Ammonium Sulfate Improves Nutrient Status and Photosynthetic Activity in Sorghum (Sorghum bicolor L.) Grown in Alkaline-Saline-Sodic Soils
DOI:
https://doi.org/10.48162/rev.39.212Keywords:
Sorghum bicolor, alkaline-saline-sodic soils (ASS), ammonium sulfate (AS) fertilization , urea (U), plant mineral nutrition, photosynthesisAbstract
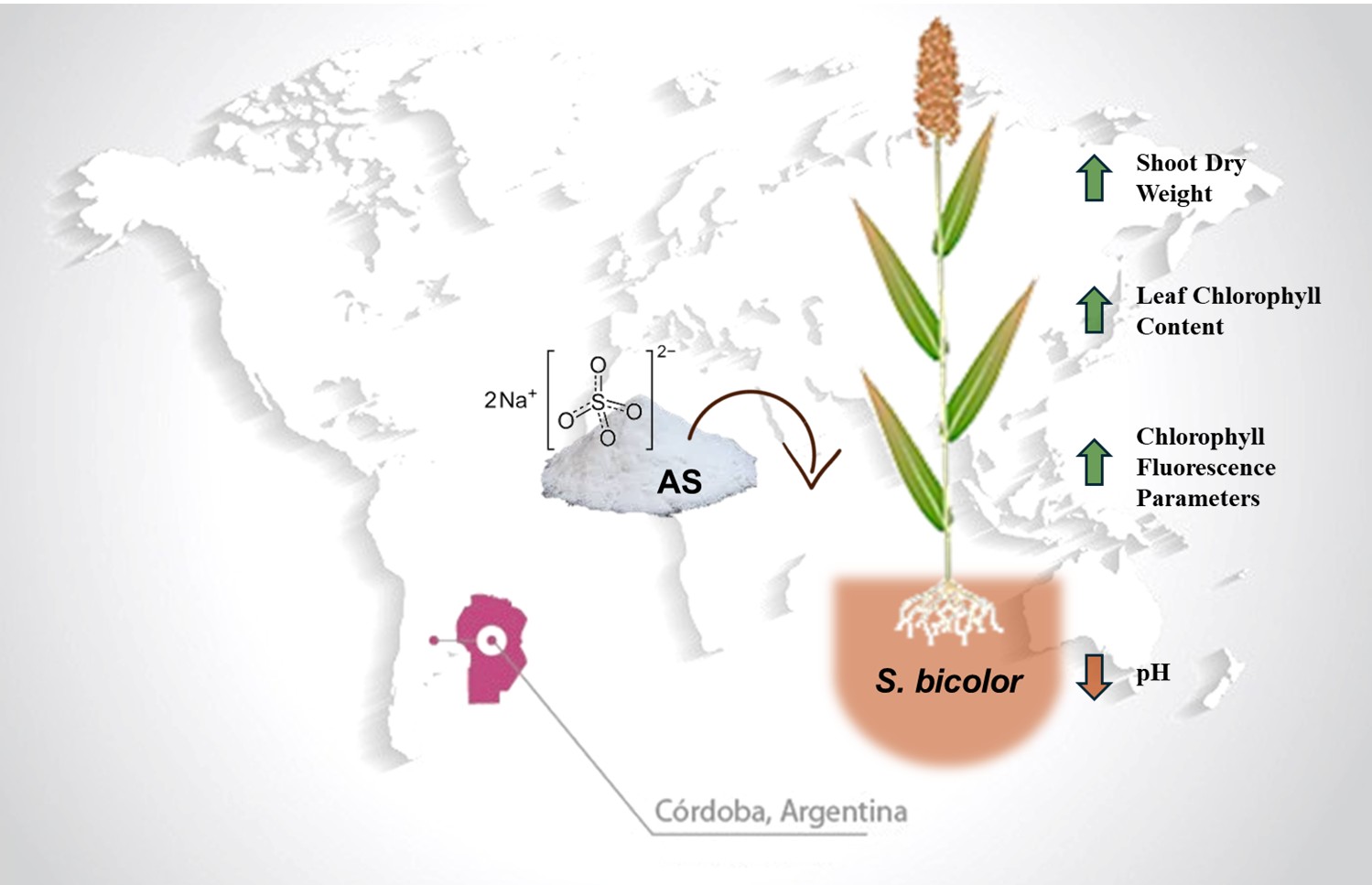
This study investigated whether fertilization with urea or ammonium sulfate could mitigate the growth limitations of sorghum imposed by alkaline-saline-sodic (ASS) soils. We hypothesized that the application of these nitrogen sources would improve the plant’s mineral status and photosynthetic activity. Experiments were conducted using three sorghum hybrids under both field and greenhouse conditions, comparing ASS soil to a non-ASS control. The treatments included fertilization with ammonium sulfate, urea, and a non-fertilized control. At the end of both experiments, soil and plant samples were analyzed to assess soil pH, nutrient content, and chlorophyll fluorescence. Our results demonstrate that ammonium sulfate, but not urea, significantly improved plant mineral status by inducing acidification in the ASS soil. This improved status led to increased photosynthetic rates and biomass production compared to unfertilized controls. These findings position ammonium sulfate as an effective tool for improving sorghum growth and productivity in challenging marginal soils.
