Enterococcus gallinarum CRL 1826 as a Probiotic for Ranaculture: in vitro Safety, Technological, and Physiological Properties
DOI:
https://doi.org/10.48162/rev.39.201Palabras clave:
Enterococcus gallinarum, características de seguridad, liofilización, probióticos para acuicultura, rana toroResumen
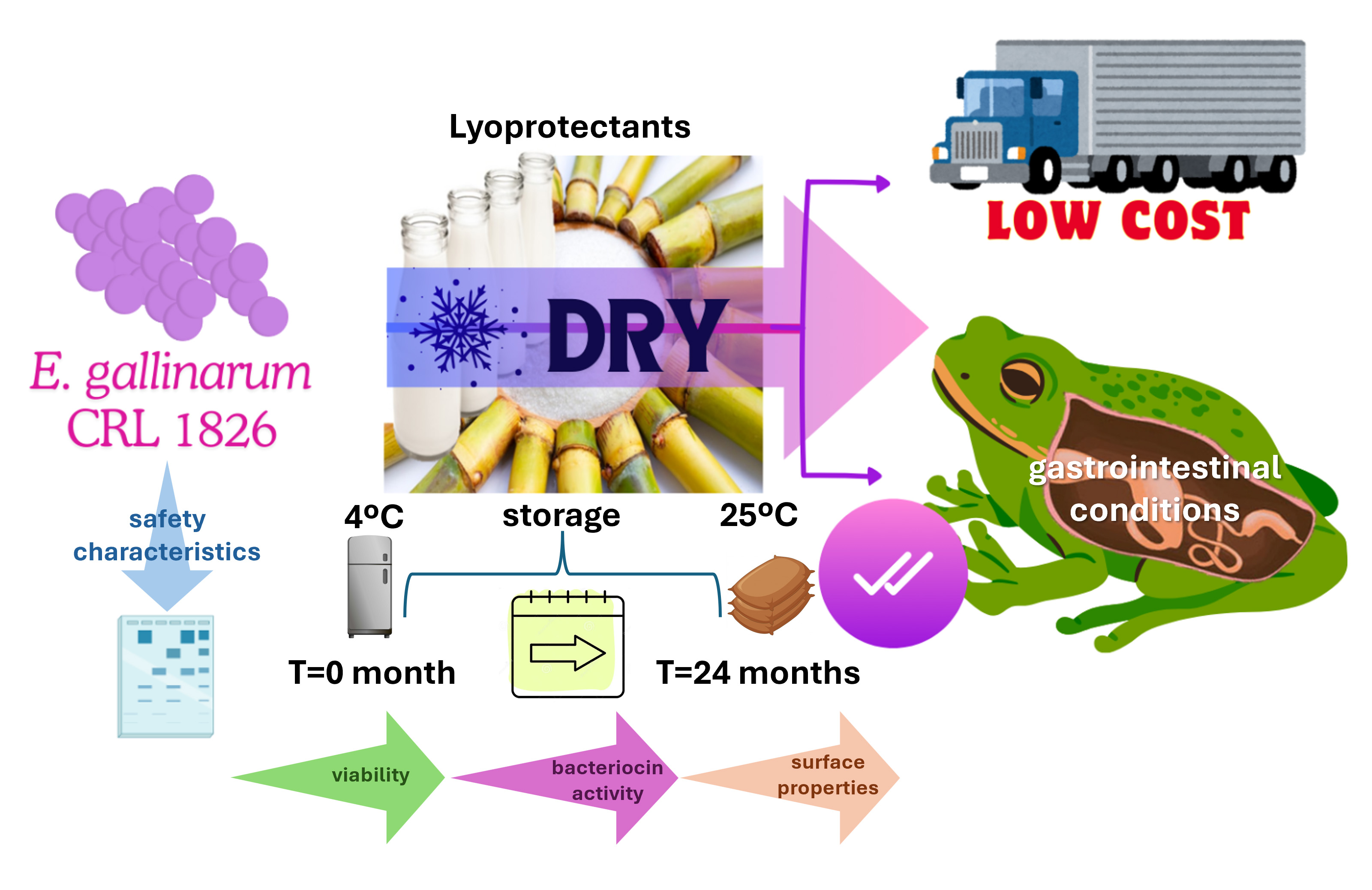
This study aimed to progress in designing a probiotic containing autochthonous Enterococcus gallinarum CRL 1826 for application during the life cycle of Lithobates catesbeianus in hatchery conditions. We assessed bacterial resistance to chemotherapeutics used in ranaculture, the presence of genes encoding virulence factors (VF) and vancomycin resistance, and bacterial survival and maintenance of beneficial properties after freeze-drying and storage. The strain exhibited resistance to antiseptics, sensitivity to most chemotherapeutics, presence of vanC, and absence of VF genes. It demonstrated resistance to freeze-drying and the highest survival when using skim milk+sucrose and storage at 4°C for 24 months. It also displayed bacteriocin activity against Listeria monocytogenes. Pre-lyophilized and lyophilized cultures grew/resisted individual gastrointestinal conditions and simulated gastrointestinal digestion, keeping bacteriocin activity and surface properties. For the first time, we demonstrated that E. gallinarum CRL 1826 is a safe bacterium with technological and physiological properties that would allow bullfrog gut colonization. These studies are essential for progressing towards selecting E. gallinarum CRL 1826 as a probiotic to prevent epizootics during bullfrog breeding and control foodborne bacteria, potentially improving growth performance of L. catesbeianus.
Highlights:
- Enterococcus gallinarum CRL 1826 was sensitive to chemotherapics used in ranaculture.
- The strain did not display virulence and transferable vancomycin resistance genes.
