Optimization of Antioxidant Extraction from Mutisia friesiana, Native Plant of the Quebrada and Puna Regions of Argentina
DOI:
https://doi.org/10.48162/rev.39.203Palabras clave:
Mutisia friesiana, antioxidantes, compuestos fenólicos, extracción asistida por ultrasonido, metodología de superficie de respuestaResumen
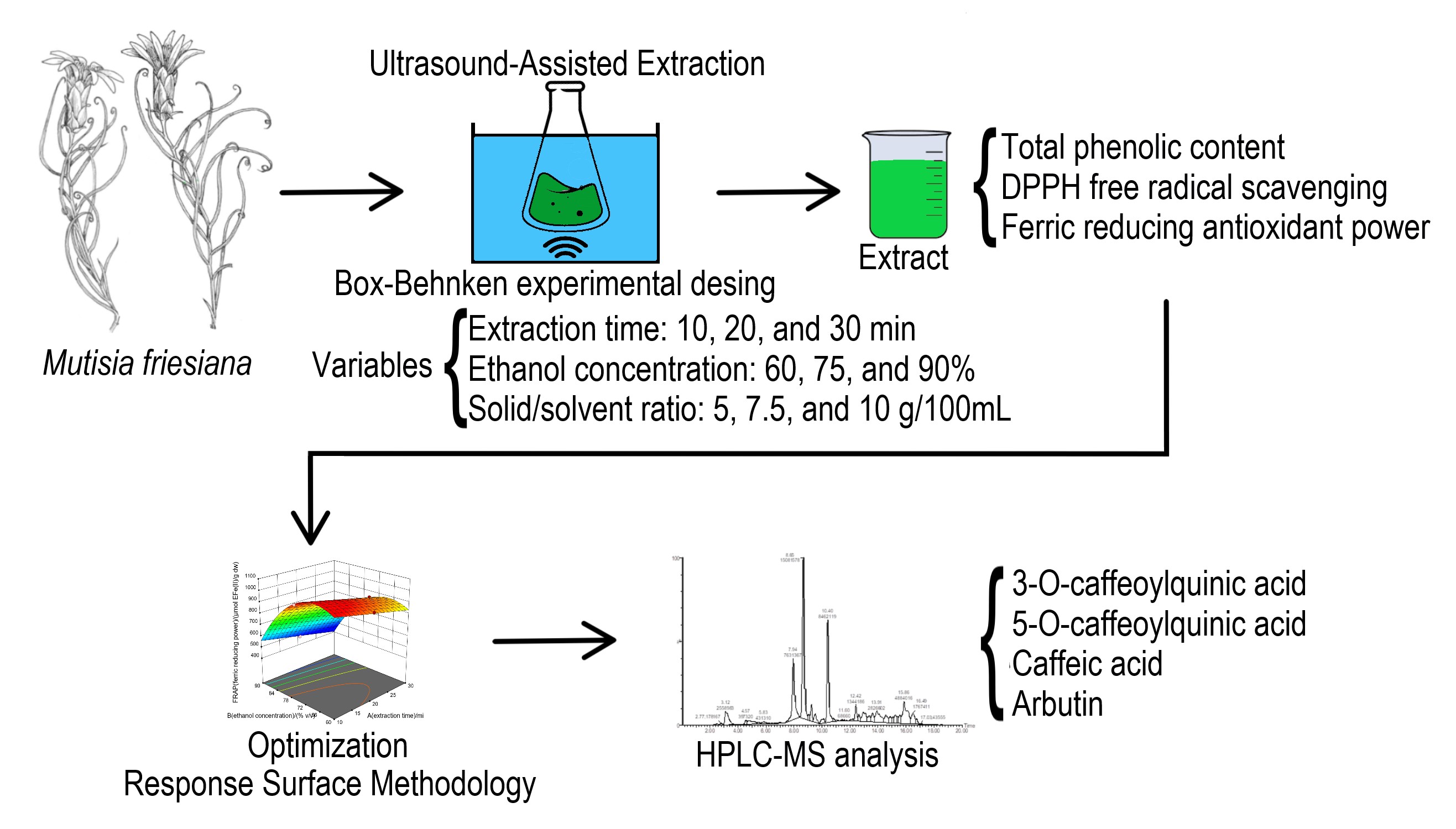
The study determined the optimal conditions for extracting antioxidant phenols from Mutisia friesiana (Asteraceae) using ultrasound-assisted extraction. We evaluated extraction time, ethanol concentration, and solid/solvent ratio, applying a Box-Behnken experimental design. Response variables were total phenolic content (TPC), DPPH free radical scavenging activity (EC50), and ferric reducing antioxidant power (FRAP). All extraction variables were optimized by the response surface methodology. The optimal conditions resulted in 15 minutes of extraction, 65% ethanol, and a solid/solvent ratio of 5 g/100 mL. Under these conditions, TPC, EC50, and FRAP values were 54.24 mg GAE/g dw, 31.40 µg/mL, and 965.32 µmol EFe(II)/g dw, respectively. Extract HPLC–MS analysis under optimal conditions identified four phenolic compounds, previously reported for their antioxidant properties. This study provides valuable insights into optimal extraction parameters of bioactive compounds from M. friesiana, positioning the species as a promising natural antioxidants source for the food industry, particularly meat products.
Highlights:
- Ultrasound-assisted extraction (UAE) was optimized to recover antioxidant phenols from Mutisia friesiana using response surface methodology.