Highlights:
- Ammonium sulfate (AS) fertilization reduced alkaline-saline-sodic (ASS) soil pH by approximately 1 unit under field conditions, whereas urea (U) shows no significant effect on soil pH.
- Unlike U, fertilization with AS improves the mineral status of sorghum by increasing the uptake of essential macro- and micronutrients, including N, P, K, and Ca, which are limited in AAS soils.
- The damage to the photosystem II electron transport chain was mitigated by AS treatment, demonstrating protection of the photosynthetic machinery from ASS stress.
- Hybrid-specific responses revealed that moderately tolerant genotypes (163 and SIP) achieved biomass production comparable to control soil conditions when treated with AS, highlighting the importance of combining appropriate genotypes with fertilization strategies.
Downloads
References
Adams, F. (1984). Crop response to lime in the southern United States. In F. Adams (Ed.). Soil acidity and liming, 12, 211-265. American Society of Agronomy. https://doi.org/10.2134/agronmonogr12.2ed.c5
Alkhader, A. M., Qaryouti, M. M., & Okasheh, T. Y. M. (2019). Effect of nitrogen on yield, quality, and irrigation water use efficiency of drip fertigated grafted watermelon (Citrullus lanatus) grown on a calcareous soil. Journal of Plant Nutrition, 42(1), 1-12. https://doi.org/10.1080/01904167.2019.1568464
Bernardo, L. M., Clark, R. B., & Maranville, J. W. (1984). Nitrate/ammonium ratio effects on mineral element uptake by sorghum. Journal of Plant Nutrition, 7(10), 1401-1414. https://doi.org/10.1080/01904168409363290
Blair, G. J., Miller, M. H., & Mitchell, W. A. (1970). Nitrate and ammonium as sources of nitrogen for corn and their influence on the uptake of other ions. Agronomy Journal, 62(4), 530-532. https://doi.org/10.2134/agronj1970.00021962006200040031x
Bontpart, T., Weiss, A., Vile, D., Gérard, F., Lacombe, B., Reichheld, J. P., & Mari, S. (2024). Growing on calcareous soils and facing climate change. Trends in Plant Science. https://doi.org/10.1016/j.tplants.2024.03.013
Bordenave, C. D., Rocco, R., Maiale, S. J., Campestre, M. P., Ruiz, O. A., Rodríguez, A. A., & Menéndez, A. B. (2019). Chlorophyll a fluorescence analysis reveals divergent photosystem II responses to saline, alkaline, and saline-alkaline stresses in the two Lotus japonicus model ecotypes MG20 and Gifu-129. Acta Physiologiae Plantarum, 41(9), 167. https://doi.org/10.1007/s11738-019-2956-0
Cavalcante, T. J., Castoldi, G., Rodrigues, C. R., Nogueira, M. M., & Albert, A. M. (2018). Marcha de absorção de macro- e micronutrientes na biomassa de sorgo. Pesquisa Agropecuária Tropical, 48(4), 364-373. https://doi.org/10.1590/1983-40632018v4851874
Chaves, M. M., Flexas, J., & Pinheiro, C. (2009). Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Annals of Botany, 103(4), 551-560. https://doi.org/10.1093/aob/mcn125
Chien, S. H., Gearhart, M. M., & Villagarcía, S. (2011). Comparison of ammonium sulfate with other nitrogen and sulfur fertilizers in increasing crop production and minimizing environmental impact: A review. Soil Science, 176(7), 327-335. https://doi.org/10.1097/SS.0b013e31821f0816
Clark, R. B., & Baligar, V. C. (2000). Acidic and alkaline soil constraints on plant mineral nutrition. Plant-Environment Interactions, 133–177.