- The dried strain showed great survival and bacteriocin activity for 24 months’ storage.
- The LAB resisted the gastrointestinal conditions and kept its bacteriocin activity.
Descargas
Citas
Anglenius, H.; Mäkivuokko, H.; Ahonen, I.; Forssten, S. D.; Wacklin, P.; Mättö, J.; Lahtinen, S.; Lehtoranta, L.; Ouwehand, A. C. 2023. In vitro screen of lactobacilli strains for gastrointestinal and vaginal benefits. Microorganisms. 11(2): 329. https://doi. org/10.3390/microorganisms11020329
Barzegar, H.; Alizadeh Behbahani, B.; Falah, F. 2021. Safety, probiotic properties, antimicrobial activity, and technological performance of Lactobacillus strains isolated from Iranian raw milk cheeses. Food Sci. Nutr. 9(8): 4094-4107. https://doi.org/10.1002/fsn3.2365
Berg, G.; Rybakova, D.; Fischer, D.; Cernava, T.; Vergès, M. C. C.; Charles, T.; Chen, X.; Cocolin, L.; Eversole, K.; Herrero Corral, G.; Kazou, M.; Kinkel, L.; Lange, L.; Lima, N.; Loy, A.; Macklin, J. A.; Maguin, E.; Mauchline, T.; McClure, R.; Mitter, B.; Ryan, M.; Sarand, I.; Smidt, H.; Schelkle, B; Roume, H.; Kiran, G. S.; Selvin, J.; Soares Correa de Souza, R.; van Overbeek, L; Singh, B. K.; Wagner, M.; Walsh, A.; Sessitsch, A.; Schloter, M. 2020. Microbiome definition re-visited: old concepts and new challenges. Microbiome. 8: 1-22. https://doi.org/10.1186/s40168- 020-00875-0
Bühler, M. I.; Sánchez Toranzo, G.; Zaltz, S. 2000. La ranicultura: una alternativa productiva. Top. Graph. Argentina.
Cai, X.; Wen, J. S.; Long, H.; Ren, W.; Zhang, X.; Huang, A. Y.; Xie, Z. Y. 2022. The probiotic effects, dose, and duration of lactic acid bacteria on disease resistance in Litopenaeus vannamei. Aquac. Rep. 26: 101299. https://doi.org/10.1016/j.aqrep.2022.101299
CDC. Vancomycin-Resistant Enterococci (VRE) and the Clinical Laboratory HAI CDC. 2020. https:// www.cdc.gov/hai/settings/lab/vreclinical-laboratory.html.