- Optimal conditions yielded an extract with high antioxidant activity.
- HPLC-MS identified four antioxidant phenols in the optimized extract.
Descargas
Citas
Barboza, G. E.; Cantero, J. J.; Núñez, C.; Pacciaroni, A.; Ariza Espinar, L. 2009. Medicinal plants: A general review and a phytochemical and ethnopharmacological screening of the native Argentine Flora. Kurtziana. 34(1-2): 7-365.
Belwal, T.; Dhyani, P.; Bhatt, I. D.; Rawal, R. S.; Pande, V. 2016. Optimization extraction conditions for improving phenolic content and antioxidant activity in Berberis asiatica fruits using response surface methodology (RSM). Food Chemistry. 207: 115-124. https://doi. org/10.1016/j.foodchem.2016.03.081
Benzie, I. F.; Strain, J. J. 1996. The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: the FRAP assay. Analytical Biochemistry. 239(1): 70-76. https://doi.org/10.1006/ abio.1996.0292
Bouafia, M.; Colak, N.; Ayaz, F. A.; Benarfa, A.; Harrat, M.; Gourine, N.; Yousfi, M. 2021 The optimization of ultrasonic-assisted extraction of Centaurea sp. antioxidative phenolic compounds using response surface methodology. Journal of Applied Research on Medicinal and Aromatic Plants. 25:100330. https://doi.org/10.1016/j.jarmap.2021.100330
Brand-Williams, W.; Cuvelier, M. E.; Berset, C. L. W. T. 1995. Use of a free radical method to evaluate antioxidant activity. LWT-Food Science and Technology. 28(1): 25-30. https://doi. org/10.1016/S0023-6438(95)80008-5
Bremer, K. 1994. Asteraceae. Cladistics and classification. Timber Press.
Cabrera, A. L. 1965. Revisión del género Mutisia (Compositae). Opera Lilloana 13. San Miguel de Tucumán. Fundación Miguel Lillo.