De Souza Miranda, R., Gomes-Filho, E., Prisco, J. T., & Alvarez-Pizarro, J. C. (2016). Ammonium improves tolerance to salinity stress in Sorghum bicolor plants. Plant Growth Regulation, 78(1), 121-131. https://doi.org/10.1007/s10725-015-0079-1
Fageria, N. K., dos Santos, A. B., & Moraes, M. F. (2010). Influence of urea and ammonium sulfate on soil acidity indices and lowland rice production. Communications in Soil Science and Plant Analysis, 41(13), 1565-1575. https://doi.org/10.1080/00103624.2010.485237
Flexas, J., Bota, J., Loreto, F., Cornic, G., & Sharkey, T. D. (2004). Diffusive and metabolic limitations to photosynthesis under drought and salinity in C3 plants. Plant Biology, 6(3), 269-279. https://doi.org/10.1055/s-2004-820867
Gahoonia, T. S., Claassen, N., & Jungk, A. (1992). Mobilization of phosphate in different soils by ryegrass supplied with ammonium or nitrate. Plant and Soil, 140(2), 241-248. https://doi.org/10.1007/BF00010600
Giorda, L., & Ortiz, S. (2012). El cultivo de sorgo en la región semiárida central de Córdoba: aportes al sistema de producción ganadera. INTA Manfredi.
González, F. J., Martínez, A. L., & Pérez, R. M. (2019). Efecto del yeso agrícola en la mejora de los suelos salinos y en el rendimiento de los cultivos. Revista de Ciencia y Tecnología Agrícola, 12(2), 85-94. https://doi.org/10.1234/rcta.2019.12.2.85
Hadi, M. R., & Karimi, N. (2012). El papel del calcio en la tolerancia a la sal de las plantas. Revista de Nutrición Vegetal, 35(6), 905-927. https://doi.org/10.1080/01904167.2012.717158
Jacoby, R., Peukert, M., Succurro, A., Koprivova, A., & Kopriva, S. (2017). The role of soil microorganisms in plant mineral nutrition-current knowledge and future directions. Frontiers in Plant Science, 8. https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2017.01617
Kalaji, H. M., Bąba, W., Gediga, K., Goltsev, V., Samborska, I. A., Cetner, M. D., Dimitrova, S., Piszcz, U., Bielecki, K., Karmowska, K., Dankov, K., & Kompała-Bąba, A. (2018). Chlorophyll fluorescence as a tool for nutrient status identification in rapeseed plants. Photosynthesis Research, 136(3), 329-343. https://doi.org/10.1007/s11120-017-0467-7
Kissel, D. E., Cabrera, M. L., & Paramasivam, S. (2008). Ammonium, ammonia, and urea reactions in soils. In J. S. Schepers & W. R. Raun (Eds.). Nitrogen in agricultural systems (p. 101-155). American Society of Agronomy. https://doi.org/10.2134/agronmonogr49.c4
Lawlor, D. W., & Cornic, G. (2002). Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant, Cell & Environment, 25(2), 275-294. https://doi.org/10.1046/j.0016-8025.2001.00814.x
Liu, C., Gu, W., Li, B., Feng, Y., Liu, C., Shi, X., & Zhou, Y. (2024). Screening key sorghum germplasms for low-nitrogen tolerance at the seedling stage and identifying from the carbon and nitrogen metabolism. Frontiers in Plant Science, 15. https://doi.org/10.3389/fpls.2024.1340509
Marschner, P., & Rengel, Z. (2012). Nutrient availability in soils. In P. Marschner (Ed.). Marschner’s mineral nutrition of higher plants (3rd ed., p. 315-330). Academic Press. https://doi.org/10.1016/B978-0-12-384905-2.00012-1
Naeem, A., Deppermann, P., & Mühling, K. H. (2023). Ammonium fertilization enhances nutrient uptake, specifically manganese and zinc, and growth of maize in unlimed and limed acidic sandy soil. Nitrogen, 4(2), 239-252. https://doi.org/10.3390/nitrogen4020017
Pierre, W. H. (1928). Nitrogenous fertilizers and soil acidity: I. Effect of various nitrogenous fertilizers on soil reaction. Agronomy Journal, 20(3), 254-269. https://doi.org/10.2134/agronj1928.00021962002000030006x
RAGT Semillas Argentina, (2025, febrero 24). Sorgo, una herramienta versátil para la conversión de reservas forrajeras. RAGT Semillas Argentina. https://ragt-semillas.com.ar/sorgo-unaherramienta-versátil-para-la-conversión-de-reservas-forrajeras
Ranaweera, Y., & De Silva, R. C. L. (2021). A laboratory study on the effects of fertilizer addition on mobilization, percolation, and leaching of calcium ions in soils of the Anuradhapura district. International Conference on Applied and Pure Sciences.