Clinical and Laboratory Standards Institute. Methods for antimicrobial dilution and disk susceptibility testing of infrequently isolated or fastidious bacteria 3rd ed. CLSI guideline M45. 2015. Clinical and Laboratory Standards Institute. Wayne. PA. USA.
Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing, 32nd ed. CLSI guideline M100. 2022. Wayne. PA. USA.
Crowe, J. H.; Crowe, L. M.; Carpenter, J. F.; Wistrom, C. A. 1987. Stabilization of dry phospholipid-bilayers and proteins by sugars. Biochem. J. 242(1): 1-10. https://doi.org/10.1042/ bj2420001
Dehkordi, R. A. F.; Karimi, I.; Karimi, B.; Eshkaftaki, R. G.; Abtahi, R.; Mohammadi, H. 2023. Effect of pancreatin on acute pancreatitis resulting from L-arginine administration in mice, a morpho-histopathological and biochemical study. Braz. J. Pharm. Sci. 59: e21494. https:// doi.org/10.1590/s2175-97902023e21494
Densmore, C. L.; Green, D. E. 2007. Diseases of amphibians. ILAR J. 48(3): 235-254. https://doi. org/10.1093/ilar.48.3.235
De Oliveira, D. M.; Forde, B. M.; Kidd, T. J.; Harris, P. N.; Schembri, M. A.; Beatson, S. A.; Paterson, D. L.; Walker, M. J. 2020. Antimicrobial resistance in ESKAPE pathogens. Microbiol. Rev. 33(3): e00181-19. https://doi.org/10.1128/cmr.00181-19
Di Rienzo, J. A.; Casanoves, F.; Balzarini, M. G.; Gonzalez, L.; Tablada, M. R. C. W.; Robledo, C. W. 2020. InfoStat versión 2020. Centro de Transferencia InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. URL http://www. infostat. com. ar
Escobar-Sánchez, M.; Carrasco-Navarro, U.; Juárez-Castelán, C.; Lozano-Aguirre, L.; Pérez-Chabela, M. L.; Ponce-Alquicira, E. 2022. Probiotic properties and proteomic analysis of Pediococcus pentosaceus 1101. Foods. 12(1): 46. https://doi.org/10.3390/ foods12010046
European Food Safety Authority Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). 2018.efsa.2018.5206
Fiocco, D.; Longo, A.; Arena, M. P.; Russo, P.; Spano, G.; Capozzi, V. 2020. How probiotics face food stress: They get by with a little help. Crit. Rev. Food Sci. Nutr. 60(9): 1552-1580. https:// doi.org/10.1080/10408398.2019.1580673
Garza, M.; Mohan, C. V.; Rahman, M.; Wieland, B.; Häsler B. 2019. The role of infectious disease impact in informing decision-making for animal health management in aquaculture systems in Bangladesh. Prev. Vet. Med. 167: 202-213. https://doi.org/10.1016/j. prevetmed.2018.03.004
Gloor, G. B.; Reid, G. 2016. Compositional analysis: a valid approach to analyze microbiome high-throughput sequencing data. Can. J. Microbiol. 62(8): 692-703. https://doi.org/10.1139/ cjm-2015-0821
Hadfield, C. A.; Whitaker, B. R. 2005. Amphibian emergency medicine and care. Seminars in avian and exotic pet medicine. 14(2): 79-89. https://doi.org/10.1053/j.saep.2005.04.003
Hidano, A.; Yamamoto, T.; Hayama, Y.; Muroga, N.; Kobayashi. S.; Nishida, T.; Tsutsui, T. 2015. Unraveling antimicrobial resistance genes and phenotype patterns among Enterococcus faecalis isolated from retail chicken products in Japan. PloS One. 10(3): e0121189. https:// doi.org/10.1371/journal.pone.0121189
Igbinosa, E. O.; Beshiru, A. 2019. Antimicrobial resistance, virulence determinants, and biofilm formation of Enterococcus species from ready-to-eat seafood. Front. Microbiol. 10: 728. https://doi.org/ 10.3389/fmicb.2019.00728