Catalano, S.; Cioni, P. L.; Flamini, G.; De Feo, V.; Morelli, I. 1995. Chemical investigation of the aerial parts of Mutisia acuminata. International Journal of Pharmacognosy. 33(1): 73-74. https:// doi.org/10.3109/13880209509088152
Catalano, S.; Cioni, P. L.; Panizzi, L.; Morelli, I. 1998. Antimicrobial activity of extracts of Mutisia acuminata var. acuminata. Journal of Ethnopharmacology. 59(3): 207-209. https://doi. org/10.1016/S0378-8741(97)00113-X
Chirinos, R.; Pedreschi, R.; Rogez, H.; Larondelle, Y.; Campos, D. 2013. Phenolic compound contents and antioxidant activity in plants with nutritional and/or medicinal properties from the Peruvian Andean region. Industrial Crops and Products. 47: 145-152. https://doi. org/10.1016/j.indcrop.2013.02.025
Chui Tinco, G. 2021. Estudio fitoquímico preliminar de cinco especies vegetales del altiplano sur y determinación de componentes mayoritarios de la especie vegetal Lampaya castellani Moldenke. Tesis de grado en Licenciatura en Ciencias Químicas. Facultad de Ciencias Puras y Naturales. Universidad Mayor de San Andrés. La Paz. Bolivia.
Cizmarova, B.; Hubkova, B.; Bolerazska, B.; Marekova, M.; Birkova, A. 2020. Caffeic acid: A brief overview of its presence, metabolism, and bioactivity. Bioactive Compounds in Health and Disease. 3(4): 74-81. https://doi.org/10.31989/bchd.v3i4.692
Daily, A.; Seligmann, O.; Nonnenmacher, G.; Fessler, B.; Wong, S. M.; Wagner, H. 1988. New chromone, coumarin, and coumestan derivatives from Mutisia acuminata var. hirsuta. Planta Medica. 54(1): 50-52. https://doi.org/10.1055/s-2006-962334
Del Vitto, L. A.; Petenatti, E. M. 2009. Asteráceas de importancia económica y ambiental: Primera parte. Sinopsis morfológica y taxonómica, importancia ecológica y plantas de interés industrial. Multequina. 18(2): 87-115.
Dirar, A. I.; Alsaadi, D. H. M.; Wada, M.; Mohamed, M. A.; Watanabe, T.; Devkota, H. P. 2019. Effects of extraction solvents on total phenolic and flavonoid contents and biological activities of extracts from Sudanese medicinal plants. South African Journal of Botany. 120: 261-267. https://doi.org/10.1016/j.sajb.2018.07.003
Fang, X.; Wang, J.; Wang, Y.; Li, X.; Zhou, H.; Zhu, L. 2014 Optimization of ultrasonic-assisted extraction of wedelolactone and antioxidant polyphenols from Eclipta prostrate L using response surface methodology. Separation and Purification Technology. 138: 55-64. https://doi.org/10.1016/j.seppur.2014.10.007
Fernández-Galleguillos, C.; Jiménez-Aspee, F.; Mieres-Castro, D.; Rodríguez-Núñez, Y. A.; Gutiérrez, M.; Guzmán, L.; Echeverría, J.; Sandoval-Yañez, C.; Forero-Doria, O. 2023. Phenolic Profile and Cholinesterase Inhibitory Properties of Three Chilean Altiplano Plants: Clinopodium gilliesii (Benth.) Kuntze [Lamiaceae], Mutisia acuminata Ruiz & Pav. var. hirsuta (Meyen) Cabrera, and Tagetes multiflora (Kunth) [Asteraceae]. Plants. 12(4): 819. https://doi.org/10.3390/plants12040819
Flores, Y.; Rodrigo, G.; Mollinedo, P.; Akesson, B.; Sterner, O.; Almanza, G. R. 2009. A 5-methylcoumarin glucoside and a coumestan derivative from Mutisia orbignyana. Revista Boliviana Química. 26(1): 21-26.
