16: 97-106. http://dx.doi.org/10.1111/ plb.12001
Reginato, M.; Travaglia, C.; Reinoso, H.; Garello, F.; Luna, V. 2015. Anatomical modifications in the halophyte Prosopis strombulifera caused by salt mixture. Flora. 218: 75-85. https://doi. org/10.1016/j.flora.2015.11.008
Reginato, M.; Turcio, E. T.; Luna, V.; Papenbrock, J. 2019. Differential effects of NaCl and Na2SO4 on the halophyte Prosopis strombulifera are explained by different responses of photosynthesis and metabolism. Plant Physiology and Biochemistry. 11: 306-314. https:// doi.org/10.1016/j.plaphy.2019.05.027
Reginato, M.; Cenzano, A. M.; Arslan, I.; Furlán, A.; Varela, C.; Cavallin, V.; Papenbrock, J.; Luna, V. 2021. Na2SO4 and NaCl salts differentially modulate the antioxidant systems in the highly stress tolerant halophyte Prosopis strombulifera. Plant Physiology and Biochemistry. 167: 748-762. https://doi.org/10.1016/j.plaphy.2021.09.003
Reich, M.; Aghajanzadeh, T.; Helm, J.; Parmar, M. J.; Hawkesford, S.; De Kok, L. J. 2017. Chloride and sulfate salinity differently affect biomass, mineral nutrient composition and expression of sulfate transport and assimilation genes in Brassica rapa. Plant and Soil. 411: 319-32. https://doi.org/10.1007/s11104-016-3026-7
Reich, M.; Aghajanzadeh, T.; Parmar, S.; Hawkesford, M. J.; De Kok, L. J. 2018. Calcium ameliorates the toxicity of sulfate salinity in Brassica rapa. Journal of Plant Physiology. 231: 1-8. https://doi.org/10.1016/j.jplph.2018.08.014
Riemenschneider, A.; Nikiforova, V.; Hoefgen, R. De Kok, L. J.; Papenbrock, J. 2005. Impact of elevated H2S on metabolite levels, activity of enzymes and expression of genes involved in cysteine metabolism. Plant Physiology and Biochemistry. 43: 473-483. https://doi.org/10.1016/j. plaphy.2005.04.001
Sigrist, M. R.; Stefanello, T. H.; de Souza, C. S.; Vargas, W.; Almeida, K. S. M.; Laroca, S.; Mansano, V. F. 2018. Phenology and pollination ecology of Prosopis rubriflora (Leguminosae, Mimosoideae), a species from the semi-arid Brazilian Chaco. Brazilian Journal of Botany. 41: 103-115. https://doi.org/10.1007/s40415-017-0433-9
Singh, V. P.; Prasad, S. M.; Bosch, S. M.; Müller, M. 2017. Phytohormones and the regulation of stress tolerance in plants: current status and future directions. Frontiers in Plant Science. 8: 1871. https://doi.org/10.3389/fpls.2017.01871
Takahashi, H.; Kopriva, S.; Giordano, M.; Saito, H.; Hell, R. 2011. Sulfur assimilation in photosynthetic organisms: molecular functions and regulations of transporters and assimilatory enzymes. Annual Review of Plant Biology. 62: 157-184. https://doi.org/10.1146/annurev-arplant-042110-103921
van Zelm, E.; Zhang, Y.; Testerink, C. 2020. Salt tolerance mechanisms of plants. Annual Review of Plant Biology. 71:403-433. https://doi.org/10.1146/annurev-arplant-050718-100005
Wilkinson, S.; Kudoyarova, G. R.; Veselov, D. S.; Arkhipova, T. N.; Davies, W. J. 2012. Plant hormone interactions: innovative targets for crop breeding and management. Journal of Experimental Botany. 63: 3499-3509. https://doi.org/10.1093/jxb/ers148
Zagorchev, L.; Seal, C. E.; Kranner, I.; Odjakova, M. A. 2013. Central role for thiols in plant tolerance to abiotic stress. International Journal of Molecular Science. 14:7405-7432. https://doi. org/10.3390/ijms14047405

Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Aquellos autores/as que tengan publicaciones con esta revista, aceptan las Políticas Editoriales.










.jpg)