Indriani, S.; Sae Leaw, T.; Benjakul, S.; Quan, T. H.; Karnjanapratum, S.; Nalinanon, S. 2022. Impact of different ultrasound-assisted processes for preparation of collagen hydrolysates from Asian bullfrog skin on characteristics and antioxidative properties. Ultrason. Sonochem. 89: 106163. https://doi.org/ 10.1016/j.ultsonch.2022.106163
Jawan, R.; Abbasiliasi, S.; Tan, J. S.; Kapri, M. R.; Mustafa, S.; Halim, M. 2022. Influence of type and concentration of lyoprotectants storage temperature and storage duration on cell viability and antibacterial activity of freeze-dried lactic acid bacterium Lactococcus lactis Gh1. Dry. Technol. 40(9): 1774-1790. https://doi.org/ 10.1080/07373937.2021.1874968
Kandil, S.; El Soda, M. 2015. Influence of freezing and freeze drying on intracellular enzymatic activity and autolytic properties of some lactic acid bacterial strains. Adv. Microbiol. 5: 371-382. https://doi.org/ 10.4236/aim.2015.56039
Kang, C. H.; Gu, T.; So, J. S. 2018. Possible probiotic lactic acid bacteria isolated from oysters (Crassostrea gigas). Probiotics Antimicrob. Proteins. 10(4): 728-739. https://doi.org/ 10.1007/s12602-017-9315-5
Ko, H. I.; Jeong, C. H.; Hong, S. W.; Eun, J. B.; Kim, T. 2022. Optimizing conditions in the acid tolerance test for potential probiotics using response surface methodology. Microb. Spectrum. 10(4): e01625-22. https://doi.org/10.1128/spectrum.01625-22
Maji, U. J.; Mohanty, S.; Mahapatra, A. S.; Maiti, N. K. 2016. Diversity and probiotic potentials of putative lactic acid bacteria for application in freshwater aquaculture. Turkish J. Fish Aquat. Sci. 16: 805-818. https://doi.org/10.4194/1303-2712-v16_4_07
Melchior, S.; Marino, M.; Innocente, N.; Calligaris, S.; Nicoli, M. C. 2020. Effect of different biopolymer-based structured systems on the survival of probiotic strains during storage and in vitro digestion. J. Sci. Food Agric. 100: 3902-3909. https://doi.org/10.1002/ jsfa.10432
Mendes, S. D. N. C.; Esteves, C. M.; Mendes, J. A. V.; Feres, M.; Figueiredo, N.; de Miranda, T. S.; Shibli, J. A.; Figueiredo, L. C. 2023. Systemic antibiotics and chlorhexidine associated with periodontal therapy: Microbiological effect on intraoral surfaces and saliva. Antibiotics. 12(5): 847. https://doi.org/ 10.3390/antibiotics12050847
Minitab, LLC. 2018. Statistical (versión 2018) [Software]. www.minitab.com
Montel Mendoza, G.; Pasteris, S. E.; Ale, C. E.; Otero, M. C.; Bühler, M. I.; Nader Macías, M. E. 2012. Cultivable microbiota of Lithobates catesbeianus and advances in the selection of lactic acid bacteria as biological control agents in raniculture. Res. Vet. Sci. 93: 1160-1167. https://doi.org/ 10.1016/j.rvsc.2012.05.007
Montel Mendoza, G.; Pasteris, S. E.; Otero, M. C., Nader Macías, M. E. F. 2014. Survival and beneficial properties of lactic acid bacteria from raniculture subjected to freeze-drying and storage. J. Appl. Microbiol. 116: 157-166. https://doi.org/ 10.1111/jam.12359
Montel Mendoza, G.; Ale, C. E.; Nader Macías, M. E. F.; Pasteris, S. E. 2015. Characterization of a bacteriocin produced by Enterococcus gallinarum CRL 1826 isolated from captive bullfrog: evaluation of its mode of action against Listeria monocytogenes and Gram-negatives. J. Bioprocess Biotech. 5: 250-256. https://doi.org/ 10.4172/2155-9821.1000250