Rengasamy, P. (2010). Soil processes affecting crop production in salt-affected soils. Functional Plant Biology, 37(7), 613-620. https://doi.org/10.1071/FP09249
Sachdev, S., Ansari, S. A., Ansari, M. I., Fujita, M., & Hasanuzzaman, M. (2021). Abiotic stress and reactive oxygen species: Generation, signaling, and defense mechanisms. Antioxidants, 10(2). https://doi.org/10.3390/antiox10020277
Saqib, M., Abbas, G., & Akhtar, J. (2020). Root-mediated acidification and resistance to low calcium improve wheat (Triticum aestivum) performance in saline-sodic conditions. Plant Physiology and Biochemistry, 156, 201-208. https://doi.org/10.1016/j.plaphy.2020.09.001
Sharma, S., Gupta, N., Chakkal, A. S., Sharma, N., Alamri, S., Siddiqui, M. H., & Haider, F. U. (2023). Changes in enzyme activities in salt-affected soils during the incubation study of diverse particle sizes of rice straw. Agriculture, 13(9), Article 1694. https://doi.org/10.3390/agriculture13091694
Sparks, D. L. (2003). The chemistry of saline and sodic soils. In D. L. Sparks (Ed.), Environmental soil chemistry (2nd ed., p. 285-300). Academic Press. https://doi.org/10.1016/B978-012656446-4/50010-4
Stirbet, A., & Govindjee, G. (2011). On the relation between the Kautsky effect (chlorophyll a fluorescence induction) and Photosystem II: Basics and applications of the OJIP fluorescence transient. Journal of Photochemistry and Photobiology B: Biology, 104(1-2), 236-257. https://doi.org/10.1016/j.jphotobiol.2010.12.010
Strasser, R. J., Tsimilli-Michael, M., & Srivastava, A. (2004). Analysis of the chlorophyll a fluorescence transient. In G. C. Papageorgiou & Govindjee (Eds.). Chlorophyll a fluorescence: A signatura of photosynthesis (p. 321-362). Springer Netherlands. https://doi.org/10.1007/978-1-4020-3218-9_12
Sui, N., Yang, Z., Liu, M., & Wang, B. (2015). Identification and transcriptomic profiling of genes involved in increasing sugar content during salt stress in sweet sorghum leaves. BMC Genomics, 16(1), 534. https://doi.org/10.1186/s12864-015-1760-5
Suzuki, Y., Ohsaki, K., Takahashi, Y., Wada, S., Miyake, C., & Makino, A. (2023). The behavior of photosystems II and I is modulated by N partitioning to Rubisco in mature leaves acclimated to low N levels and in senescent leaves in rice. Plant and Cell Physiology, 64(1), 55-63. https://doi.org/10.1093/pcp/pcac139
Tupa-Andrade, L.; Caro-Castro, J.; León-Quispe, J. (2025). Isolation of Polyhydroxyalkanoate (PHA)-Producing Azotobacter spp. from Crop Rhizospheres Located in Lima, Peru. Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. 57(2): 45-56. DOI: https://doi.org/10.48162/rev.39.170.
Wakeel, A. (2013). Potassium–sodium interactions in soil and plant under saline-sodic conditions. Journal of Plant Nutrition and Soil Science, 176(3), 344-354. https://doi.org/10.1002/jpln.201200417
Zhang, H., Xu, N., Wu, X., Wang, J., Ma, S., Li, X., & Sun, G. (2018). Effects of four types of sodium salt stress on plant growth and photosynthetic apparatus in sorghum leaves. Journal of Plant Interactions, 13(1), 506-513. https://doi.org/10.1080/17429145.2018.1526978.
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Aquellos autores/as que tengan publicaciones con esta revista, aceptan las Políticas Editoriales.










.jpg)