Ghitescu, R. E.; Volf, I.; Carausu, C.; Bühlmann, A. M.; Gilca, I. A.; Popa, V. I. 2015. Optimization of ultrasound-assisted extraction of polyphenols from spruce wood bark. Ultrasonics Sonochemistry. 22: 535-541. https://doi.org/10.1016/j.ultsonch.2014.07.013
Juárez, B. E.; Mendiondo, M. E. 2003. Flavonoids from Mutisia acuminata. Pharmaceutical Biology. 41(4): 291-292. https://doi.org/10.1076/phbi.41.4.291.15669
Katinas, L.; Pruski, J.; Sancho, G.; Tellería, M. C. 2008. The subfamily Mutisioideae (Asteraceae). The Botanical Review. 74(4): 469-716. https://doi.org/10.1007/s12229-008-9016-6
Kumar, K.; Srivastav, S.; Sharanagat, V. S. 2021. Ultrasound assisted extraction (UAE) of bioactive compounds from fruit and vegetable processing by-products: A review. Ultrasonics Sonochemistry. 70:105325. https://doi.org/10.1016/j.ultsonch.2020.105325
Llorent-Martínez, E. J.; Zengin, G.; Sinan, K. I.; Polat, R.; Canlı, D.; Picot-Allain, M. C. N.; Mahomoodally, M. F. 2020. Impact of different extraction solvents and techniques on the biological activities of Cirsium yildizianum (Asteraceae: Cynareae). Industrial Crops and Products. 144:112033. https://doi.org/10.1016/j.indcrop.2019.112033
Lopez, V. L.; Cellini, J. M. 2022. Plantas medicinales asociadas a bosques de Polylepis tarapacana del altiplano jujeño: Riqueza y cobertura en gradientes geográficos, topográficos y de estructura forestal. Ecología Austral. 32(3): 894-907. https://doi.org/10.25260/EA.22.32.3.0.1905
Lourenço, S. C.; Moldão-Martins, M.; Alves, V. D. 2019. Antioxidants of natural plant origins: From sources to food industry applications. Molecules. 24(22): 4132. https://doi.org/10.3390/ molecules24224132
Montgomery, D. C. 2004. 2° ed. Métodos de superficies de respuesta y otros enfoques para la optimización de procesos. In John Wiley & Sons (Ed.). Diseño y análisis de experimentos. Limusa. 427-510.
Moreira-Muñoz, A.; Scherson, R. A.; Luebert, F.; Román, M. J.; Monge, M.; Diazgranados, M.; Silva, H. 2020. Biogeography, phylogenetic relationships and morphological analyses of the South American genus Mutisia Lf (Asteraceae) shows early connections of two disjunct biodiversity hotspots. Organisms Diversity & Evolution. 20: 639-656. https://doi. org/10.1007/s13127-020-00454-z
Olszowy, M. 2019. What is responsible for antioxidant properties of polyphenolic compounds from plants? Plant Physiology and Biochemistry. 144: 135-143. https://doi.org/10.1016/j. plaphy.2019.09.039
Palmieri, S.; Pellegrini, M.; Ricci, A.; Compagnone, D.; Lo Sterzo, C. 2020. Chemical composition and antioxidant activity of thyme, hemp and coriander extracts: A comparison study of maceration, Soxhlet, UAE and RSLDE techniques. Foods. 9(9): 1221. https://doi. org/10.3390/foods9091221
Pham, D. C.; Nguyen, H. C.; Nguyen, T. H. L.; Ho, H. L.; Trinh, T. K.; Riyaphan, J.; Weng, C. F. 2020. Optimization of ultrasound‐assisted extraction of flavonoids from Celastrus hindsii leaves using response surface methodology and evaluation of their antioxidant and antitumor activities. BioMed Research International. 2020(1): 3497107. https://doi. org/10.1155/2020/3497107
Pillco Paulo, D. 2023. Perfil de componentes fenólicos mediante cromatografía líquida y espectrometría de masas de alta resolución en tándem, MS/MS, de cinco especies del género Mutisia. Tesis de grado en Química. Facultad de Ciencias Químicas, Físicas y Matemáticas. Universidad Nacional de San Antonio Abad del Cusco. http:// hdl.handle.net/20.500.12918/7286
Singleton, V. L.; Rossi, J. A. 1965. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. American Journal of Enology and Viticulture. 16(3): 144-158. https://doi. org/10.5344/ajev.1965.16.3.144
Sobhani, A.; Noormohammadi, N.; Moradi, K.; Ebrahimi, M.; Khanahmadi, M. 2022. Optimization of heat and ultrasound assisted extraction of bioactive compounds from Echinacea purpurea using response surface methodology. Journal of Applied Research on Medicinal and Aromatic Plants. 31:100399. https://doi.org/10.1016/j.jarmap.2022.100399
Sova, M. 2012. Antioxidant and antimicrobial activities of cinnamic acid derivatives. Mini-Reviews in Medicinal Chemistry. 12(8): 749-767. https://doi.org/10.2174/138955712801264792
Sova, M.; Saso, L. 2020. Natural sources, pharmacokinetics, biological activities and health benefits of hydroxycinnamic acids and their metabolites. Nutrients. 12(8): 2190. https://doi. org/10.3390/nu12082190
Tirado-Kulieva, V. A.; Oblitas, J.; León-Roque, N.; Castro, W. 2026. Physicochemical Properties, Bioactive Compounds, and Antioxidant Activity of Andean Fruits: Optimization of Extraction by Response Surface Methodology. Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina. 58(1): e9133.