Niederle, M. V.; Bosch, J.; Ale, C. E.; Nader Macías, M. E.; Aristimuno Ficoseco, C.; Toledo, L. F.; Valenzuela, Sánchez, A.; Soto Azat, C.; Pasteris, S. E. 2019. Skin-associated lactic acid bacteria from North American bullfrogs as potential control agents of Batrachochytrium dendrobatidis. PLoS One. 14(9): e0223020. https://doi.org/ 10.1371/journal. pone.0223020
Pasteris, S. E.; Bühler, M. I.; Nader Macías, M. E. 2006. Microbiological and histological studies in farmed-bullfrog (Rana catesbeiana) displaying red-leg syndrome. Aquaculture. 251: 11-18. https://doi.org/ 10.1016/j.aquaculture.2005.05.007
Pasteris, S. E.; Roig Babot, G.; Otero, M. C.; Nader Macías, M. E. 2009. Beneficial properties of lactic acid bacteria isolated from a Rana castesbeiana hatchery. Aquac. Res. 40: 1605-1615. https://doi.org/10.1111/j.1365-2109.2009.0226.x
Pasteris, S. E.; Vera Pingitore, E.; Roig Babot, G.; Otero, M. C.; Bühler, M. I.; Nader Macías, M. E. 2009. Characterization of the beneficial properties of lactobacilli isolated from bullfrog (Rana catesbeiana) hatchery. Antonie van Leeuwenhoek. 95: 375-385. https://doi. org/10.1007/s10482-009-9329-4
Pasteris, S. E.; Montel Mendoza, G.; Llanos, R. J.; Pucci Alcaide, F.; Nader Macías, M. E. F. 2017. Preliminary assessment of in vivo safety of potentially probiotic lactic acid bacteria for American bullfrog culture. Aquac. Res. 48(5): 2157-2172. https://doi.org/ 10.1111/ are.13053
Pereira, S. A.; Jerônimo, G. T.; Marchiori, N. C.; Oliveira, H. M.; Jesus, G. F. A.; Schmidt, E. C.; Bouzon, Z. L.; Vieira, F. N.; Martins, M. L.; Mouriño, J. L. P. 2018. Tadpoles fed supplemented diet with probiotic bacterium isolated from the intestinal tract of bullfrog Lithobates catesbeianus: Haematology, cell activity and electron microscopy. Microb. Pathog. 114: 255-263. https:// doi.org/10.1016/j.micpath.2017.11.033
Pereira, W. A.; Mendonça, C. M. N.; Urquiza, A. V.; Marteinsson, V.; LeBlanc, J. G.; Cotter, P. D.; Villalobos, E. F.; Romero, J.; Oliveira, R. P. 2022. Use of probiotic bacteria and bacteriocins as an alternative to antibiotics in aquaculture. Microorganisms. 10(9): 1705. https://doi. org/10.3390/microorganisms10091705
Pospiech, A.; Neumann, B. 1995. A versatile quick-prep of genomic DNA from Gram-positive bacteria. Trends Genet. 11: 217-218.
Romyasamit, C.; Saengsuwan, P.; Boonserm, P.; Thamjarongwong, B.; Singkhamanan, K. 2022. Optimization of cryoprotectants for freeze-dried potential probiotic Enterococcus faecalis and evaluation of its storage stability. Dry. Technol. 40(11): 2283-2292. https://doi.org/1 0.1080/07373937.2021.1931294
Sahoo, T. K.; Jena, P. K.; Nagar, N.; Patel, A. K.; Seshadri, S. 2015. In vitro evaluation of probiotic properties of lactic acid bacteria from the gut of Labeo rohita and Catla catla. Probiotics Antimicrob. Proteins. 7(2): 126-136. https://doi.org/10.1007/s12602-015-9184-8
Salama, S. A. E. H. 2022. Bacterial pathogens causing the blue crab (Callinectes sapidus) mortality at Suez Canal (El-Temsah Lake) in Ismailia Governorate. Egypt J. Aquat. Biol. Fish. 26(2): 151-168. https://doi.org/10.21608/ejabf.2022.226167
Sambrook, J.; Gething, M. J. 1989. Protein structure Chaperones paperones. Nature. 342(6247): 224-225.
Silla, A. J.; Calatayud, N. E.; Trudeau, V. L. 2021. Amphibian reproductive technologies: approaches and welfare considerations. Conserv. Physiol. 9(1): coab011. https://doi.org/10.1093/ conphys/coab011
Stevens, E. C.; Hume, I. D. 1995. Digestion of carbohydrate, lipids and protein. In Comparative Physiology of the Vertebrate Digestive System. 152-171. Cambridge Academic Press 2nd ed.
Stone, G. G.; Newell, P.; Bradford, P. A. 2018. In vitro activity of ceftazidime-avibactam against isolates from patients in a phase 3 clinical trial for treatment of complicated intra-abdominal infections. Antimicrob. Agents. Chemother. 62(7): e02584-17. https://doi.org/10.1128/ aac.02584-17
Szopa, K.; Szajnar, K.; Pawlos, M.; Znamirowska-Piotrowska, A. 2023. Probiotic fermented goat’s and sheep’s milk: effect of type and dose of collagen on survival of four strains of probiotic bacteria during simulated in vitro digestion conditions. Nutrients. 15(14): 3241. doi:10.3390/nu15143241
Teixeira, R. D.; Mello, S. C. P.; dos Santos, C. A. L. 2001. The world market for frog legs. Food and Agriculture Organization of the United Nations, GLOBEFISH. Fishery Industries Division. 68: 1-44.
Timmons, P. B.; Hewage, C. M. 2021. Conformation and membrane interaction studies of the potent antimicrobial and anticancer peptide palustrin-Ca. Sci. Rep. 11(1): 22468. https://doi. org/10.1038/s41598-021-01769-3
Tong, Q.; Cui, L. Y.; Bie, J.; Han, X. Y.; Hu, Z. F.; Wang, H. B.; Zhang, J. T. 2021. Changes in the gut microbiota diversity of brown frogs (Rana dybowskii) after an antibiotic bath. BMC Vet. Res. 17(1): 1-13. https://doi.org/10.1186/s12917-021-03044-z
Vera Pingitore, E.; Bru, E.; Nader Macías, M. E. 2012. Effect of lyophilization and storage temperature on the activity of salivaricin CRL 1328, a potential bioactive ingredient of a urogenital probiotic product. J. Gen. Appl. Microbiol. 58(2): 71-81. https://doi.org/10.2323/ jgam.58.71
Wang, H.; Zhou, N.; Zhang, R.; Wu, Y.; Zhang, R.; Zhang, S. 2014. Identification and localization of gastrointestinal hormones in the skin of the bullfrog Rana catesbeiana during periods of activity and hibernation. Acta Histochem. 116(8): 1418-1426. https://doi.org/10.1016/j. acthis.2014.09.005
Yang, P.; Zheng, Y.; Zou, X.; Sun, Y.; Liu, Y. 2023. Comparative transcriptomic analysis of gene expression profiles in the liver and spleen of American bullfrog (Lithobates catesbeianus) in response to Citrobacter freundii infection. J. World Aquac. Soc. 55(1): 1-20. https://doi. org/10.1111/jwas.12999

Descargas
Publicado
Cómo citar
Número
Sección
Licencia

Esta obra está bajo una licencia internacional Creative Commons Reconocimiento-NoComercial-CompartirIgual 3.0.
Aquellos autores/as que tengan publicaciones con esta revista, aceptan las Políticas Editoriales.










.jpg)