Villamil-Galindo, E.; Piagentini, A. 2024. Green solvents for the recovery of phenolic compounds from strawberry (Fragaria x ananassa Duch) and apple (Malus domestica) agro-industrial bio-wastes. Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina. 56(1): 149-160. DOI: https://doi.org/10.48162/rev.39.131
Viturro, C. I.; Molina, A.; Schmeda‐Hirschmann, G. 1999. Free radical scavengers from Mutisia friesiana (Asteraceae) and Sanicula graveolens (Apiaceae). Phytotherapy Research. 13(5): 422-424. https://doi.org/10.1002/(SICI)1099-1573(199908/09)13:5%3C422::AID-PTR462%3E3.0.CO;2-M
Viturro, C. I.; de la Fuente, J. 2000. Chemical study of the essential oil of Mutisia friesiana. Molecules. 5(3): 568-570. https://doi.org/10.3390/50300568
Viturro, C. I.; Maier, M. S.; Stortz, C. A.; de la Fuente, J. R. 2001. Antifungal diastereomeric furanones from Mutisia friesiana: structural determination and conformational analysis. Tetrahedron: Asymmetry. 12(7): 991-998. https://doi.org/10.1016/S0957-4166(01)00163-X
Viturro, C. I.; de la Fuente, J. R.; Maier, M. S. 2003. Antifungal methylphenone derivatives and 5-methylcoumarins from Mutisia friesiana. Zeitschrift für Naturforschung C. 58(7-8): 533-540. https://doi.org/10.1515/znc-2003-7-815
Viturro, C. I.; de la Fuente, J. R.; Maier, M. S. 2004. 5-Methylcoumaranones from Mutisia friesiana and their bioactivity. Journal of Natural Products. 67(5): 778-782. https://doi.org/10.1021/ np0304058
Viturro, C. I.; de la Fuente, J. R.; Maier, M. S. 2005. One new prenylated furanone and other non- polar constituents from Mutisia friesiana. Zeitschrift für Naturforschung B. 60(5): 585-589. https://doi.org/10.1515/znb-2005-0519
Yancheshmeh, B. S.; Panahi, Y.; Allahdad, Z.; Abdolshahi, A.; Zamani, Z. 2022. Optimization of ultrasound-assisted extraction of bioactive compounds from Achillea kellalensis using response surface methodology. Journal of Applied Research on Medicinal and Aromatic Plants. 28:100355. https://doi.org/10.1016/j.jarmap.2021.100355
Descargas
Publicado
Cómo citar
Número
Sección
Licencia

Esta obra está bajo una licencia internacional Creative Commons Reconocimiento-NoComercial-CompartirIgual 3.0.
Aquellos autores/as que tengan publicaciones con esta revista, aceptan las Políticas Editoriales.










.jpg)



